WholeBody Imaging of Multiple Myeloma Diagnostic Criteria Alpio

Whole-Body Imaging of Multiple Myeloma: Diagnostic Criteria Alípio G. Ormond Filho, MD 1; Bruno C. Carneiro, MD 1; Daniel Pastore, MD, Ph. D 1; Igor P. Silva, MD 1; Sâmia R. Yamashita, MD 1; Flávio D. Consolo, MD 1; Vânia T. M. Hungria, MD, Ph. D 2; Alex F. Sandes, MD, Ph. D 3; Edgar G. Rizzatti, MD, Ph. D 3; Marcelo A. C. Nico, MD 1 1 Department of Musculoskeletal Radiology, Fleury Medicina e Saúde Higienópolis, São Paulo, SP, Brazil 2 Department of Hematology, Clínica São Germano, São Paulo, SP, Brazil 3 Department of Hematology and Flow Cytometry, Fleury Medicina e Saúde Higienópolis, São Paulo, SP, Brazil

Whole-Body Imaging of Multiple Myeloma: Diagnostic Criteria Recipient of a Certificate of Merit award for an education exhibit at the 2017 RSNA Annual Meeting (MK 258 -ED-X). Address correspondence to Bruno C. Carneiro, MD Department of Musculoskeletal Radiology, Fleury Medicina e Saúde Higienópolis, Rua Mato Grosso 306, 1º Andar, Higienópolis, São Paulo, SP, Brazil 01239 -040 +55 11 3179 -0822, e-mail bruno. carneiro@grupofleury. com. br Acknowledgments: We acknowledge Xavier Stump, MD, for all of his years teaching the diseases of the musculoskeletal system to generations of radiologists and always helping our group solve the most difficult cases. We also thank Julio B. Guimarães, MD, Ph. D, for helping us with a variety of issues in this work, and Julie C. Dutoit, MD, Ph. D, for allowing us to use a figure from her article.
MULTIPLE MYELOMA (MM) ü Heterogeneous clonal plasma cell proliferative disease ü Primary infiltration of the bone marrow ü Second most common hematologic malignancy after lymphoma ü Most frequent malignancy involving the skeleton, with exception of metastasis ü Different whole-body (WB) imaging modalities indicated in distinct disease situations

TABLE of CONTENTS ü Summary of disease spectrum ü MM criteria scenario ü 2014 updated criteria ü Imaging modalities and their applications ü Treatment response evaluation ü Complications ü Conclusion

Learning Objectives 1. Describe the disease spectrum of MM 2. Discuss the updated diagnostic criteria and indications, advantages, and limitations of the WB imaging modalities 3. Identify and interpret the imaging findings of MM for diagnosis, follow-up, complications assessment, and treatment response evaluation

DISEASE SPECTRUM – MONOCLONAL GAMMOPATHIES MONOCLONAL GAMMOPATHY OF UNDETERMINED SIGNIFICANCE (MGUS) ü Asymptomatic stage ü Low rates of progression to MM ü Present in approximately 3%– 4% of the population older than 50 years SMOLDERING MULTIPLE MYELOMA (SMM) ü Intermediary stage ü Heterogeneous group with different rates of progression to MM MM ü End-organ damage, CRAB (hypercalcemia, renal failure, anemia, and bone lesions) symptoms ü Skeletal involvement in 80%– 90% of patients ü Complications Br J Haematol 2011; 154: 32 -75. Lancet Oncol 2014; 15: e 538 -e 548.

MM SCENARIO 2003 CRITERIA ü End-organ damage (ie, one or more CRAB features) needed for diagnosis ü High-risk SMM did not show any benefit from treatment • Toxic effects • Early intervention did not extend survival ü WB radiography is reference-standard imaging modality Hematol J 2003; 4(6): 379 -398. Lancet Oncol 2014; 15: e 538 -e 548.

MM SCENARIO 2014 CRITERIA ü Recognition of biomarkers that can be used to identify a subset of patients at very high risk of progression to active disease (once referred to as high-risk SMM) ü Major advance in treatment options • Safer and more effective treatments • Early intervention can extend survival ü Cross-sectional imaging is reference-standard approach Hematol J 2003; 4(6): 379 -398. Lancet Oncol 2014; 15: e 538 -e 548.
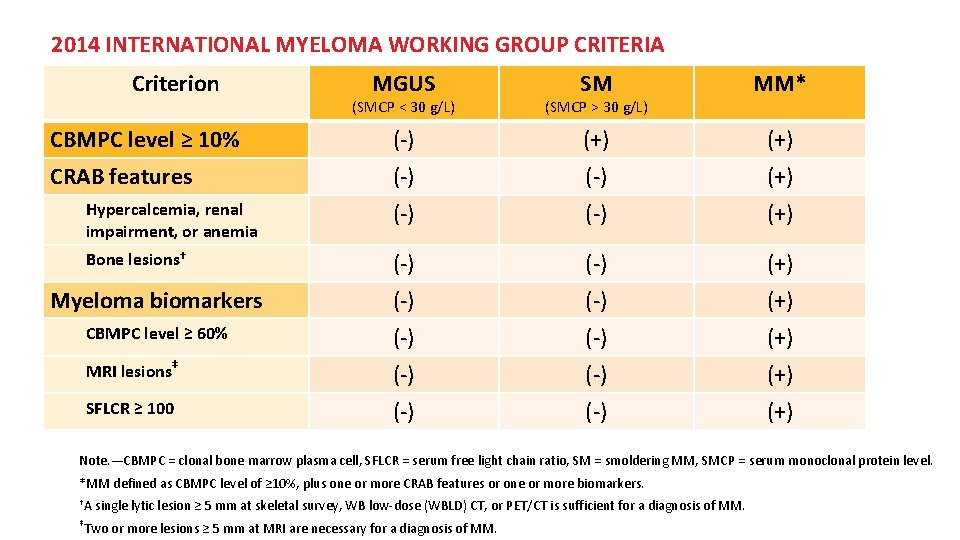
2014 INTERNATIONAL MYELOMA WORKING GROUP CRITERIA Criterion MGUS (SMCP < 30 g/L) SM (SMCP > 30 g/L) MM* CBMPC level ≥ 10% (-) (+) CRAB features (-) (+) Hypercalcemia, renal impairment, or anemia (-) (+) Bone lesions† (-) (+) Myeloma biomarkers (-) (+) CBMPC level ≥ 60% (-) (+) MRI lesions‡ (-) (+) SFLCR ≥ 100 (-) (+) Note. —CBMPC = clonal bone marrow plasma cell, SFLCR = serum free light chain ratio, SM = smoldering MM, SMCP = serum monoclonal protein level. *MM defined as CBMPC level of ≥ 10%, plus one or more CRAB features or one or more biomarkers. †A ‡ single lytic lesion ≥ 5 mm at skeletal survey, WB low-dose (WBLD) CT, or PET/CT is sufficient for a diagnosis of MM. Two or more lesions ≥ 5 mm at MRI are necessary for a diagnosis of MM.

IMAGING MODALITIES ü WB radiography ü WBLD CT ü WB MRI ü PET/CT

WB Radiography ü Previously the standard method for detecting bone lesions ü Low accuracy ü Requires 30%– 50% bone destruction for detection of a lesion ü 30%– 70% false-negative results ü Lower sensitivity in the axial skeleton ü Low accuracy for detection of complications ü No longer indicated unless it is the only option Haematologica 2015; 100(10): 1254 -1266.
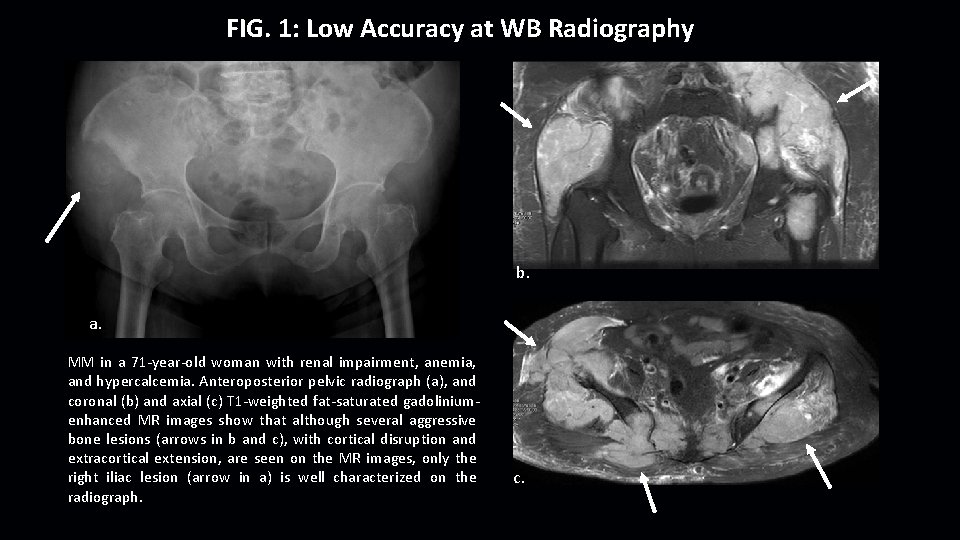
FIG. 1: Low Accuracy at WB Radiography b. a. MM in a 71 -year-old woman with renal impairment, anemia, and hypercalcemia. Anteroposterior pelvic radiograph (a), and coronal (b) and axial (c) T 1 -weighted fat-saturated gadoliniumenhanced MR images show that although several aggressive bone lesions (arrows in b and c), with cortical disruption and extracortical extension, are seen on the MR images, only the right iliac lesion (arrow in a) is well characterized on the radiograph. c.
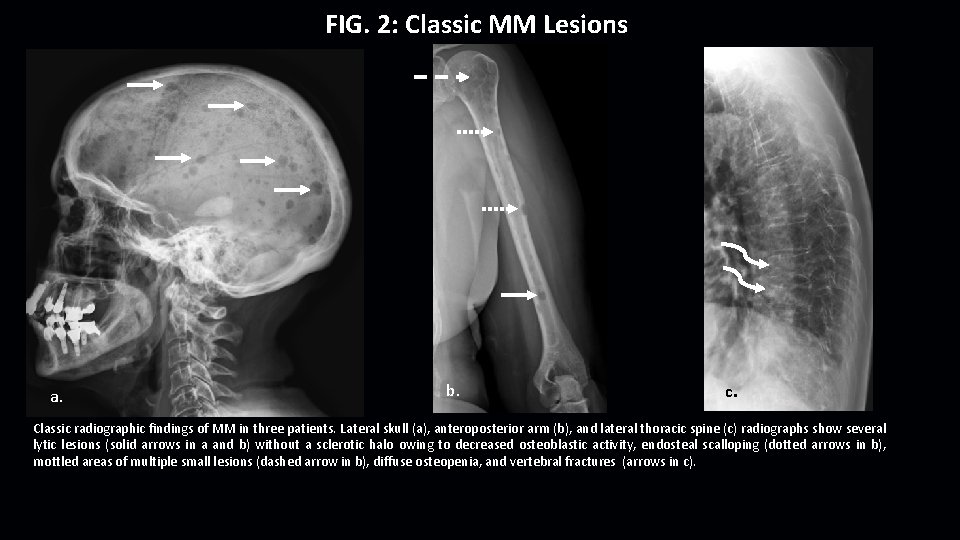
FIG. 2: Classic MM Lesions a. b. c. Classic radiographic findings of MM in three patients. Lateral skull (a), anteroposterior arm (b), and lateral thoracic spine (c) radiographs show several lytic lesions (solid arrows in a and b) without a sclerotic halo owing to decreased osteoblastic activity, endosteal scalloping (dotted arrows in b), mottled areas of multiple small lesions (dashed arrow in b), diffuse osteopenia, and vertebral fractures (arrows in c).

WBLD CT ü Significantly higher accuracy compared with WB radiography ü Involves better sensitivity, faster acquisition, and less expense compared with MRI and PET/CT ü Excellent interobserver correlation ü New standard initial procedure in the setting of MM according to European Myeloma Network ü Not so good for detection of extramedullary lesions ü Not indicated for treatment response evaluation Oncol Lett 2017; 13: 2490 -2494. Haematologica 2015; 100(10): 1254 -1266.
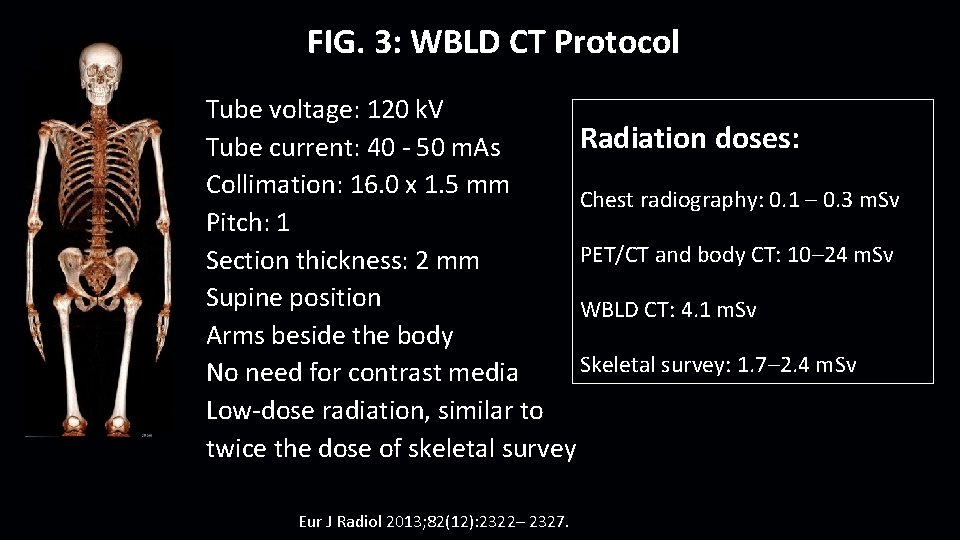
FIG. 3: WBLD CT Protocol Tube voltage: 120 k. V Radiation doses: Tube current: 40 - 50 m. As Collimation: 16. 0 x 1. 5 mm Chest radiography: 0. 1 – 0. 3 m. Sv Pitch: 1 PET/CT and body CT: 10– 24 m. Sv Section thickness: 2 mm Supine position WBLD CT: 4. 1 m. Sv Arms beside the body Skeletal survey: 1. 7– 2. 4 m. Sv No need for contrast media Low-dose radiation, similar to twice the dose of skeletal survey Eur J Radiol 2013; 82(12): 2322– 2327.
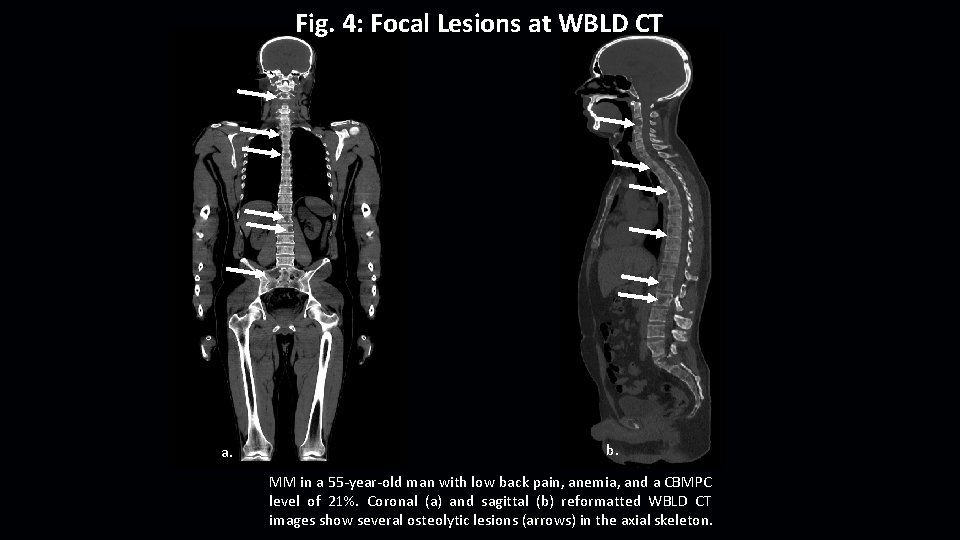
Fig. 4: Focal Lesions at WBLD CT a. b. MM in a 55 -year-old man with low back pain, anemia, and a CBMPC level of 21%. Coronal (a) and sagittal (b) reformatted WBLD CT images show several osteolytic lesions (arrows) in the axial skeleton.
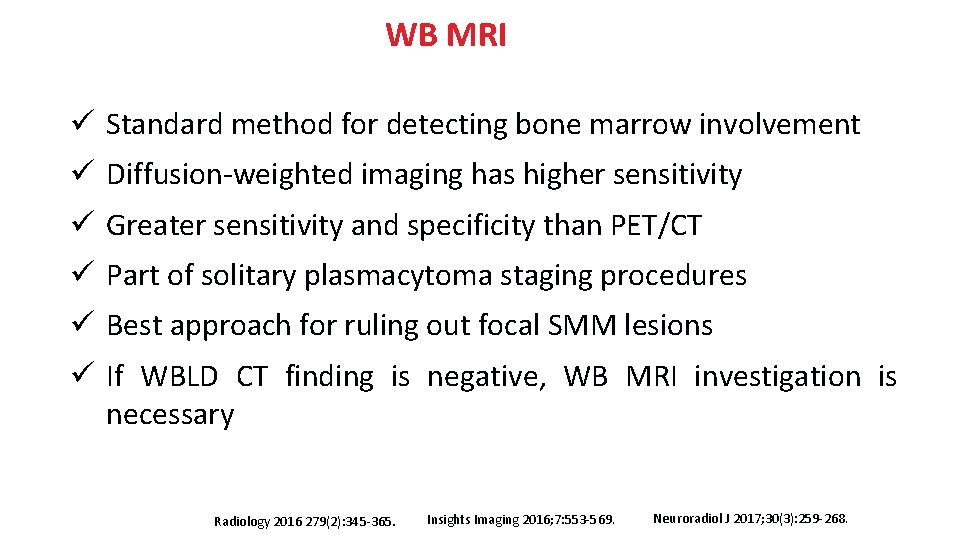
WB MRI ü Standard method for detecting bone marrow involvement ü Diffusion-weighted imaging has higher sensitivity ü Greater sensitivity and specificity than PET/CT ü Part of solitary plasmacytoma staging procedures ü Best approach for ruling out focal SMM lesions ü If WBLD CT finding is negative, WB MRI investigation is necessary Radiology 2016 279(2): 345 -365. Insights Imaging 2016; 7: 553 -569. Neuroradiol J 2017; 30(3): 259 -268.

WB MRI ü Has prognostic value ü Not as effective as PET/CT for treatment response assessment ü Standard procedure for evaluation of painful complications ü Contrast media not routinely necessary ü Whole-spine MRI and pelvic MRI also are options if WB MRI is not available Radiology 2016; 279(2): 345 -365. Insights Imaging 2016; 7: 553 -569. Neuroradiol J 2017; 30(3): 259 -268.
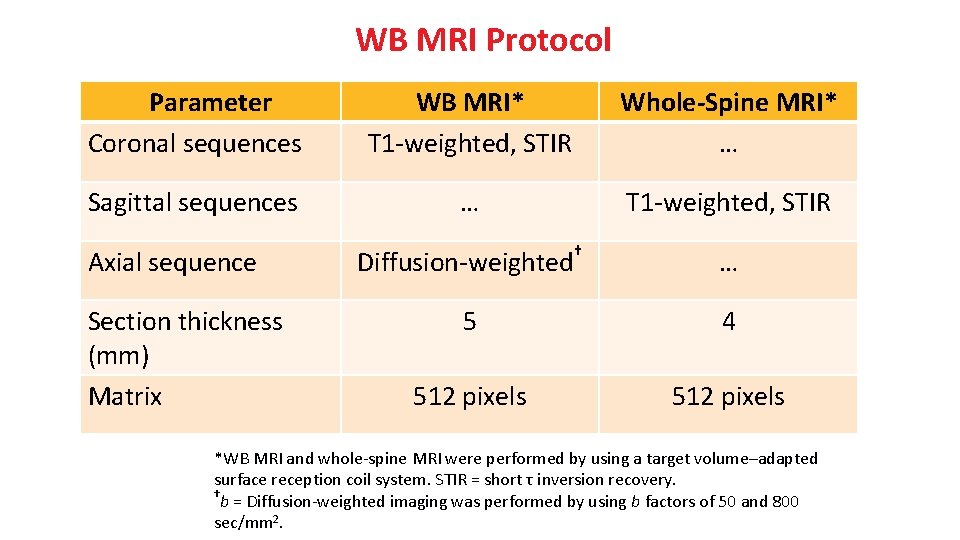
WB MRI Protocol Parameter Coronal sequences WB MRI* T 1 -weighted, STIR Whole-Spine MRI* … Sagittal sequences … T 1 -weighted, STIR Diffusion-weighted† … 5 4 512 pixels Axial sequence Section thickness (mm) Matrix *WB MRI and whole-spine MRI were performed by using a target volume–adapted surface reception coil system. STIR = short τ inversion recovery. † b = Diffusion-weighted imaging was performed by using b factors of 50 and 800 sec/mm 2.
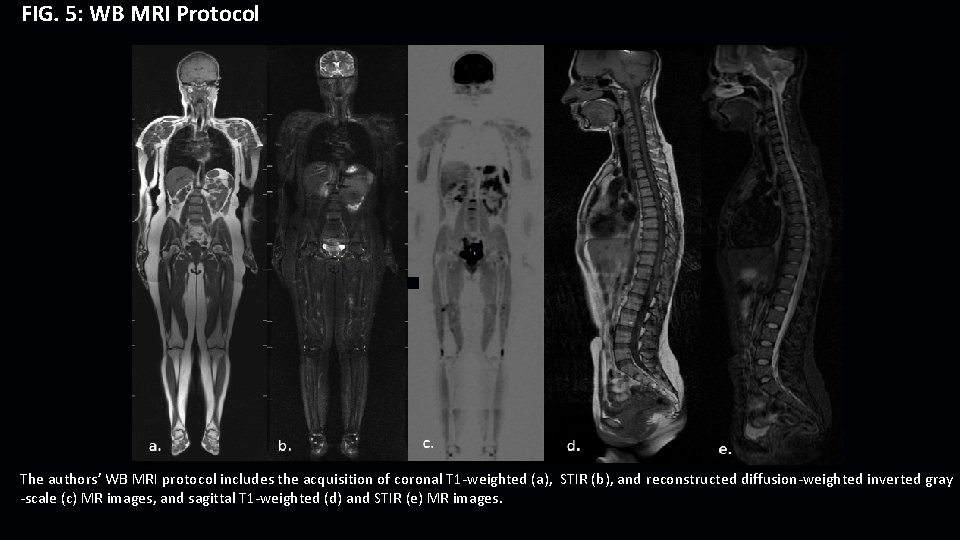
FIG. 5: WB MRI Protocol The authors’ WB MRI protocol includes the acquisition of coronal T 1 -weighted (a), STIR (b), and reconstructed diffusion-weighted inverted gray -scale (c) MR images, and sagittal T 1 -weighted (d) and STIR (e) MR images.
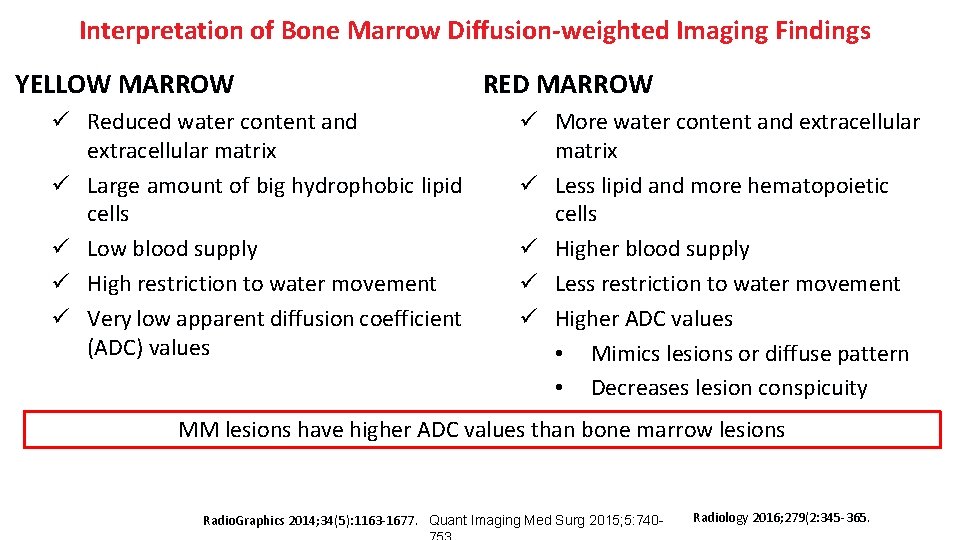
Interpretation of Bone Marrow Diffusion-weighted Imaging Findings YELLOW MARROW ü Reduced water content and extracellular matrix ü Large amount of big hydrophobic lipid cells ü Low blood supply ü High restriction to water movement ü Very low apparent diffusion coefficient (ADC) values RED MARROW ü More water content and extracellular matrix ü Less lipid and more hematopoietic cells ü Higher blood supply ü Less restriction to water movement ü Higher ADC values • Mimics lesions or diffuse pattern • Decreases lesion conspicuity MM lesions have higher ADC values than bone marrow lesions Radio. Graphics 2014; 34(5): 1163 -1677. Quant Imaging Med Surg 2015; 5: 740 - Radiology 2016; 279(2: 345 -365.
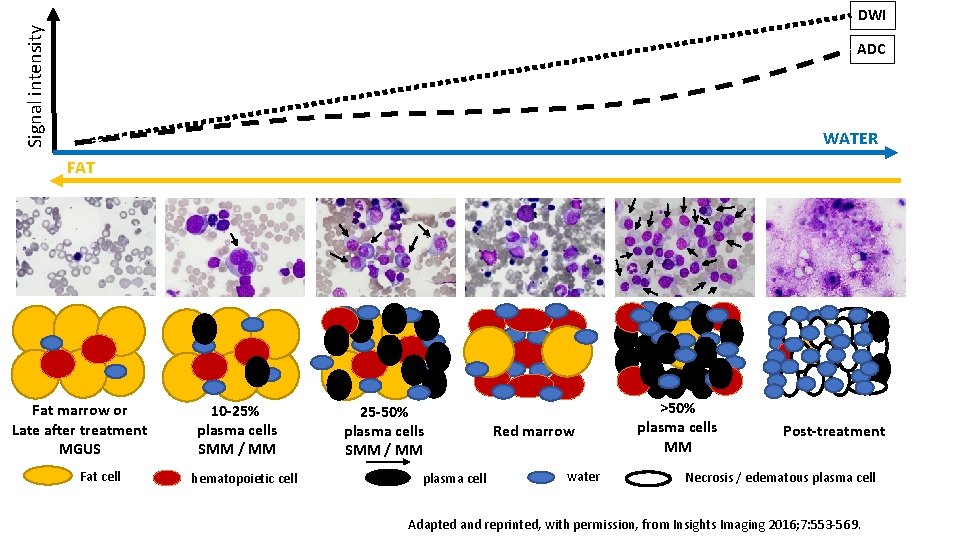
Signal intensity DWI ADC WATER FAT Fat marrow or Late after treatment MGUS Fat cell 10 -25% plasma cells SMM / MM hematopoietic cell 25 -50% plasma cells SMM / MM plasma cell Red marrow water >50% plasma cells MM Post-treatment Necrosis / edematous plasma cell Adapted and reprinted, with permission, from Insights Imaging 2016; 7: 553 -569.
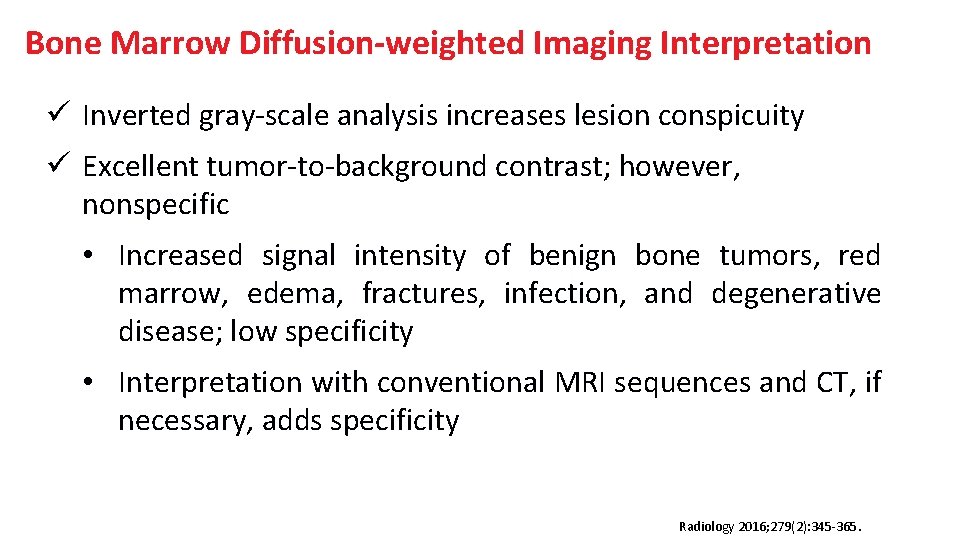
Bone Marrow Diffusion-weighted Imaging Interpretation ü Inverted gray-scale analysis increases lesion conspicuity ü Excellent tumor-to-background contrast; however, nonspecific • Increased signal intensity of benign bone tumors, red marrow, edema, fractures, infection, and degenerative disease; low specificity • Interpretation with conventional MRI sequences and CT, if necessary, adds specificity Radiology 2016; 279(2): 345 -365.
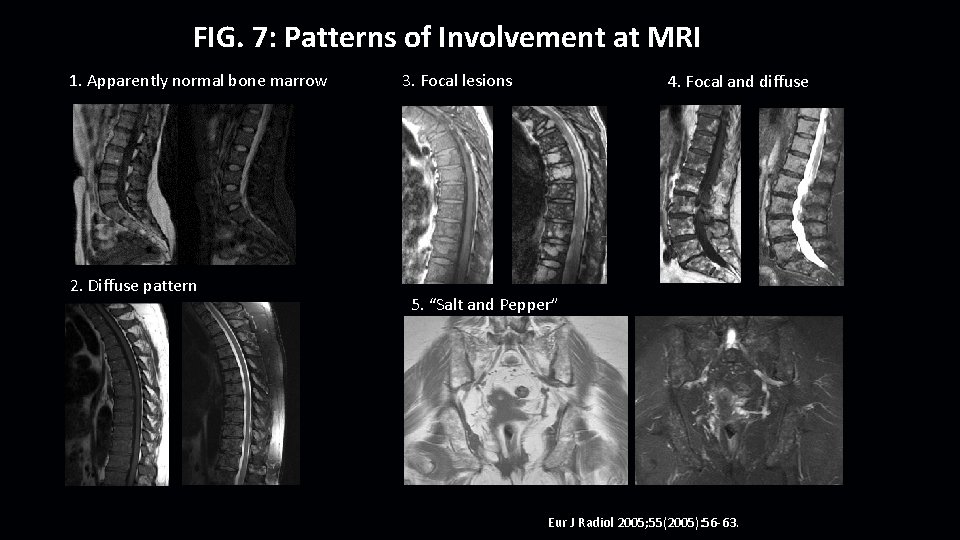
FIG. 7: Patterns of Involvement at MRI 1. Apparently normal bone marrow 2. Diffuse pattern 3. Focal lesions 4. Focal and diffuse 5. “Salt and Pepper” Eur J Radiol 2005; 55(2005): 56 -63.
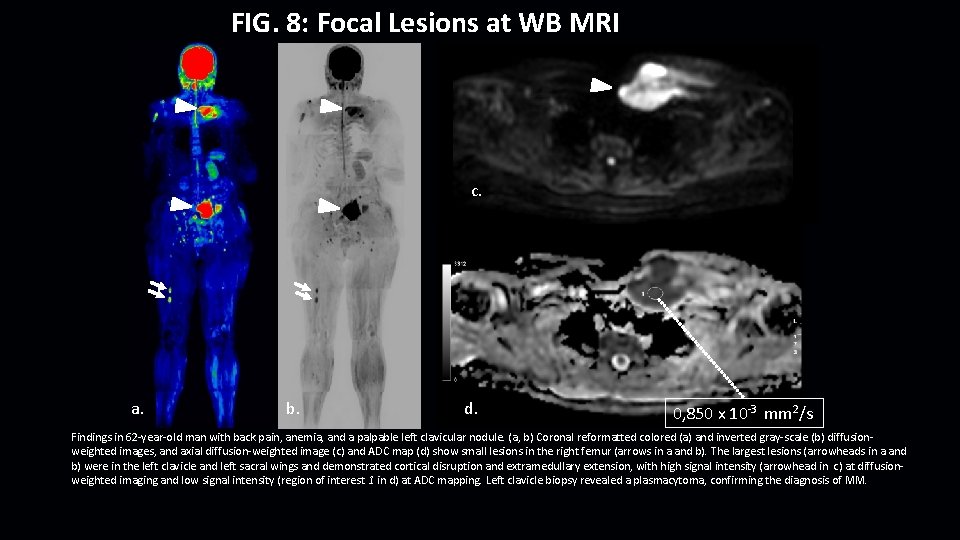
FIG. 8: Focal Lesions at WB MRI c. a. b. d. 0, 850 x 10 -3 mm 2/s Findings in 62 -year-old man with back pain, anemia, and a palpable left clavicular nodule. (a, b) Coronal reformatted colored (a) and inverted gray-scale (b) diffusionweighted images, and axial diffusion-weighted image (c) and ADC map (d) show small lesions in the right femur (arrows in a and b). The largest lesions (arrowheads in a and b) were in the left clavicle and left sacral wings and demonstrated cortical disruption and extramedullary extension, with high signal intensity (arrowhead in c) at diffusionweighted imaging and low signal intensity (region of interest 1 in d) at ADC mapping. Left clavicle biopsy revealed a plasmacytoma, confirming the diagnosis of MM.
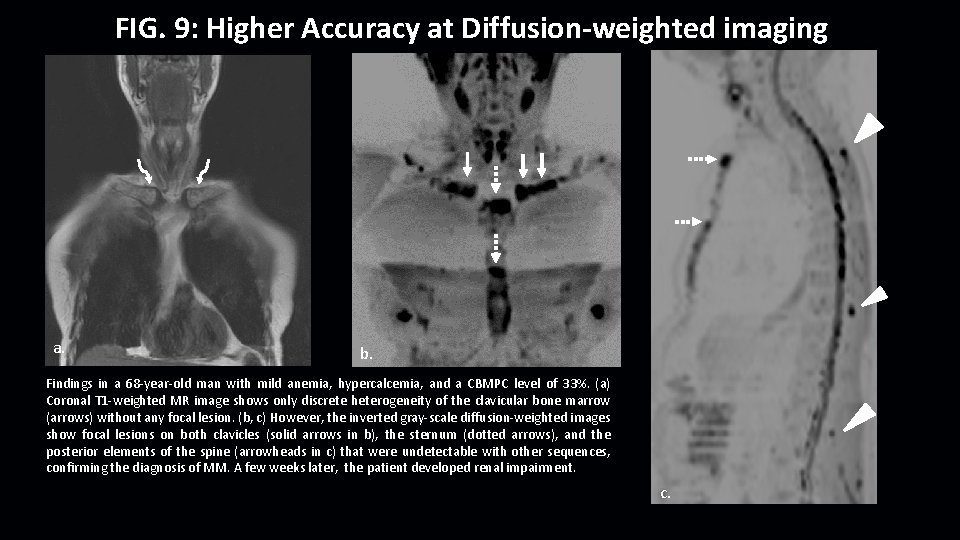
FIG. 9: Higher Accuracy at Diffusion-weighted imaging a. b. Findings in a 68 -year-old man with mild anemia, hypercalcemia, and a CBMPC level of 33%. (a) Coronal T 1 -weighted MR image shows only discrete heterogeneity of the clavicular bone marrow (arrows) without any focal lesion. (b, c) However, the inverted gray-scale diffusion-weighted images show focal lesions on both clavicles (solid arrows in b), the sternum (dotted arrows), and the posterior elements of the spine (arrowheads in c) that were undetectable with other sequences, confirming the diagnosis of MM. A few weeks later, the patient developed renal impairment. c.
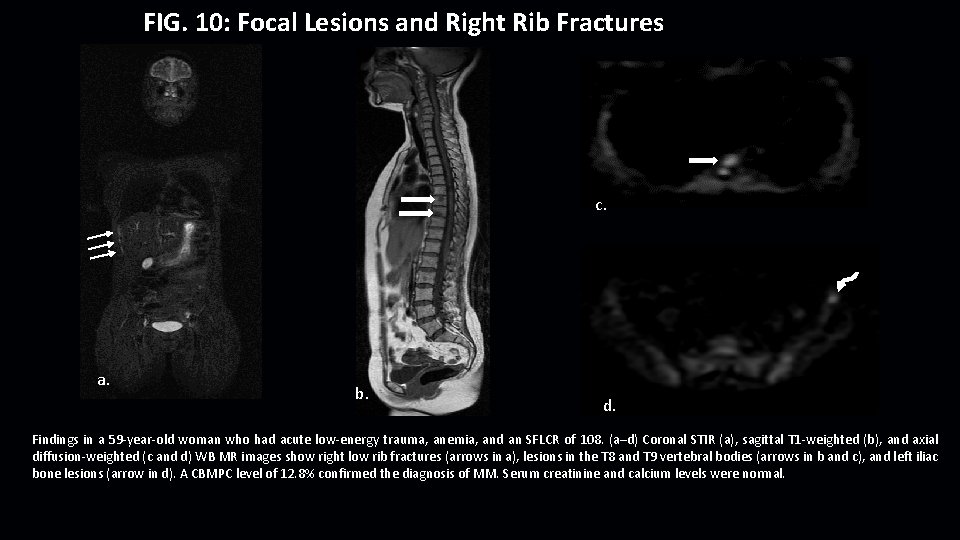
FIG. 10: Focal Lesions and Right Rib Fractures c. a. b. d. Findings in a 59 -year-old woman who had acute low-energy trauma, anemia, and an SFLCR of 108. (a–d) Coronal STIR (a), sagittal T 1 -weighted (b), and axial diffusion-weighted (c and d) WB MR images show right low rib fractures (arrows in a), lesions in the T 8 and T 9 vertebral bodies (arrows in b and c), and left iliac bone lesions (arrow in d). A CBMPC level of 12. 8% confirmed the diagnosis of MM. Serum creatinine and calcium levels were normal.
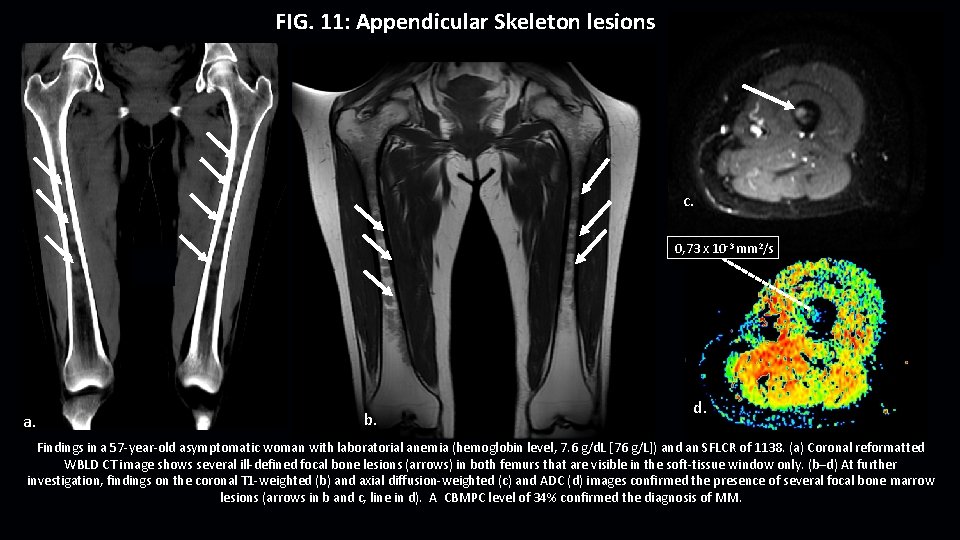
FIG. 11: Appendicular Skeleton lesions c. 0, 73 x 10 -3 mm 2/s a. b. d. Findings in a 57 -year-old asymptomatic woman with laboratorial anemia (hemoglobin level, 7. 6 g/d. L [76 g/L]) and an SFLCR of 1138. (a) Coronal reformatted WBLD CT image shows several ill-defined focal bone lesions (arrows) in both femurs that are visible in the soft-tissue window only. (b–d) At further investigation, findings on the coronal T 1 -weighted (b) and axial diffusion-weighted (c) and ADC (d) images confirmed the presence of several focal bone marrow lesions (arrows in b and c, line in d). A CBMPC level of 34% confirmed the diagnosis of MM.
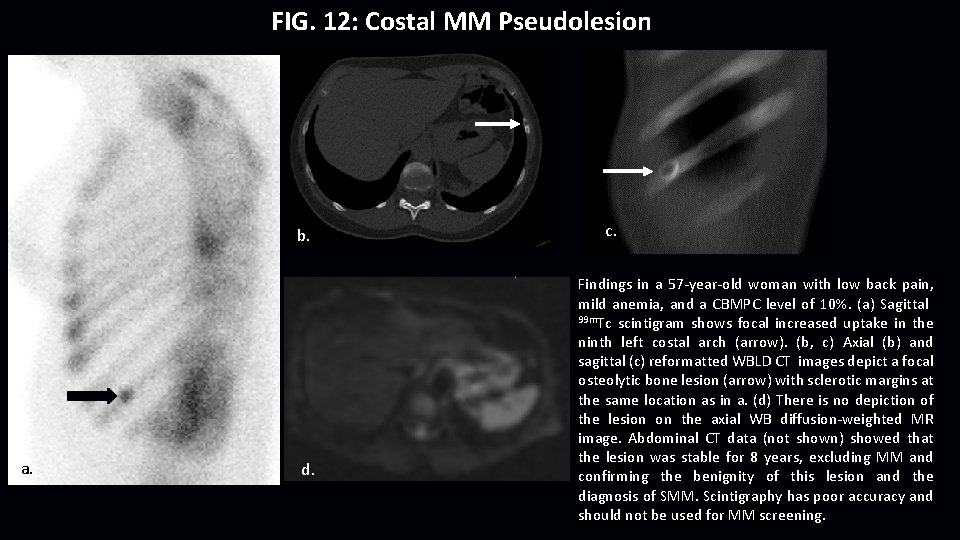
FIG. 12: Costal MM Pseudolesion b. a. d. c. Findings in a 57 -year-old woman with low back pain, mild anemia, and a CBMPC level of 10%. (a) Sagittal 99 m. Tc scintigram shows focal increased uptake in the ninth left costal arch (arrow). (b, c) Axial (b) and sagittal (c) reformatted WBLD CT images depict a focal osteolytic bone lesion (arrow) with sclerotic margins at the same location as in a. (d) There is no depiction of the lesion on the axial WB diffusion-weighted MR image. Abdominal CT data (not shown) showed that the lesion was stable for 8 years, excluding MM and confirming the benignity of this lesion and the diagnosis of SMM. Scintigraphy has poor accuracy and should not be used for MM screening.
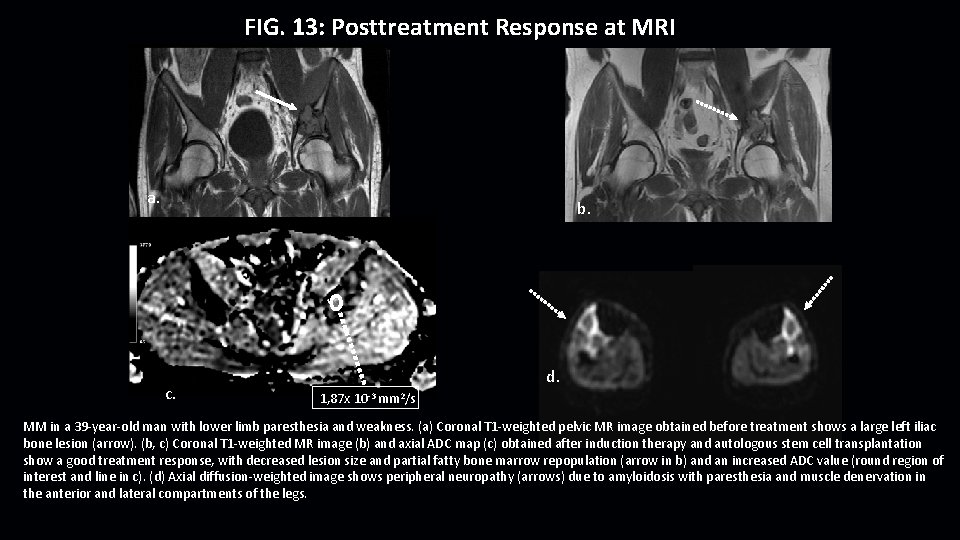
FIG. 13: Posttreatment Response at MRI a. b. c. d. 1, 87 x 10 -3 mm 2/s MM in a 39 -year-old man with lower limb paresthesia and weakness. (a) Coronal T 1 -weighted pelvic MR image obtained before treatment shows a large left iliac bone lesion (arrow). (b, c) Coronal T 1 -weighted MR image (b) and axial ADC map (c) obtained after induction therapy and autologous stem cell transplantation show a good treatment response, with decreased lesion size and partial fatty bone marrow repopulation (arrow in b) and an increased ADC value (round region of interest and line in c). (d) Axial diffusion-weighted image shows peripheral neuropathy (arrows) due to amyloidosis with paresthesia and muscle denervation in the anterior and lateral compartments of the legs.

PET/CT ü Best tool for posttreatment evaluation ü Has prognostic relevance ü Good option for disease diagnosis ü High sensitivity for detection of bone lesions and extramedullary involvement ü Sufficient for excluding focal lesions if WB MRI is not available ü Diffuse FDG uptake does not meet the criteria Lancet Oncol 2017; 18: e 206 -e 217.
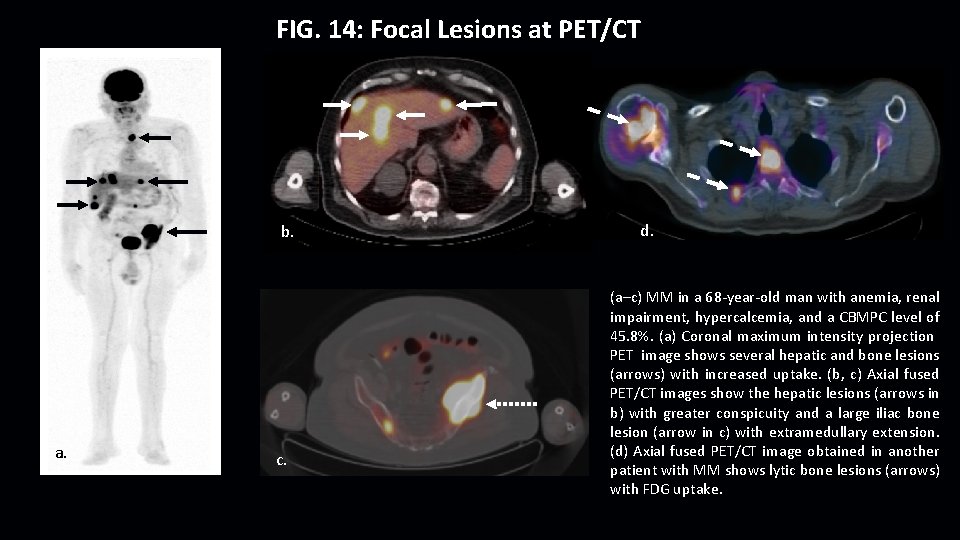
FIG. 14: Focal Lesions at PET/CT b. a. c. d. (a–c) MM in a 68 -year-old man with anemia, renal impairment, hypercalcemia, and a CBMPC level of 45. 8%. (a) Coronal maximum intensity projection PET image shows several hepatic and bone lesions (arrows) with increased uptake. (b, c) Axial fused PET/CT images show the hepatic lesions (arrows in b) with greater conspicuity and a large iliac bone lesion (arrow in c) with extramedullary extension. f. Axial fused PET/CT image obtained in another (d) patient with MM shows lytic bone lesions (arrows) with FDG uptake.
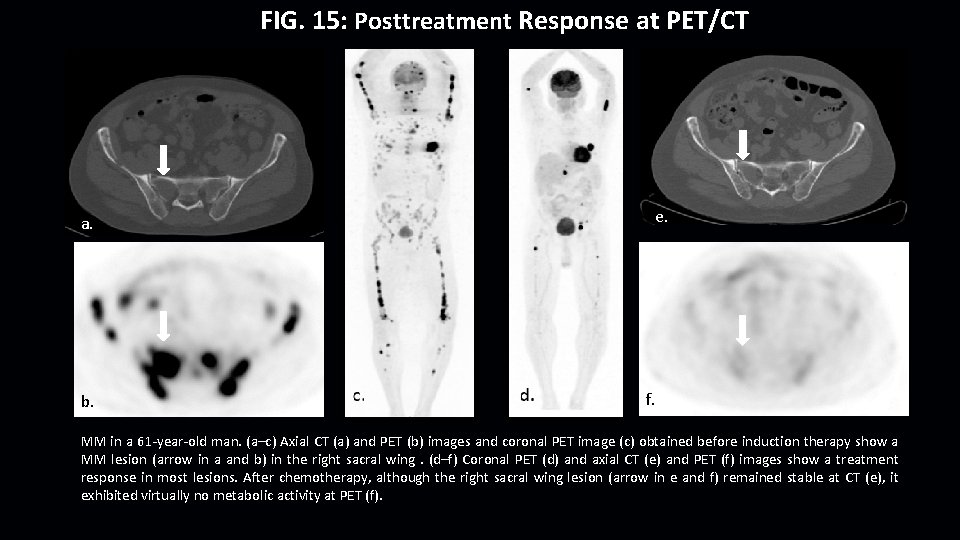
FIG. 15: Posttreatment Response at PET/CT e. a. b. f. MM in a 61 -year-old man. (a–c) Axial CT (a) and PET (b) images and coronal PET image (c) obtained before induction therapy show a MM lesion (arrow in a and b) in the right sacral wing. (d–f) Coronal PET (d) and axial CT (e) and PET (f) images show a treatment response in most lesions. After chemotherapy, although the right sacral wing lesion (arrow in e and f) remained stable at CT (e), it exhibited virtually no metabolic activity at PET (f).

COMPLICATIONS ü Infections are the main cause of death ü Vertebral fractures may be neoplastic or osteoporotic ü MRI can differentiate between these ü Spinal cord or nerve root compression ü Fractures or extramedullary tumor extension ü Osteonecrosis: usually treatment related ü Meningeal myelomatosis ü Neuropathy ü Venous thrombosis and/or thromboembolism
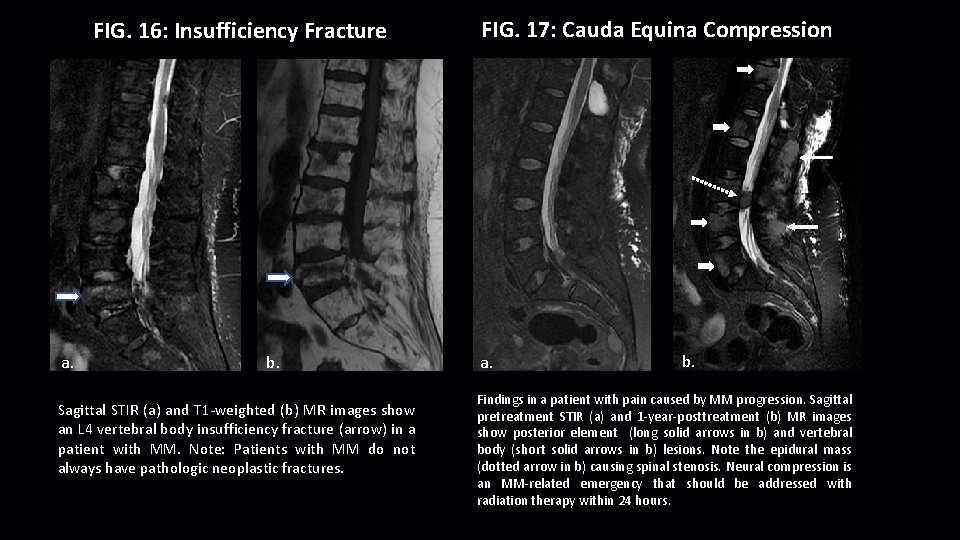
FIG. 16: Insufficiency Fracture a. b. Sagittal STIR (a) and T 1 -weighted (b) MR images show an L 4 vertebral body insufficiency fracture (arrow) in a patient with MM. Note: Patients with MM do not always have pathologic neoplastic fractures. FIG. 17: Cauda Equina Compression a. b. Findings in a patient with pain caused by MM progression. Sagittal pretreatment STIR (a) and 1 -year-posttreatment (b) MR images show posterior element (long solid arrows in b) and vertebral body (short solid arrows in b) lesions. Note the epidural mass (dotted arrow in b) causing spinal stenosis. Neural compression is an MM-related emergency that should be addressed with radiation therapy within 24 hours.
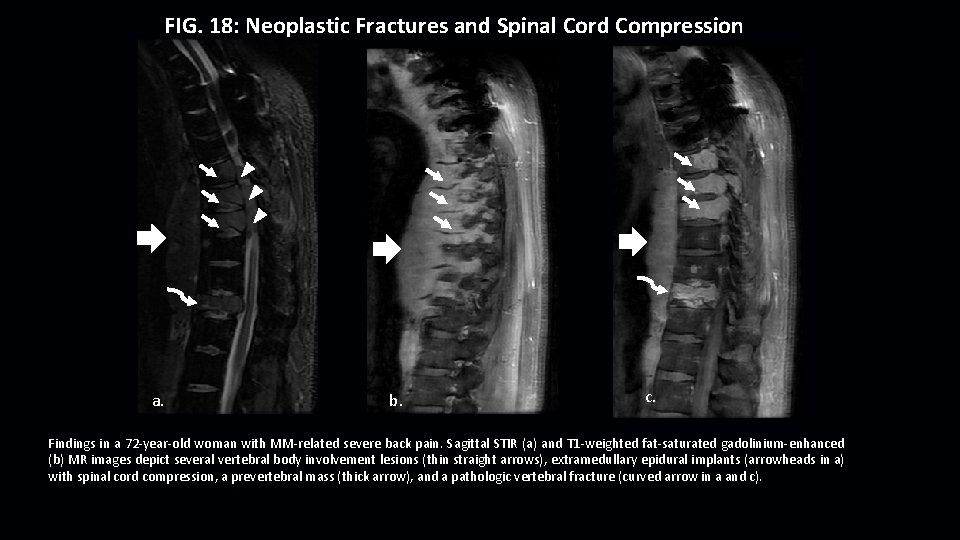
FIG. 18: Neoplastic Fractures and Spinal Cord Compression a. b. c. Findings in a 72 -year-old woman with MM-related severe back pain. Sagittal STIR (a) and T 1 -weighted fat-saturated gadolinium-enhanced (b) MR images depict several vertebral body involvement lesions (thin straight arrows), extramedullary epidural implants (arrowheads in a) with spinal cord compression, a prevertebral mass (thick arrow), and a pathologic vertebral fracture (curved arrow in a and c).
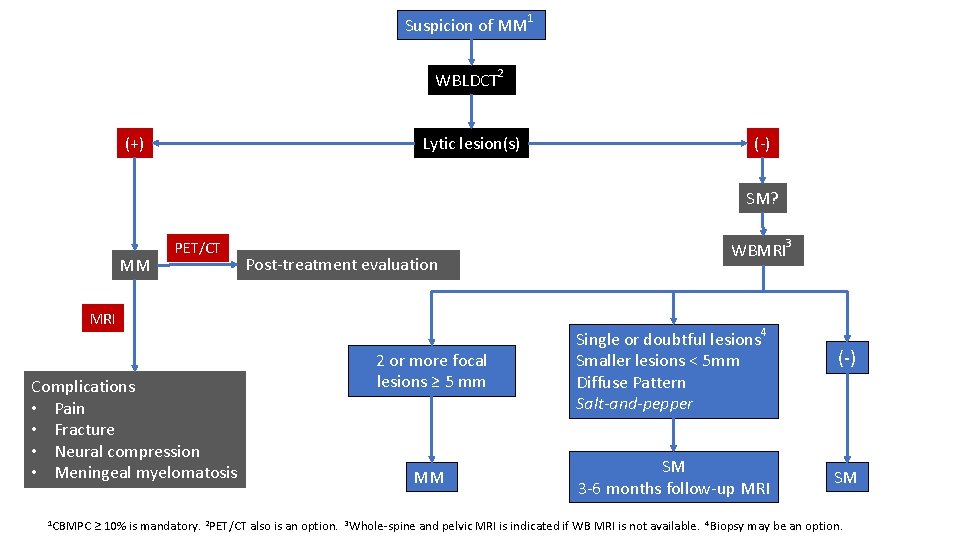
Suspicion of MM 1 WBLDCT 2 (+) Lytic lesion(s) (-) SM? MM PET/CT Post-treatment evaluation MRI Complications • Pain • Fracture • Neural compression • Meningeal myelomatosis 1 CBMPC WBMRI 3 2 or more focal lesions ≥ 5 mm Single or doubtful lesions 4 Smaller lesions < 5 mm Diffuse Pattern Salt-and-pepper MM SM 3 -6 months follow-up MRI (-) SM ≥ 10% is mandatory. 2 PET/CT also is an option. 3 Whole-spine and pelvic MRI is indicated if WB MRI is not available. 4 Biopsy may be an option.

Conclusions ü WB radiography is a poor option unless it is the only choice. ü WBLD CT is the initial standard imaging procedure. ü WB MRI is the most sensitive imaging modality for detecting “early” MM and excluding SMM bone lesions. ü Standard MRI is the method of choice for evaluation of painful complications. ü PET/CT is the best tool for assessment of treatment response.

Conclusions ü 2014 International Myeloma Working Group updated criteria • Major role of radiologists in diagnosis • Greater importance of imaging, with different WB techniques indicated in distinct disease scenarios ü Radiologists must know the disease criteria, advantages and limitations of each imaging modality, and recommendations for early follow-up and treatment response evaluation.

Suggested Readings • Baur-Melnyk A, Buhmann S, Dürr HR, Reiser M. Role of MRI for the diagnosis and prognosis of multiple myeloma. Eur J Radiol 2005; 55(1): 56– 63. • Bhojwani N, Szpakowski P, Partovi S. Diffusion-weighted imaging in musculoskeletal radiologyclinical applications and future directions. Quant Imaging Med Surg 2015; 5(5): 740– 753. • Bird JM, Owen RG, D'Sa S, et al. Guidelines for the diagnosis and management of multiple myeloma 2011. Br J Haematol 2011; 154(1): 32– 75. • Cavo M, Terpos E, Nanni C, et al. Role of 18 F-FDG PET/CT in the diagnosis and management of multiple myeloma and other plasma cell disorders: a consensus statement by the International Myeloma Working Group. Lancet Oncol 2017; 18(4): e 206–e 217. • Dimopoulos MA, Hillengass J, Usmani S, et al. Role of magnetic resonance imaging in the management of patients with multiple myeloma: a consensus statement. J Clin Oncol 2015; 33(6): 657– 664.

Suggested Readings • Durie BG, Kyle RA, Belch A, et al. Myeloma management guidelines: a consensus report from the Scientific Advisors of the International Myeloma Foundation. Hematol J 2003; 4(6): 379– 398. [Published correction appears in Hematol J 2004; 5(3): 285. ] https: //doi. org/10. 1038/sj. thj. 6200312. • Dutoit JC, Verstraete KL. MRI in multiple myeloma: a pictorial review of diagnostic and post-treatment findings. Insights Imaging 2016; 7: 553– 569. • Ippolito D, Besostri V, Bonaffini PA, Rossini F, Di Lelio A, Sironi S. Diagnostic value of whole-body low-dose computed tomography (WBLDCT) in bone lesions detection in patients with multiple myeloma (MM). Eur J Radiol 2013; 82(12): 2322– 2327. • Lambert L, Ourednicek P, Meckova Z, Gavelli G, Straub J, Spicka I. Whole-body lowdose computed tomography in multiple myeloma staging: superior diagnostic performance in the detection of bone lesions, vertebral compression fractures, rib fractures and extraskeletal findings compared to radiography with similar radiation exposure. Oncol Lett 2017; 13(4): 2490– 2494.

Suggested Readings • Lasocki A, Gaillard F, Harrison SJ. Multiple myeloma of the spine. Neuroradiol J 2017; 30(3): 259– 268. • Lecouvet FE. Whole-Body MR Imaging: Musculoskeletal Applications. Radiology 2016; 279(2): 345– 365. • Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014; 15: e 538–e 548. • Subhawong TK, Jacobs MA, Fayad LM. Diffusion-weighted MR imaging for characterizing musculoskeletal lesions. Radio. Graphics 2014; 34(5): 1163– 1177. • Terpos E, Kleber M, Engelhardt M, et al. European Myeloma Network guidelines for the management of multiple myeloma-related complications. Haematologica 2015; 100(10): 1254– 1266.
- Slides: 42