Who was Democritus How did Dalton describe atoms

Who was Democritus? How did Dalton describe atoms? Greek model (400 B. C. )

DALTON’S ATOMIC THEORY OF MATTER His atomic theory of matter contains four hypotheses: Elements are made of tiny particles called atoms. All atoms of a given element are identical. The atoms of a given element are different from those of any other element Atoms of one element can combine chemically with atoms of another element to form compounds. A given compound always has the same relative number and types of atoms. Atoms are indivisible in chemical processes. Atoms are not created or destroyed in chemical reactions. A chemical reaction simply changes the way the atoms Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
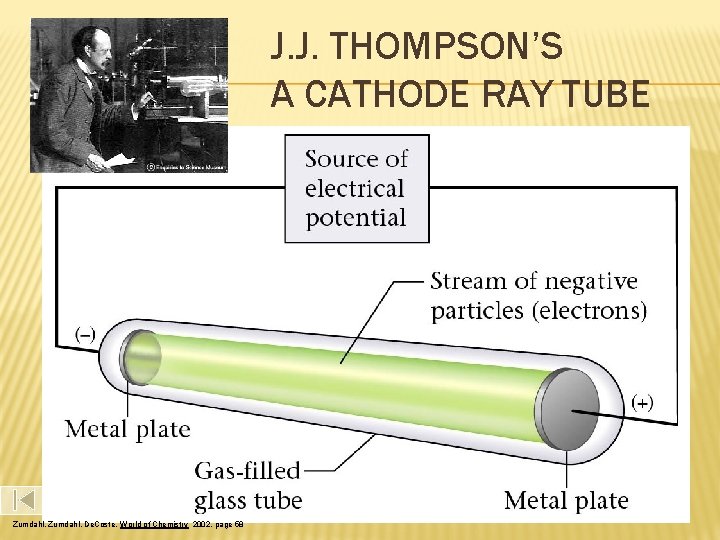
J. J. THOMPSON’S A CATHODE RAY TUBE Zumdahl, De. Coste, World of Chemistry 2002, page 58

CATHODE RAY EXPERIMENT Thompson’s 1897 Experiment Using a cathode ray tube, Thomson was able to deflect cathode rays with an electrical field. The rays bent towards the positive pole, indicating that they are negatively charged.

THOMSON MODEL OF THE ATOM J. J. Thomson discovered the electron and knew that electrons could be emitted from matter (1897). Thomson proposed that atoms consist of small, negative electrons embedded in a massive, positive sphere. The electrons were like raisins in a plum pudding, called the ‘plum pudding’ model of the atom.
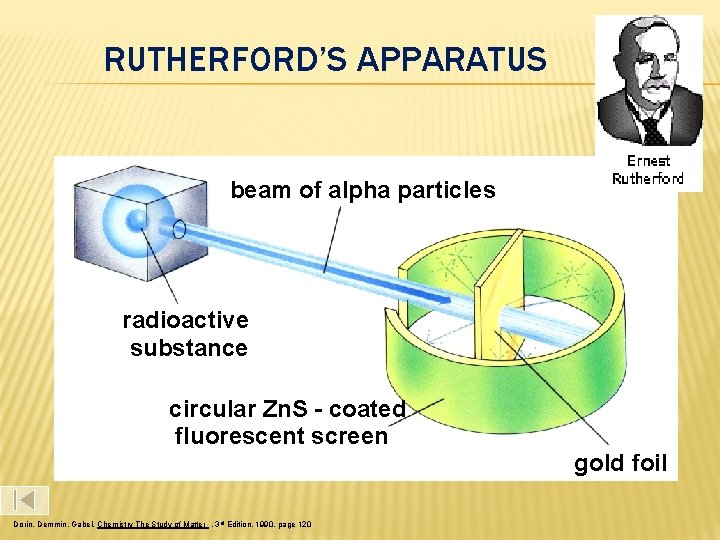
RUTHERFORD’S APPARATUS beam of alpha particles radioactive substance circular Zn. S - coated fluorescent screen gold foil Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 120

Rutherford Model In 1911, Ernest Rutherford conducted his gold foil experiment which helped improve our understanding of atomic structure. He directed a narrow beam of alpha particles at a very thin sheet of gold foil. particles (a) are He atoms that have been stripped of their electrons Alpha
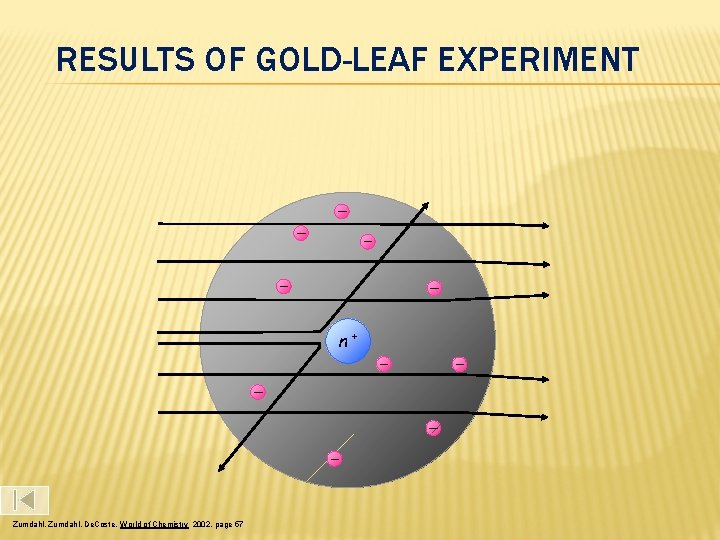
RESULTS OF GOLD-LEAF EXPERIMENT n+ Zumdahl, De. Coste, World of Chemistry 2002, page 57
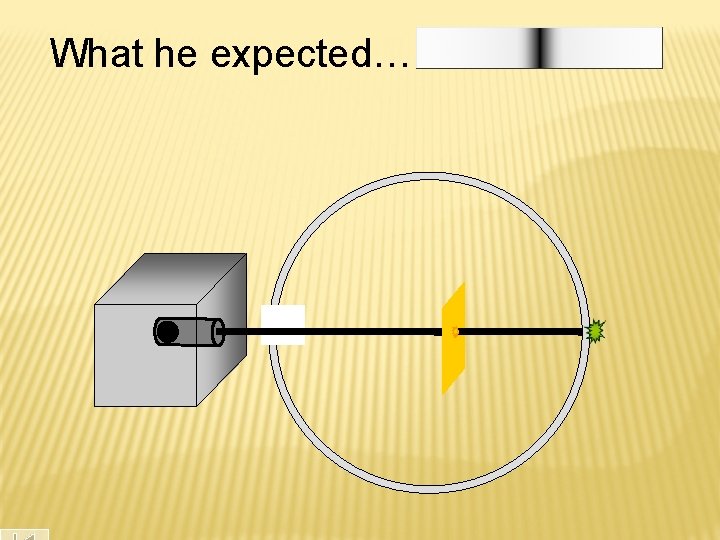
What he expected…
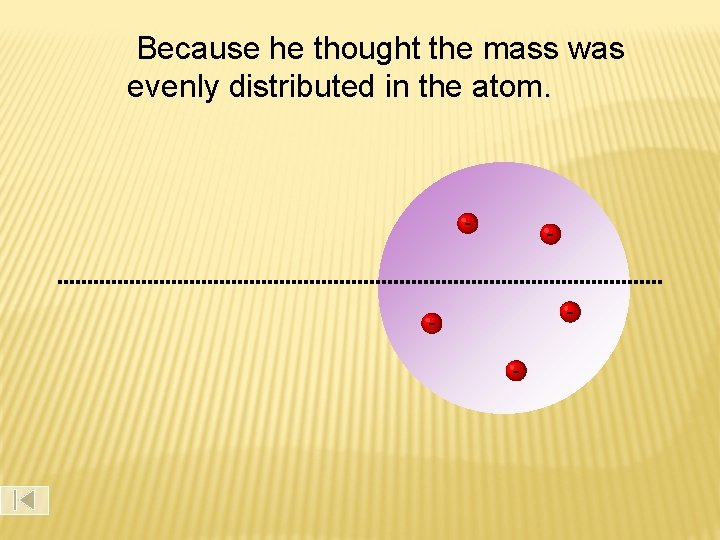
Because he thought the mass was evenly distributed in the atom. - -
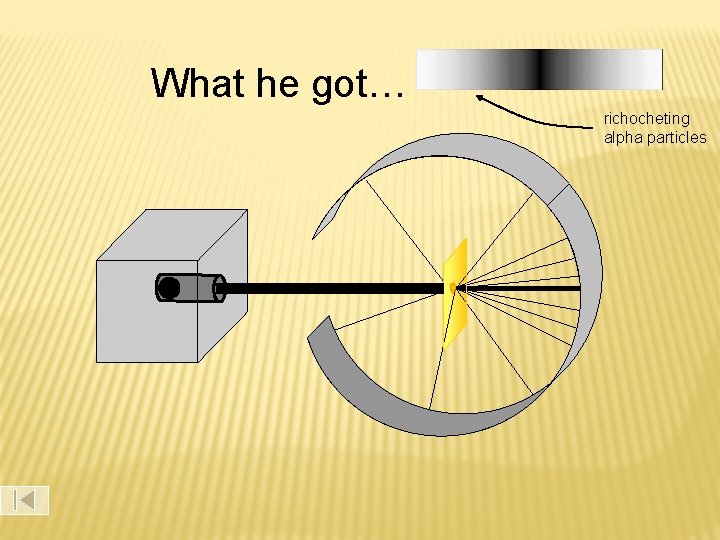
What he got… richocheting alpha particles

According to Thomson’s model, the heavy, positive alpha particles should pass easily through the gold, with only a slight deflection And mostly that’s how it happened. However, they found 1 in every 8000 particles had actually been deflected back toward the source. Rutherford Model


Rutherford suggested a new structural model of the atom. He stated that all the positive charge and the mass is concentrated in a small core in the center of the atom, AKA nucleus And that the atom is mostly empty space with electrons surrounding the positively charged nucleus like planets around the sun. - + Rutherford’s model (1909) Rutherford Model
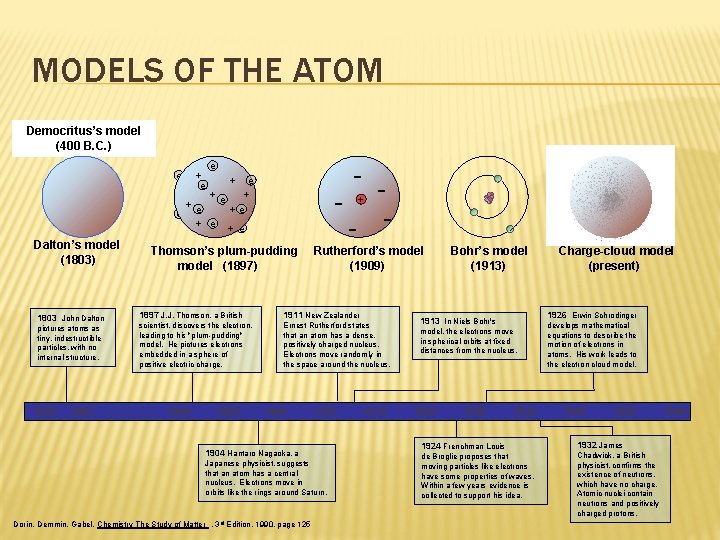
MODELS OF THE ATOM Democritus’s model (400 B. C. ) e + e e - + e +e +e e + e Dalton’s model (1803) 1803 John Dalton pictures atoms as tiny, indestructible particles, with no internal structure. 1800 - Thomson’s plum-pudding model (1897) - + Rutherford’s model (1909) 1897 J. J. Thomson, a British 1911 New Zealander scientist, discovers the electron, leading to his "plum-pudding" model. He pictures electrons embedded in a sphere of positive electric charge. Ernest Rutherford states that an atom has a dense, positively charged nucleus. Electrons move randomly in the space around the nucleus. 1805. . . . . 1895 1900 1905 1910 1904 Hantaro Nagaoka, a Japanese physicist, suggests that an atom has a central nucleus. Electrons move in orbits like the rings around Saturn. Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 125 1915 Bohr’s model (1913) 1926 Erwin Schrodinger 1913 In Niels Bohr's model, the electrons move in spherical orbits at fixed distances from the nucleus. 1920 1925 Charge-cloud model (present) 1930 1924 Frenchman Louis de Broglie proposes that moving particles like electrons have some properties of waves. Within a few years evidence is collected to support his idea. develops mathematical equations to describe the motion of electrons in atoms. His work leads to the electron cloud model. 1935 1940 1932 James Chadwick, a British physicist, confirms the existence of neutrons, which have no charge. Atomic nuclei contain neutrons and positively charged protons. 1945

THE SAD STATE OF CHEMISTRY HUMOR Two atoms are walking down the street. One atom says to the other, “Hey! I think I lost an electron!” The other says, “Are you sure? ” “Yes, I’m positive!” A neutron walks into a restaurant and orders a couple of drinks. As she is about to leave, she asks the waiter how much she owes. The waiter replies, “For you, No Charge!!!”

SIZE OF AN ATOM Atoms are incredibly tiny. Measured in picometers (10 -12 meters) Nucleus tiny compared to atom California WEB Hydrogen atom, 32 pm radius Radius of the nucleus near 10 -15 m. Density near 1014 g/cm 3 IF the atom was the size of a stadium, the nucleus would be the size of a marble.

PARTICLE ACCELERATOR 7 Km and 27 km


electron e- proton p+ neutron n 0 -1 +1 0 Structure of the Atom 0 Outside of nucleus 1 Inside nucleus

STRUCTURE OF THE ATOM There are two regions 1. Nucleus 2. Contains protons and neutrons Has a positive charge Almost all the mass Single Atom Electron Cloud Where electrons are found Has a negative charge Almost all the volume Water Molecule

ELEMENTS In 1813, a system of representing elements with symbols was introduced. Each symbol consists of one or two letters. Two letters are needed for a chemical symbol when the first letter of that element’s name has already been used. Aluminum Al Bromine Br Calcium Ca Carbon C Gold Au Helium He Hydrogen H Nitrogen N

Element Guide

Element Guide 2
Element Guide 2

Element Guide Proton Number-tells what element it is. (also the atomic number) 2

Element Guide Proton Number-tells what element it is. (also the atomic number) 2 He

Element Guide Proton Number-tells what element it is. (also the atomic number) 2 He

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. 2 He

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. 2 He 4

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. 2 He 4

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. Explains the total atomic weight-The number of protons PLUS the number of neutrons. 2 He 4

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. Explains the total atomic weight-The number of protons PLUS the number of neutrons. 2 He 4
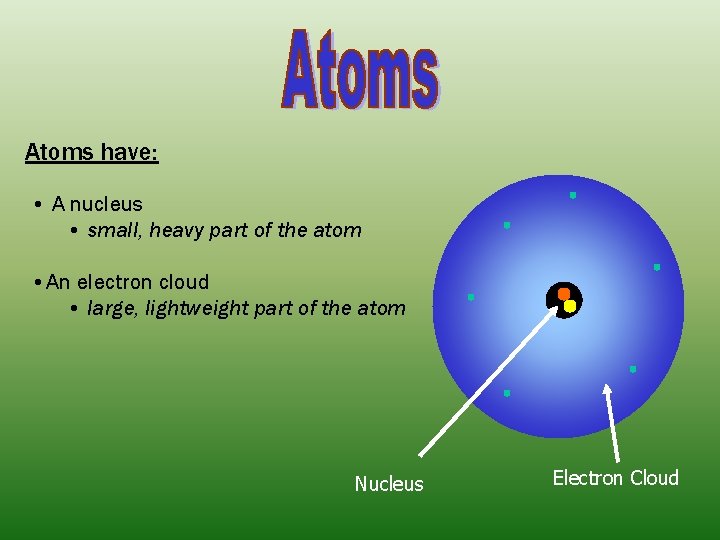
Atoms have: • A nucleus • small, heavy part of the atom • An electron cloud • large, lightweight part of the atom Nucleus Electron Cloud
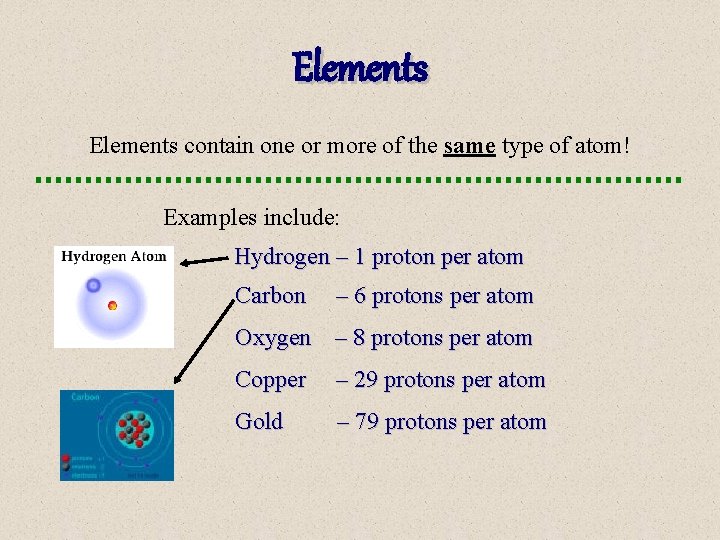
Elements contain one or more of the same type of atom! Examples include: Hydrogen – 1 proton per atom Carbon – 6 protons per atom Oxygen – 8 protons per atom Copper – 29 protons per atom Gold – 79 protons per atom
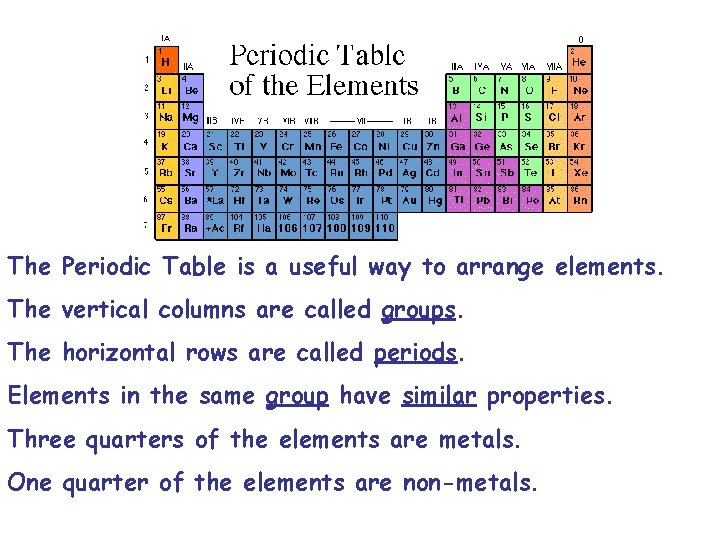
The Periodic Table is a useful way to arrange elements. The vertical columns are called groups. The horizontal rows are called periods. Elements in the same group have similar properties. Three quarters of the elements are metals. One quarter of the elements are non-metals.

Rules for chemical symbols in the periodic table: 1. The symbol is usually the first one or two letters of the name. 2. Sometimes the old (Latin) name is used. 3. The first letter of a symbol id always a capital letter. 4. The second letter of a symbol is always a small letter. 5. Every element has a different symbol.

Now look in your periodic table and find the symbols of the following elements, then find out if it is a metal or a non-metal: Copper ……. . Iron ………. . Magnesium ………. . Chlorine ………. . Carbon ………… Sodium ……. Lead ………. Gold ……… Calcium ……. . Fluorine ……….

Now look in your periodic table and find name that corresponds to the following symbol: Fe ……. . Mg ………. . Ag ………. . K ………. . C ………… Ca ……. Na ………. N ……… Ne ……. . Pb ……….

Fe

Cu

N

F

K

Zn

Li

Al

O

The Periodic Table Q 1. The symbol of Nitrogen is: a. Na b. N Q 2. Hg is the symbol of: a. Hydrogen b. Helium c. Ni c. Mercury Q 3. The percentage of metals in the periodic table is: a. 75% b. 50% c. 25% Q 4. Water is not in the periodic table because: a. It is a liquid substance b. It is a natural substance c. It is a compound not an element

The Periodic Table Q 1. The symbol of Nitrogen is: a. Na b. N Q 2. Hg is the symbol of: a. Hydrogen b. Helium c. Ni c. Mercury Q 3. The percentage of metals in the periodic table is: a. 75% b. 50% c. 25% Q 4. Water is not in the periodic table because: a. It is a liquid substance b. It is a natural substance c. It is a compound not an element
- Slides: 50