WHO Technical Briefing seminar Geneva 16 20 November

WHO Technical Briefing seminar Geneva, 16 – 20 November 2009. Challenges in Medicines Supply Systems: Country case studies Helen Tata & Magali Babaley EMP/MAR 1| WHO Technical Briefing Seminar 19 November 2009 Geneva

Context l 11 African countries planned in 2007 to evaluate their medicines supply systems (Cameroon, Senegal, Rwanda, ( Burundi, Mali, Congo-Brazzaville, Ghana, Nigeria, Tanzania and Zambia) l The countries carried out either 1 or 2 of these surveys: 1. Mapping of all partners involved in procurement and distribution of essential medicines and medical supplies; 2. In-Depth Assessment of medicines supply management system in countries. 2| WHO Technical Briefing Seminar 19 November 2009 Geneva

Mapping/in-depth assessment of medicines supply systems Objectives Support the Ministry of Health to : l Map out all partners involved in medicines procurement and distribution – Use evidence as basis of advocating strategies to strengthen coordination of partners involved in the supply system. l Carry-out an in-depth assessment of the national medicines supply management system in order to: – determine strengths and weaknesses of the system – Identify gaps in the system and proposed strategies to fill in the gaps. 3| WHO Technical Briefing Seminar 19 November 2009 Geneva

Relevance to MOH l To identify information gaps l To have an advocacy tool, an information link and feedback to support planning for a coordinated, coherent and efficient national medicines supply system. l To mobilize resources for under served programme areas within the macro frame of the sector needs l To re-schedule procurement priorities, facilitate forecasting and maintain an interrupted supply chain l To ensure full supply and security for essential medicines 4| WHO Technical Briefing Seminar 19 November 2009 Geneva

Relevance to Partners Factual evidence for: l Providing a plate form for information exchange between partners working in medicines supply system l Providing information on under-financed product categories l Targeting gaps and redefining strategies for strengthening medicines supply systems l Providing information for program monitoring and future projects 5| WHO Technical Briefing Seminar 19 November 2009 Geneva

Tools for Mapping/in-depth assessment l WHO/AFRO in collaboration with WHO/HQ developed the methodology and questionnaires to support countries in this activity l Tools: 6 questionnaires – – – 1) Partners 2)Ministry of Health, 3)Programmes, 4)Central Medical Stores, 5)Regional/District Stores and 5)Health Facilities. l Assessment team: – National coordinator – External consultant or expert from WHO/AFRO or HQ – WHO Medicines Adviser in country office 6| WHO Technical Briefing Seminar 19 November 2009 Geneva
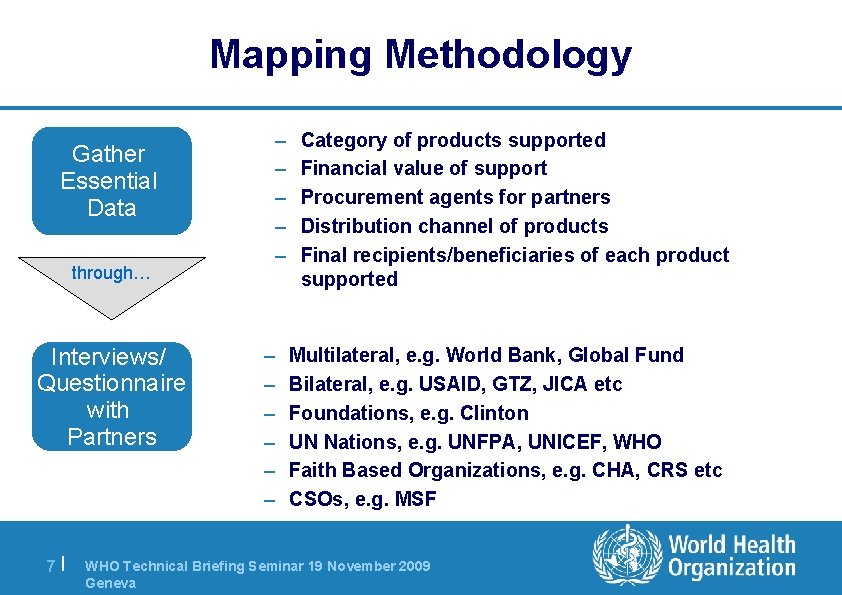
Mapping Methodology Gather Essential Data through… Interviews/ Questionnaire with Partners 7| – – – Category of products supported Financial value of support Procurement agents for partners Distribution channel of products Final recipients/beneficiaries of each product supported Multilateral, e. g. World Bank, Global Fund Bilateral, e. g. USAID, GTZ, JICA etc Foundations, e. g. Clinton UN Nations, e. g. UNFPA, UNICEF, WHO Faith Based Organizations, e. g. CHA, CRS etc CSOs, e. g. MSF WHO Technical Briefing Seminar 19 November 2009 Geneva

Tool: Level III Indicators l Medicines management cycle – – – – 8| Selection Quantification/Forecasting Procurement Ordering Storage/Stock management Distribution Quality assurance system Rational use Financing Information management Monitoring and evaluation Human resources Policy framework WHO Technical Briefing Seminar 19 November 2009 Geneva

Mapping results l Burundi l Congo Republic l Ghana l Mali l Nigeria l Rwanda l Senegal l Tanzania l Zambia l ALL MAPS 9| WHO Technical Briefing Seminar 19 November 2009 Geneva
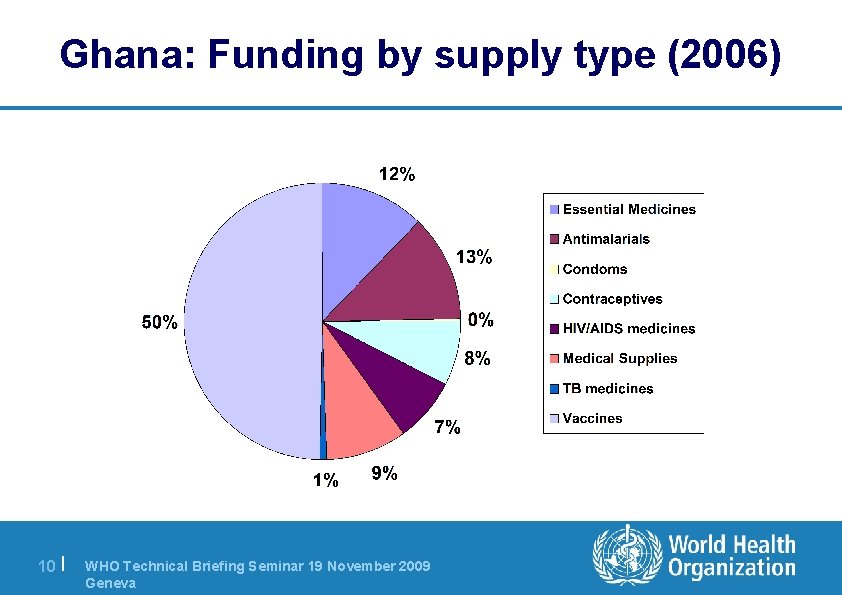
Ghana: Funding by supply type (2006) 10 | WHO Technical Briefing Seminar 19 November 2009 Geneva
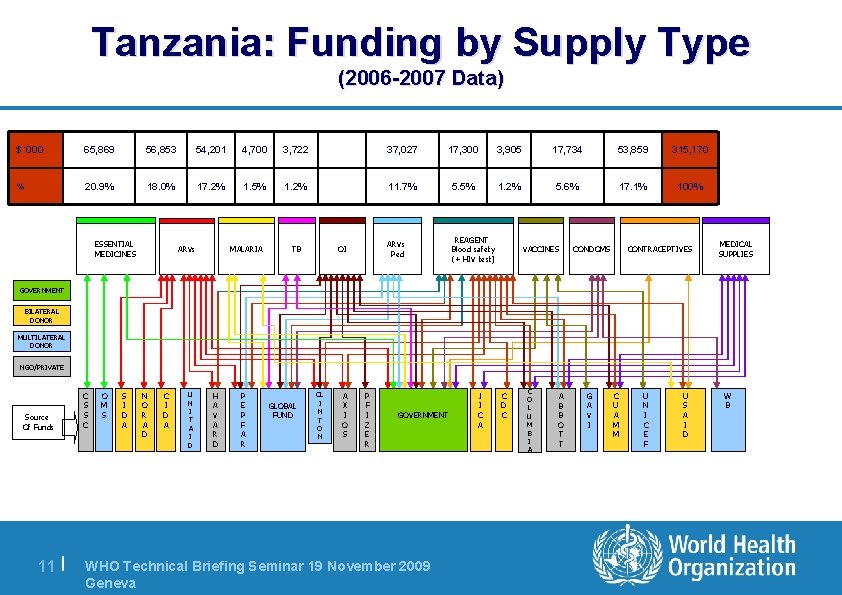
Tanzania: Funding by Supply Type (2006 -2007 Data) $ ‘ 000 65, 869 56, 853 54, 201 4, 700 3, 722 37, 027 17, 300 3, 905 17, 734 53, 859 315, 170 % 20. 9% 18. 0% 17. 2% 1. 5% 1. 2% 11. 7% 5. 5% 1. 2% 5. 6% 17. 1% 100% ARVs Ped REAGENT Blood safety (+ HIV test) ESSENTIAL MEDICINES ARVs MALARIA TB OI VACCINES CONDOMS CONTRACEPTIVES MEDICAL SUPPLIES GOVERNMENT BILATERAL DONOR MULTILATERAL DONOR NGO/PRIVATE Source Of Funds 11 | C S S C O M S S I D A N O R A D C I D A U N I T A I D H A V A R D P E P F A R GLOBAL FUND CL I N T O N A X I O S P F I Z E R GOVERNMENT WHO Technical Briefing Seminar 19 November 2009 Geneva J I C A C D C C O L U M B I A A B B O T T G A V I C U A M M U N I C E F U S A I D W B
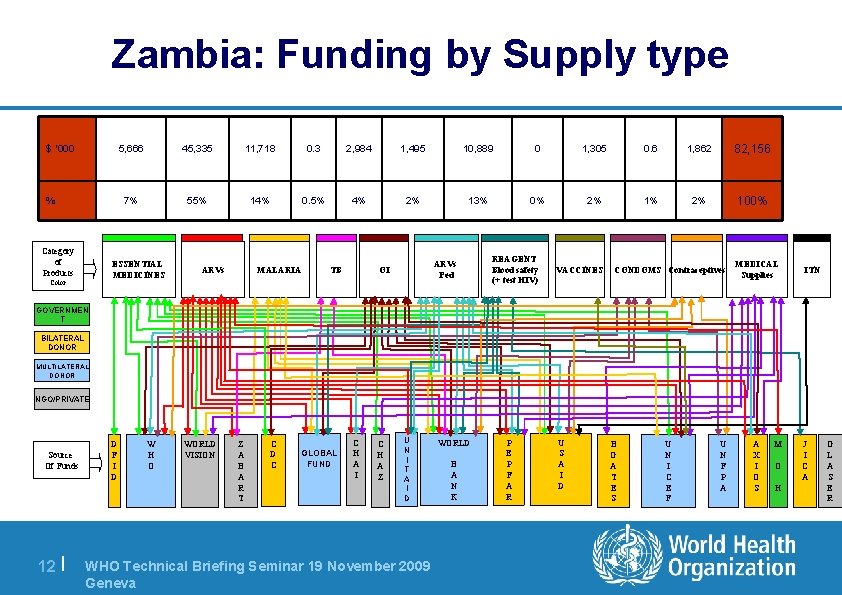
Zambia: Funding by Supply type $ ‘ 000 % Category of Products 5, 666 45, 335 11, 718 0. 3 2, 984 1, 495 10, 889 0 1, 305 0. 6 1, 862 82, 156 7% 55% 14% 0. 5% 4% 2% 13% 0% 2% 100% ESSENTIAL MEDICINES Color ARVs MALARIA TB ARVs Ped OI REAGENT Blood safety (+ test HIV) VACCINES CONDOMS Contraceptives MEDICAL Supplies ITN GOVERNMEN T BILATERAL DONOR MULTILATERAL DONOR NGO/PRIVATE Source Of Funds 12 | D F I D W H O WORLD VISION Z A B A R T C D C GLOBAL FUND C H A I C H A Z U N I T A I D WHO Technical Briefing Seminar 19 November 2009 Geneva WORLD B A N K P E P F A R U S A I D B G A T E S U N I C E F U N F P A A X I O S M O H J I C A G L A S E R

Study results: Strengths l Increased funding for procurement of specific categories of essential medicines from donors such as: – Global Fund to fight HIV/AIDS, TB, malaria – UNITAID – PEPHAR – Clinton Initiative – World Bank etc l Several partners offering technical support, though not coordinated, to strengthen national capacities in medicines procurement and supply management l Some categories of products receive Logistic support to improve distribution and physical availability l Medicines supply systems (public and private) exist in all the countries l Motivated human resource team in a difficult work environment 13 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Study results: Challenges (1) l Structure Supply system: – Complex and magnitude not known to the actors – Responsibilities and tasks of each of the actors not clearly defined – Transparency not always assured – CMS is rarely involved in forecasting/procurement l Selection: – Supply outside the EML/STG still exists – Procurement of non registered medicines in countries still exists l Quantification: – Lack of coordination in planning between Mo. H/CMS and all partners results in stock-outs, shortages, overstocking and expired products – Adequate logistic information system not in place (due to the complexity of the system? ) 14 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Study results: Challenges (2) Procurement: Difficult to manage the various procurement and financial procedures specific to each of the multitude of partners. Stock management: – Specific stock management tools available for products of some partners, leading to increased work load for personnel and adding to the complexity of stock management. – CMS may not have adequate storage capacity due to lack of coordination in procurement planning Distribution: – Inadequate funds for distribution. – Where funds are available, no coordination between different programs leading to high operational costs for all programs. 15 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Study results: Challenges (3) Financing: – Most of today's donor funding target vertical disease programs such as HIV/AIDS, tuberculosis, malaria and EPI – The above may lead to some programs being under-supported while others are over-supported – Financial figures not always available – Logistic support is under- funded Monitoring-Evaluation: – Each unit/project having its stand alone M&E project for its products (e. g. condoms, ARVs, PMTCT, ITN, Coartem, Test Kits etc – Not easy to manage due to the number of different reporting tools which increase the burden of work for personnel Human resources: – Allocation of scarce human and financial resources distorted. – Single staff at lower level of care not well equipped for multipurpose tasks – Incentive systems distorted leading to internal brain drain 16 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Next steps Providing further support to these countries in: • Coordination strategies/mechanisms at country and global levels • Strengthening systems and tools on needs assessment/quantification • Promoting compliance for Drug Donation Guidelines • Information sharing across national programs/partners for all categories of products studied 17 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Drug donation guidelines Article no. 12 states: – "Costs of international and local transport, warehousing, port clearance and appropriate storage and handling should be paid by the donor agency, unless specifically agreed otherwise with the recipient in advance. " Additional guidelines for drug donations as part of development aid: – "It should be recognized that drugs do not arrive in an administrative vacuum. Drug donations should not create an abnormal situation which may obstruct or delay national capacity building in selection, procurement, storage, distribution and rational use of drugs. " – "Administratively, the drugs should be treated as if they were procured. This means that they should be registered or authorized for use in the country through the same procedure that is used for government tenders. They should be entered into the inventory, distributed through the existing distribution channels and be subject to the same quality assurance procedures. If cost-sharing procedures are operational in the recipient country, the donated drugs should not automatically be distributed free of charge. " 18 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Paris Declaration l http: //www. mfdr. org/sourcebook/1 st. Edition/2 -1 Paris. pdf l Ownership: country sets the agenda l Alignment: donor country – Aligning with country agenda – Using country's systems l Harmonization: donor-donor – Ensure that actions are harmonized, transparent, and collectively effective; • Establishing common arrangements • Simplifying procedures • Complementarities: more effective division of labor • Sharing information to avoid duplication l Managing for results: – Countries and donors to shift focus to development results and get results measured. l Mutual Accountability – 19 | Donors and partners are accountable for development results. WHO Technical Briefing Seminar 19 November 2009 Geneva

Partner Alignment: The Global Fund example in Tanzania l GF is inline with GOT system l GF gives fund to the GOT (Paymaster General) l Disbursed to beneficiary programs- NACP/NMCP l Deposited to MSD (NACP/NMCP) l MSD then procure, store, and Distribute l MSD charge 8% for operating costs (the rest 6% covered by MOHSW) 20 | WHO Technical Briefing Seminar 19 November 2009 Geneva

Thank You 21 | WHO Technical Briefing Seminar 19 November 2009 Geneva
- Slides: 21