WHO Prequalification of Diagnostics WHO Technical Briefing Seminar

WHO Prequalification of Diagnostics WHO Technical Briefing Seminar on Essential Medicines and Health Products WHO HQ, Geneva, 28 October - 1 November 2013 Mercedes Pérez González Department of Essential Medicines & Health Products

Regulation of diagnostics (IVDs) l Regulation specifically for diagnostics is often poorly understood and/or poorly enforced l Production of many diagnostic products has been moved to countries with less strict regulatory framework. l Different categories of IVDs regulated differently – HIV IVDs, particularly for blood screening, attract greatest stringency – Degree of stringency is usually risk-based – Risk perception is different in different settings l Different regulatory versions of the same product 2 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Who sets international standards? Organization International Organization for Standardization (ISO) Certification of ISO compliance is made by an independent agency. Global Harmonization Task Force (GHTF) Comprised on national regulators & industry. Issues guidance on specific topics related to medical devices including IVDs. International Medical Device Regulators Forum (IMDRF) - replaced GHTF Comprised on national regulators. Maintains GHTF guidance documents. Clinical and Laboratory Standards Institute (CLSI) Issues guidance documents specific for testing processes. 3 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Role of WHO l To provide normative guidance to Member States on when and how to use IVDs to guide clinical decision-making – WHO ART guidelines l To provide recommendations on quality and performance of IVDs through the WHO Prequalification of Diagnostics programme according to international standards l To increase in-country capacity to effectively regulate & to monitor quality of diagnostics in their market 4 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Aim of WHO Prequalification of Diagnostics l To promote and facilitate access to safe & appropriate diagnostic technologies of good quality in an equitable manner – Through adoption of GHTF guidance and ISO requirements l Customers – WHO Member States – UN agencies – Funding and procurement agencies 5 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
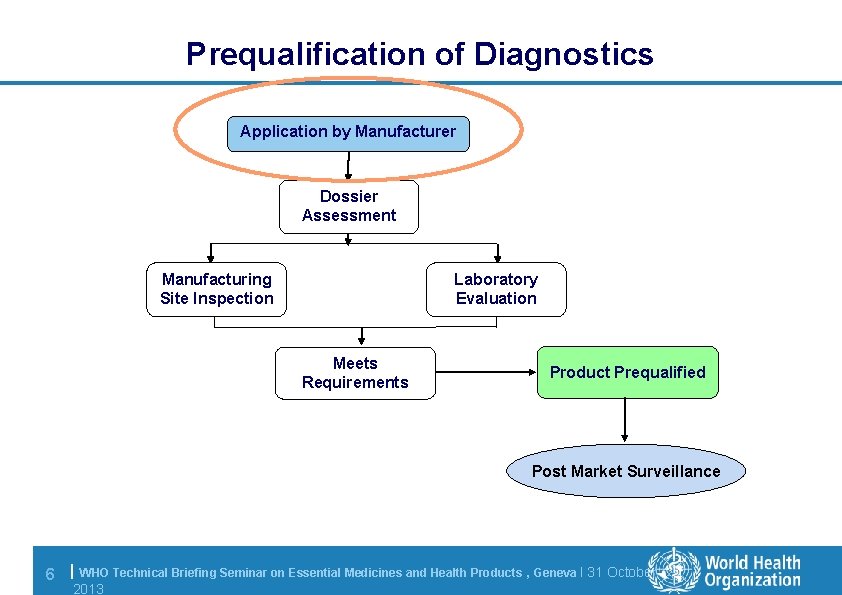
Prequalification of Diagnostics Application by Manufacturer Dossier Assessment Manufacturing Site Inspection Laboratory Evaluation Meets Requirements Product Prequalified Post Market Surveillance 6 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Application: requirements l Manufacturer may submit application at any time to diagnostics@who. int – Must use the Prequalification of Diagnostics application form – Instructions for the completion of the application form contains information to help fill the form 7 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Prioritization of PQDx applications Current prioritization criterion Comment Already listed on WHO/UN procurement scheme and procured by UN organizations in significant levels Ensure continuity of supply and quality of products procured Assist diagnosis of infection with HIV-1/HIV-2, Focus on priority disease areas – highest or malaria historical procurement Rapid test format Bringing testing closer to the community Original product manufacturers Ensure known supply chain; no duplication of effort, best possible prices Few other prequalified products exist in the product category such as CD 4, VL Focus on unmet market / procurement needs 8 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
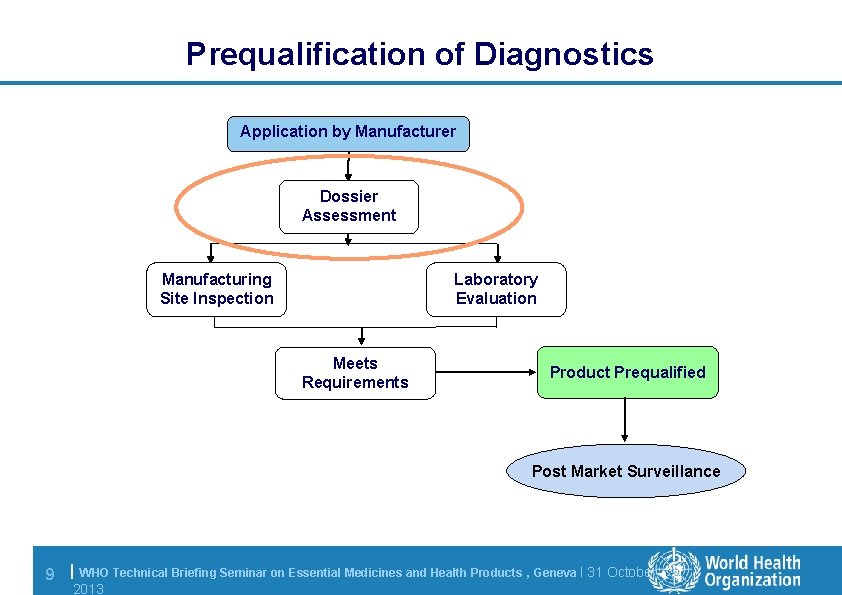
Prequalification of Diagnostics Application by Manufacturer Dossier Assessment Manufacturing Site Inspection Laboratory Evaluation Meets Requirements Product Prequalified Post Market Surveillance 9 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Dossier: requirements l Based on best international practice (ISO, EN, GHTF, CLSI); follow the content of the GHTF STED l Looks into critical aspects for WHO Member States often not dealt with/dealt with from a local prospective by SRAs (stability, IFU, RA, etc. ). l Dossier must demonstrate that the IVD conforms to the Essential Principles of Safety and Performance of Medical Devices (GHTF/SG 1/N 41 R 9: 2005) 10 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Dossier: requirements Key Components Product description Design and manufacturing information Product performance specifications & associated validation and verification studies Labelling Commercial history Regulatory history Quality management system 11 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Dossier: submission l Clinical evidence to validate performance claims – One clinical evaluation* performed by Manufacturer – One clinical evaluation* performed independently • Must clearly relate to the product undergoing prequalification (same name, same product code, same regulatory version) Performance characteristics Clinical (diagnostic sensitivity) including seroconversion sensitivity Clinical (diagnostic) specificity Positive and negative predictive values (high/low prevalence) Different clinical stages Geographical distribution (consider intended use setting) Genotypic differences *The EC Common Technical Specifications (CTS) for IVDs 2009 are a useful guide http: //eur-lex. europa. eu/Lex. Uri. Serv. do? uri=OJ: L: 2009: 039: 0034: 0049: EN: PDF 12 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
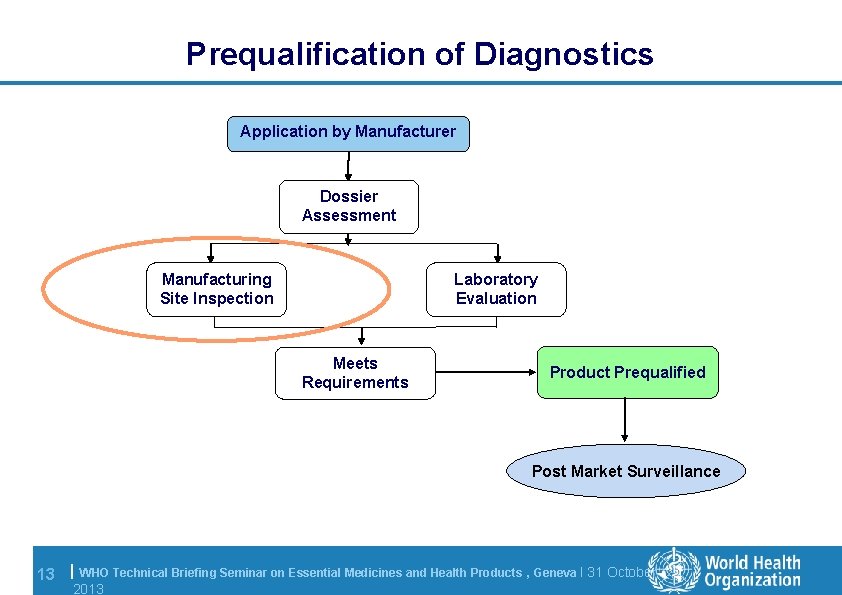
Prequalification of Diagnostics Application by Manufacturer Dossier Assessment Manufacturing Site Inspection Laboratory Evaluation Meets Requirements Product Prequalified Post Market Surveillance 13 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Inspection: requirements § The manufacturer must demonstrate that the IVD is produced under a functional quality management system e. g. conforms to ISO 13485: 2003 Key Components Quality management system including documentation requirements Management responsibility including customer focus, quality policy Resource management including human resources, work environment Product realization including production and service provision, control of monitoring and measuring devices Measurement, analysis and improvement including control of nonconforming product, improvement 14 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Inspection: requirements l Dossier submission data – to confirm is true l Production l QC and lot release – QC panels should be challenging enough to detect failure or drift – Independence and adequately staffed QA/QC department – Deviation reporting procedures observed l WHO related/end user issues 15 | IFU stability (transport, in-use, expiry dates) training complaints reporting mechanisms WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
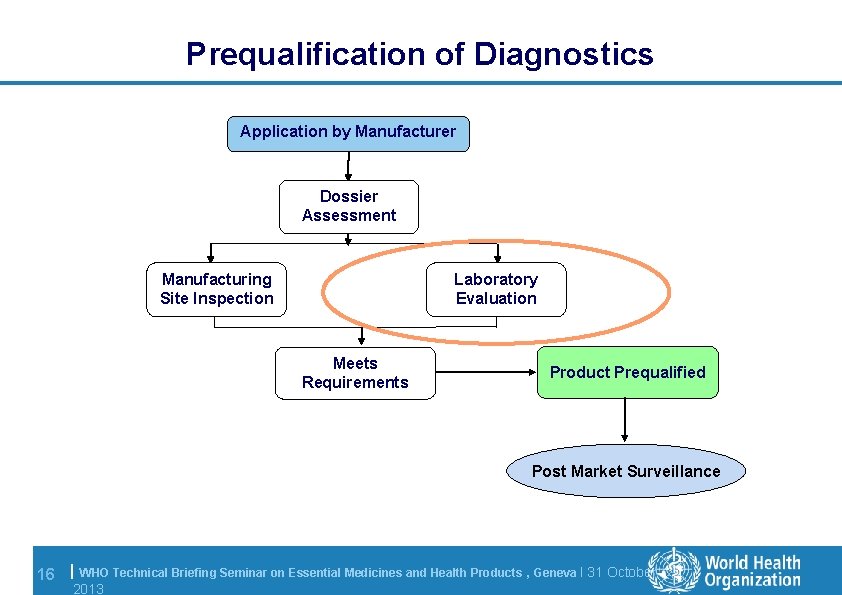
Prequalification of Diagnostics Application by Manufacturer Dossier Assessment Manufacturing Site Inspection Laboratory Evaluation Meets Requirements Product Prequalified Post Market Surveillance 16 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Evaluations performed l WHO evaluation protocols followed, based on existing international standards and best practice and adapted to assay type l Performance and operational characteristics l WHO Collaborating Centres performs evaluation under supervision of WHO l WHO Composite Reports of all products produced – Report 17 to be released 17 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Laboratory evaluation: outcome EIA (Laboratory) l If RDT, results are read by 3 independent readers l Two production lots are submitted to assess lot-tolot variation l Results of the WHO laboratory evaluation must meet the acceptance criteria 18 | RDT (Point of Care or Laboratory) HIV serology Sensitivity: 100% Specificity: ≥ 98% Sensitivity ≥ 99% Specificity ≥ 98% Inter-reader variability ≤ 5% Invalid rate ≤ 5% HCV serology Sensitivity: 100% Specificity: ≥ 98% Sensitivity ≥ 98% Specificity ≥ 97% Inter-reader variability ≤ 5% Invalid rate ≤ 5% HBs. Ag serology Sensitivity: 100% Specificity: ≥ 98% Sensitivity 100% Specificity ≥ 98% Inter-reader variability ≤ 5% Invalid rate ≤ 5% WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
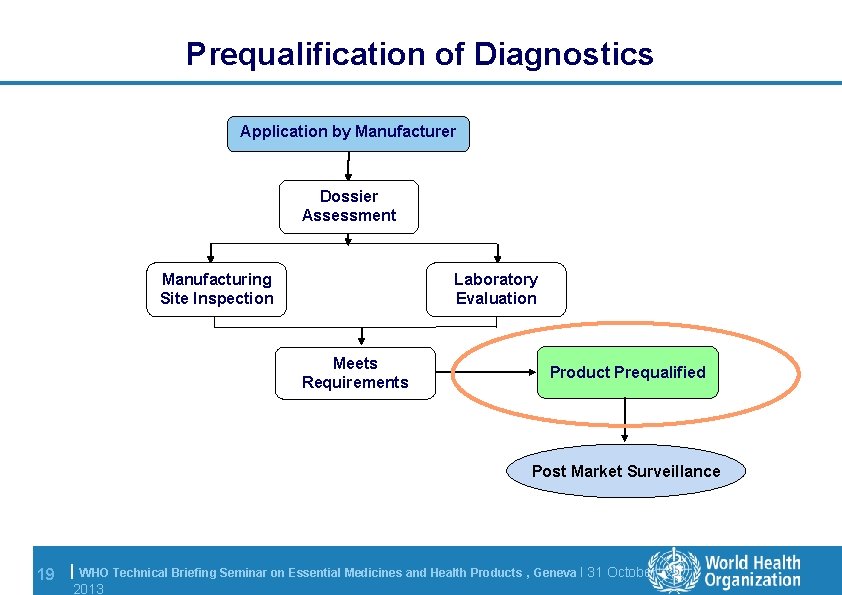
Prequalification of Diagnostics Application by Manufacturer Dossier Assessment Manufacturing Site Inspection Laboratory Evaluation Meets Requirements Product Prequalified Post Market Surveillance 19 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Prequalification: decision l Final prequalification outcome depends on: – Results of dossier assessment and acceptance of action plan – Results of inspection and acceptance of action plan • no critical nonconformities outstanding – Meeting the acceptance criteria for the laboratory evaluation l WHO PQDx Public Report is posted on WHO website and product is added to the list of WHO prequalified products l Product is then eligible for WHO and UN procurement 20 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

PQDx fast track procedure l The process relies on provision of relevant “evidence” of certification issued by a stringent regulatory authority (SRA) or their authorised representative(s) l Submitted evidence will be considered for its suitability in satisfying WHO requirements. l This applies to – the dossier assessment (product design and development), – and/or inspections of the site of manufacture and the quality system – And/or the laboratory evaluation 21 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

PQDx assessment status for all products l WHO website updates the status of each product undergoing PQDx assessment monthly http: //www. who. int/diagnostics_laboratory/pq_status/en/index. html 22 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
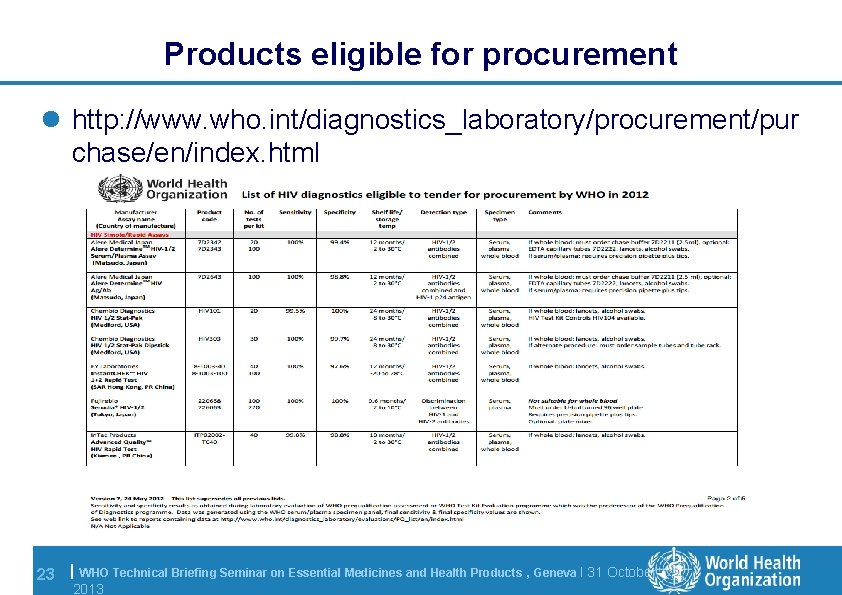
Products eligible for procurement l http: //www. who. int/diagnostics_laboratory/procurement/pur chase/en/index. html 23 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
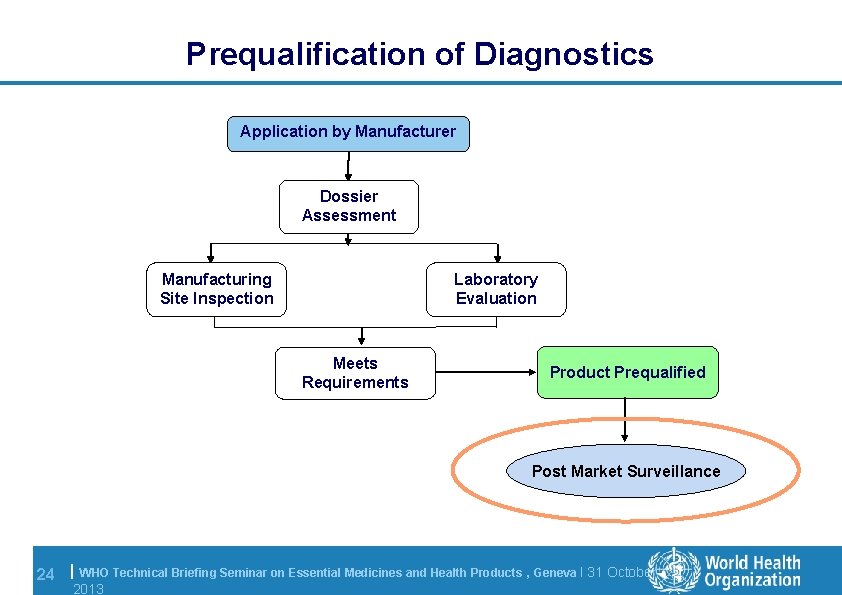
Prequalification of Diagnostics Application by Manufacturer Dossier Assessment Manufacturing Site Inspection Laboratory Evaluation Meets Requirements Product Prequalified Post Market Surveillance 24 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Post-market surveillance l Should be the onus of the manufacturer, but poorly executed l WHO PQDx complaint form for end users to report issues – http: //www. who. int/diagnostics_laboratory/procurement/complaints/ en/index. html – GHTF/SG 2 -N 54 R 8: 2006 • Medical Devices Post Market Surveillance: Global Guidance for Adverse Event Reporting for Medical Devices – GHTF/SG 2 -N 57 R 8: 2006 • Medical Devices Post Market Surveillance: Content of Field Safety Notices l Pilot project on PMS – Regulatory capacity building – Capacity building at the NRL level 25 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013

Contact us l Contact us by email diagnostics@who. int WHO Prequalification of Diagnostics programme website http: //www. who. int/diagnostics _laboratory/evaluations/en/ 26 | WHO Technical Briefing Seminar on Essential Medicines and Health Products , Geneva l 31 October 2013
- Slides: 26