WHO Model Formulary WMF Dinesh K Mehta September

WHO Model Formulary (WMF) Dinesh K Mehta September 2004

What healthcare workers want to know about medicines • What medicine for a given condition? • Is the medicine suitable for my patient? • What else do I need to know to prescribe it? • What do I need to know about a previously prescribed medicine?

Requirements for prescribing information Authoritative Practical Comprehensive Widely distributed Up-to-date Locally relevant
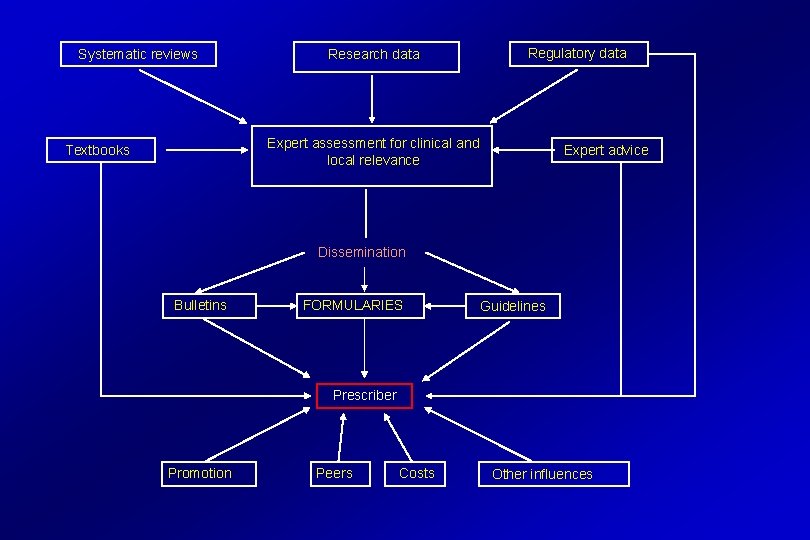
Systematic reviews Research data Regulatory data Expert assessment for clinical and local relevance Textbooks Expert advice Dissemination Bulletins FORMULARIES Guidelines Prescriber Promotion Peers Costs Other influences

Anatomy of a formulary • ‘Front matter’ • Notes • Monographs • Appendices

How is it presented? • The book • Digitally • Adaptations and translations

Adapting information for local use • • • Language Training Affordability Prevalence of conditions and priorities Healthcare provision Sophistication of technology

WHO Model Formulary: production

Validation of existing text Compare WMF text against · New WHO publications · Changes between editions of BNF · Martindale

Sources · WHO experts and Expert Committee · WHO programmes · WHO publications · Summaries of Product Characteristics (SPCs) · Original papers and systematic reviews (e. g. Cochrane reviews) · BNF · Martindale · Drug withdrawals · Textbooks · Readers’ comments

Verification of new text New and amended text: · Checked independently by 2 editors · Extra check of dose statements · Sent to WHO for verification

Style · WHO style manual · Use of XML to code database ensures consistent style of individual elements

How you can help · Make thorough use of the resource · Report: · Areas of disagreement · Ambiguity or incomplete information · Mistakes! · Make suggestions for improvement/expansion

The future • Evolution of scope and extent • Research on effectiveness • Digital versions e. g. – PDA (handheld) • Translated versions (book and digital)
- Slides: 14