WHO guidance on postmarket surveillance EMP Technical briefing

WHO guidance on post-market surveillance EMP Technical briefing seminar 3 - 7 November 2014 Anita Sands Prequalification Team – Diagnostics Department of Essential Medicines & Health Products

About post-market surveillance l Some issues related to quality, safety and performance may not be apparent during pre-market assessment or may only arise after a product is placed on the market l ISO 9001: 2000, ISO 13485: 2003 and ISO 14971: 2000 list the requirements on post-market activities to be performed by manufacturer l Post-market surveillance is necessary to detect, investigate and act on any issues – Sometimes leading to field safety corrective action by the manufacturer 2 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

WHO post-market surveillance of IVDs WHO prequalified product Reactive PMS Complaints Field Safety Corrective Action Field Safety Notice 3 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014 Proactive PMS Lot verification testing

PMS roles and responsibilities l Key stakeholders – End users (testers, procurers, implementers), manufacturers, NRAs, NRLs, WHO l Each stakeholder ensures that post-market surveillance data is detected, assessed and properly acted upon – – 4 End users are crucial as they usually first report problems; NRLs perform lot testing NRAs often lack capacity for post-market oversight; WHO promotes the practice of PMS, ensures coordination of lot testing and ensuring manufacturers undertake appropriate investigation and any action, if required. Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

PMS: definitions l Adverse event (also called an incident) – A product defect (i. e. malfunction or failure, deterioration in characteristics or performance, or inadequacy of labelling or of instructions for use) that, directly or indirectly, has led or might have led to serious medical consequences, namely death or serious deterioration in the state of health of the patient, user or another person – May be classified as mild, moderate, severe l Complaint – Any written, electronic, or oral communication that alleges deficiencies related to the identity, quality, durability, reliability, safety, effectiveness, or performance of a device after it is released for distribution 5 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

PMS: definitions l Field safety corrective action – Action taken by the manufacturer to reduce a risk of death or serious deterioration in the state of health associated with the use of a medical device that is already placed on the market – A FSCA is triggered when there is an unacceptable increase in risk associated with use of the device – FSCAs may include: • return of device to supplier, device modification, device exchange, device destruction, retrofit by purchased of device according to Mx modificationdesign control 6 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Reactive PMS: definitions l Field safety corrective action cont'd – A device modification can include: • permanent or temporary changes to the labelling or instructions for use; • software upgrades including those carried out by remote access; • modification to the clinical management of patients to address a risk of death or serious deterioration in state of health related specifically to the characteristics of the device. – A FSCA should be notified to NRA (and WHO) through Field Safety Corrective Action Report 7 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

PMS: definitions l Field safety notice – A communication sent out by the manufacturer or its representative to the device users in relation to a field safety corrective action – It tells their customers about a FSCA they are undertaking • It should be sent to all users for their attention and possible action • It should be sent to all NRAs in the country of supply 8 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Reactive PMS: complaint reporting for users l User identifies problem l User documents the problem l User reports problem to their NRA (or national IVD focal point) l National IVD focal point – Reports all complaints (including AEs) to manufacturer – Reviews trends for mild/moderate adverse events (AEs) – Reports all serious AEs, and any change in trends of mild/moderate AEs to WHO 9 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Reactive PMS: WHO complaint reporting for Mx l Manufacturer receives complaint from national IVD focal point and/or WHO – Investigates problem and takes appropriate action including root cause analysis, and if appropriate, any FSCA – Forwards initial and final investigation reports to WHO – If it is required, forwards FSCA report to WHO l WHO will ensure that the manufacturer undertakes the appropriate investigation and, if required FSCA 10 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

What will WHO do? l WHO will monitor the manufacturer's investigation including risk assessment, and any required FSCA – – Course (the direction the investigation is taking) Conduct (how the investigation is being carried out) Progress (how quickly the investigation is being carried out) Outcome (whether the results of device analysis are satisfactory) l WHO may inform other NRAs, may decide if WHO PQ status is affected, and may decide to take no further action l WHO may issue an Information Notice for Users where the manufacturer has not undertaken an appropriate FSCA and/or has not issued a FSN, including if not done quickly 11 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014
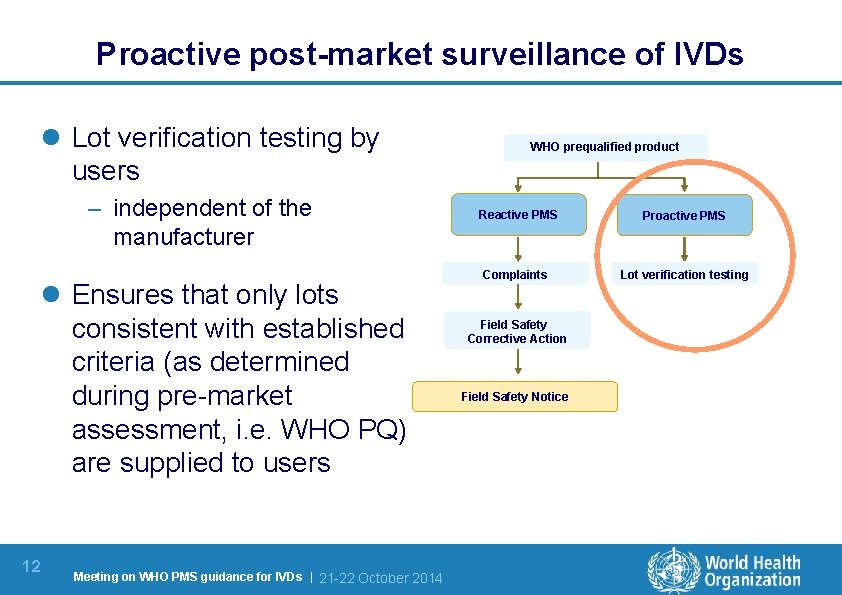
Proactive post-market surveillance of IVDs l Lot verification testing by users – independent of the manufacturer l Ensures that only lots consistent with established criteria (as determined during pre-market assessment, i. e. WHO PQ) are supplied to users 12 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014 WHO prequalified product Reactive PMS Proactive PMS Complaints Lot verification testing Field Safety Corrective Action Field Safety Notice

Proactive PMS of IVDs: lot testing l Lot verification – Through physical inspection of packaging, labelling and instructions for use; and – Testing of samples from a lot against a characterised panel of specimens l Lot testing should be centrally performed by qualified laboratory l Lot acceptance criteria must be in place (pass/fail) 13 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Proactive PMS of IVDs: panels for lot testing l Same panel of locally sourced specimens of known reactivity for each lot of the same IVD l Minimum requirements – – – Analytical sensitivity Clinical sensitivity Specificity Invalid rates Anomalies • Incomplete clearing, high background, faint line, etc. l Acceptance criteria for performance 14 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Proactive PMS of IVDs: sampling for lot testing l Pre-distribution l Post-distribution – Systematic or random sampling of lots – Easier to prevent delivery of poorly performing lots to the testing sties – Requires more resources – Random lots – More difficult to recall defective lots – What to do for individuals already tested using those lots – Less resource-intensive l May use a risk-managed approach to decide on sampling interval, depends on size of lot 15 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Proactive PMS of IVDs: physical inspection l With each new lot: – Inspection of packaging integrity may alert to mishandling – Inspection of package contents will reveal labelling, inclusion errors – Review of instructions for use will detect any method changes l Keep records – Lot numbers and expiry dates, date of first use, all test results and band intensities, rates of invalid results, false positive rates Cannot verify “error” without good records 16 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014

Objective of WHO guidance on PMS l Narrative l Forms – – – IVD complaint reporting form Manufacturer investigation reporting form Field safety corrective action reporting form Lot testing data collection form Lot testing reporting form l Notices – Field Safety Notice 17 Meeting on WHO PMS guidance for IVDs | 21 -22 October 2014
- Slides: 17