WHO Good Governance for Medicines programme Technical Briefing

WHO Good Governance for Medicines programme Technical Briefing Seminar 3 November 2010, Geneva Dr Guitelle Baghdadi-Sabeti Department of Essential Medicines and Pharmaceutical Policies

Corruption requires two parties: the corrupter and the corruptee "Whose is the greater blame? She who sins for pay or he who pays for sin? " Sor Juana Inés de la Cruz Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 2

What is the definition of corruption? "The abuse of entrusted power for personal gain" Transparency International Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 3

Unethical practices can be found throughout medicines chain R&D and clinical trials R&D priorities Patent Unlawful appropriation royalties Counterfeit/ substandard Tax evasion Conflict of interest Manufacturing Registration Pricing Cartels Selection Unethical donations Collusion Pressure Procurement & import Overinvoicing Thefts Distribution Inspection Falsification safety/ efficacy data Bribery Prescription Dispensing State Capture Pharmacovigilance Unethical promotion Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 4 Promotion

Corruption identified as the single greatest obstacle to economic and social development q Health and pharmaceutical sectors attractive targets ì US$ 5. 3 trillion spent on health services annually "Corruption is a worldwide problem, ì Global pharmaceutical market: > US$ 750 b existing in both high- and lowq No global estimate on financial losses, but: income countries… no country ì 10 to 25% procurement spending lost into should feel offended and restrained corruption to talk about it". ì Some countries report losses: § § 2/3 medicines supplies lost in hospitals Dr H. Hogerzeil, Director, 10% national expenditures on health care. WHO Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 5

Unethical practices can have significant impact on health systems q q q Health impact ì Unsafe medicines on the market ì Lack EM in health facilities ì Irrational use of medicines Economical impact ì Waste limited public/donor funding ì Not stable environment ì Not easy to conduct business Image and trust impact ì Erodes public trust ì Reduces credibility of health profession Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 6

WHO Good Governance for Medicines Programme: an innovative initiative q Goal ì q To contribute to health systems strengthening and prevent corruption by promoting good governance in the pharmaceutical sector Specific objectives ì To raise awareness on the impact of corruption in the pharmaceutical sector and bring this to the national health policy agenda ì To increase transparency and accountability in medicine regulatory and supply management systems ì To promote individual and institutional integrity in the pharmaceutical sector ì To institutionalize good governance in pharmaceutical systems by building national capacity and leadership Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 7

Numerous technical guidelines already exist… the challenge is to balance them with ethical practices Ethical practices Technical guidelines q q GMP GCP q Counterfeits q Manual on Marketing Authorization q Rule of law q Accountability q Transparency q Participation q Merit system q WHO model list of EM q q Good procurement practices Evidence-based decisionmaking q Honesty q Ethical criteria q q Etc… Efficiency and effectiveness q Etc… Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 8
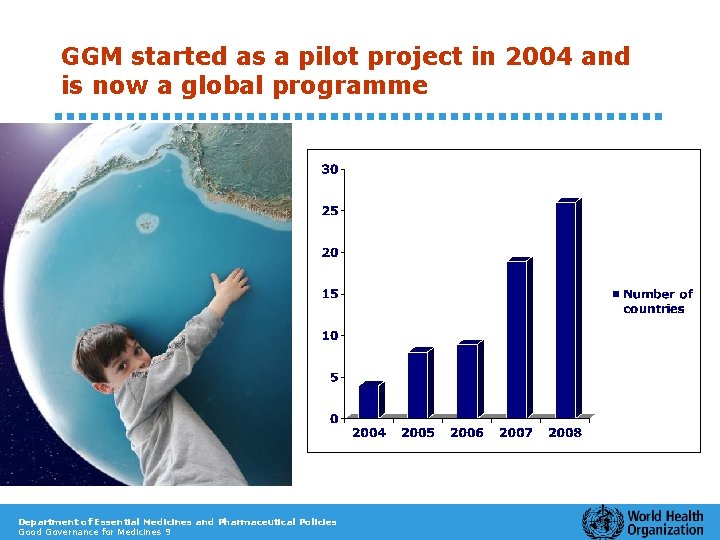
GGM started as a pilot project in 2004 and is now a global programme Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 9
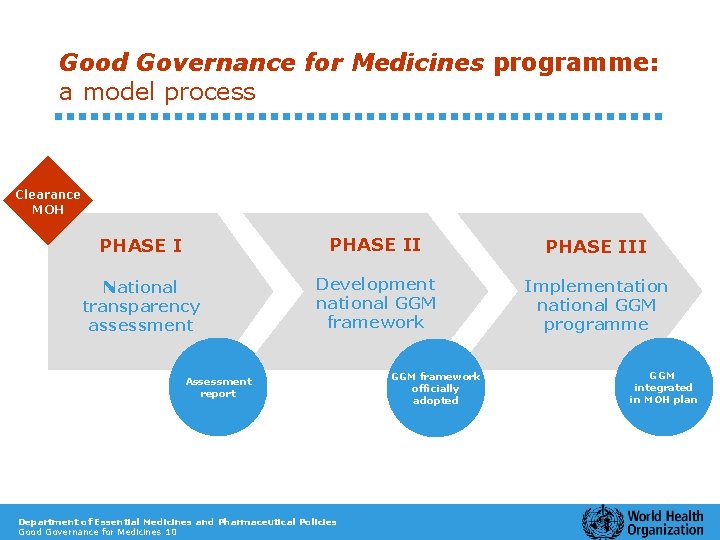
Good Governance for Medicines programme: a model process Clearance MOH PHASE III National transparency assessment Development national GGM framework Implementation national GGM programme Assessment report Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 10 GGM framework officially adopted GGM integrated in MOH plan

Bottom-up approach in policy development lead to the 'GGM technical package' Country case studies and compilation of best practices Working draft PHASE I Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 11 PHASE II In process PHASE III

PHASE III Summary qualitative findings Common strengths Registration Promotion Inspection Selection Procurement Areas of improvement - Written procedures for applications - Conflict of interest policy - Operational committees - Committee composition & TOR - Provisions exist (major variations) - Complete provisions to cover all - No DTCA for prescription-only med. - Establish responsible unit - Inspection report required - Conflict of interest policy - Reports subject to internal review - Appeals mechanisms - Clear criteria for selection process - Conflict of interest policy - Committees (multi-discipl. Experts) - Selection criteria for members - Written procedures - Conflict of interest policy - Well-established tender committees - Audit results publicly available Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 12

PHASE III Efforts to address corruption need coordinated application of two basic strategies q "Discipline-based approach" (top-down) ì ì q Laws, policies and procedures against corruption and for pharmacy practice with adequate punitive consequence for violation Attempts to prevent corrupt practices through fear of punishment "Values-based approach" (bottom-up) ì ì Promotes institutional integrity through promotion moral values and ethical principles Attempts to motivate ethical conduct of public servant Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 13
PHASE II GGM frameworks effective only if translated into action: some preliminary achievements q Lower costs for quality medicines procurement q National pharmaceutical laws, regulations and procedures revised q Web-based pharmaceutical activities (registration, licensing) and information q Conflict of interest policies developed and implemented q Integrity and Leadership training programmes for health officials q Good governance introduced in University curriculum q Communications and advocacy campaigns q Culture of transparency is emerging in institutions Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 14 PHASE III
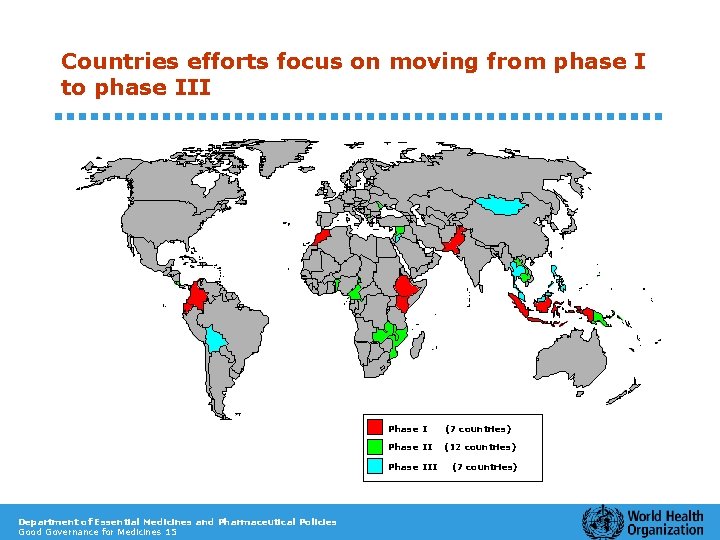
Countries efforts focus on moving from phase I to phase III Phase I (7 countries) Phase II (12 countries) Phase III Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 15 (7 countries)

Key observations and lessons learnt 1. Great interest in subject area (preventive approach appealing) 2. A dedicated and motivated national team to champion the issue 3. Involvement of high-level and technical officials essential 4. Collaboration with key stakeholders 5. Promotion of integrity together with legislative reforms 6. Timeframe different between countries 7. Institutionalization needed for sustainability Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 16

Priorities for 2010 - 2012 q Identify best practices in phase III countries q Integrate corruption on the health agenda (global & countries) q Institutionalization of the GGM q Communications strategy q Monitoring and evaluation q Training phase III and GGM resources q Fundraising Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 17

"I never worry about action, but only inaction. " Winston CHURCHILL Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 18

A few questions for reflection… q Do you believe that corruption exists in your country? Isolated cases or permissive culture? q What are the most common forms? q What are the possible causes? q What makes the pharmaceutical sector so vulnerable to corruption? q What do you think needs to be done to tackle corruption in the pharmaceutical sector on the long run? Department of Essential Medicines and Pharmaceutical Policies Good Governance for Medicines 19
- Slides: 19