Whitley Academy Science Faculty Combined Science Higher Chemistry

Whitley Academy Science Faculty Combined Science Higher Chemistry Paper 1 Knowledge Organiser

For Higher Chemistry Paper 1, you will need to revise. . • • • Energy changes Acids and salts Electrolysis Atomic structure Development of the Periodic table including group 1, group 7 and group 0 Ionic bonding Covalent bonding Reactions with metals Relative molecular mass. Balancing equations Required practicals – acids and salts, Metals reactions

Atoms, elements, compounds and mixtures - PART 1 All substances are made of atoms that cannot be chemically broken down. It is the smallest part of an element. There about 100 different elements Elements are made of only one type of atom. Each element has its own symbol. e. g. Na is sodium. Compounds contain more than one type of atom. They are formed from elements by chemical reactions, which always involve the formation of one or more new substance, often involving an energy change. You need to: • Know the names and symbols for the first 20 elements and all of group 1, 7 and 0 • Name compounds when given the formulae or symbol equations Mg. Cl The components of a compound cannot magnesium be separated by physical means. They can 2 be separated only by chemical means. Magnesium chloride chlorine

Atoms, elements, compounds and mixtures - PART 1 Compounds contain two or more elements chemically combined in fixed proportions and can be represented by formulae using the symbols of atoms from which they formed. Naming a compound with two elements For example: (usually a metal and a non metal) apply these Na 2 S = sodium sulfide rules: K 2 O = potassium oxide • The metal name does not change When a compound contains a • The non-metal's name ends in ide transition metal, the names become a bit more complicated. Naming a compound with a metal To distinguish them, Roman that reacts with ions that consist of numerals indicate the charge two or more non-metal atoms on the metal ion covalently bonded together apply e. g. iron(II) chloride. these rules: • The metal name does not change • The non-metal's name ends in ate For example: Na CO = sodium carbonate 2 3 if oxygen is present KNO 3 = potassium nitrate

Atoms, elements, compounds and mixtures - PART 1 Chemical equations: Show the reactants (what we start with) and the products (what we end with). No atoms are lost or made. The mass of the products equals the mass of the reactants. Word equation: methane + oxygen carbon dioxide + water Symbol equation: CH 4 + 2 O 2 CO 2 + 2 H 2 O There are 4 hydrogens here, bonded together. + There are 2 molecules of oxygen not bonded together. You need to: • Write word equations • Balanced symbol equations There are 4 hydrogens here. You multiply the big number by the little number. + Equations MUST balance • We can ONLY add BIG numbers to the front of a substance • We can tell elements within a compound by BIG letters • We can check an equation is balanced by counting the number of each type of atom on either side

Atoms, elements, compounds and mixtures - PART 1 We can add state symbols to a symbol equation to show whether the reactants and products in a chemical reaction are solids, gases, liquids or dissolved in water. Word equation: Symbol equation: Solid = s Liquid = l Gas = g Aqueous (dissolved in water) = aq sodium + water sodium hydroxide + hydrogen 2 Na(s) + 2 H 2 O(l) → 2 Na. OH(aq) + H 2(g) The reaction between copper sulfate and sodium hydroxide is: copper sulfate + sodium hydroxide → sodium sulfate + copper hydroxide Cu. SO 4(aq) + 2 Na. OH(aq) → Na 2 SO 4(aq) + Cu(OH)2(s) You can tell that the copper hydroxide forms a solid (the precipitate) because its state symbol is (s) for solid, rather than (aq) for aqueous (dissolved in water). HT only – write balanced half equations and ionic equations The reaction can also be shown by an ionic equation: Cu 2+(aq) + 2 OH–(aq) → Cu(OH)2(s)
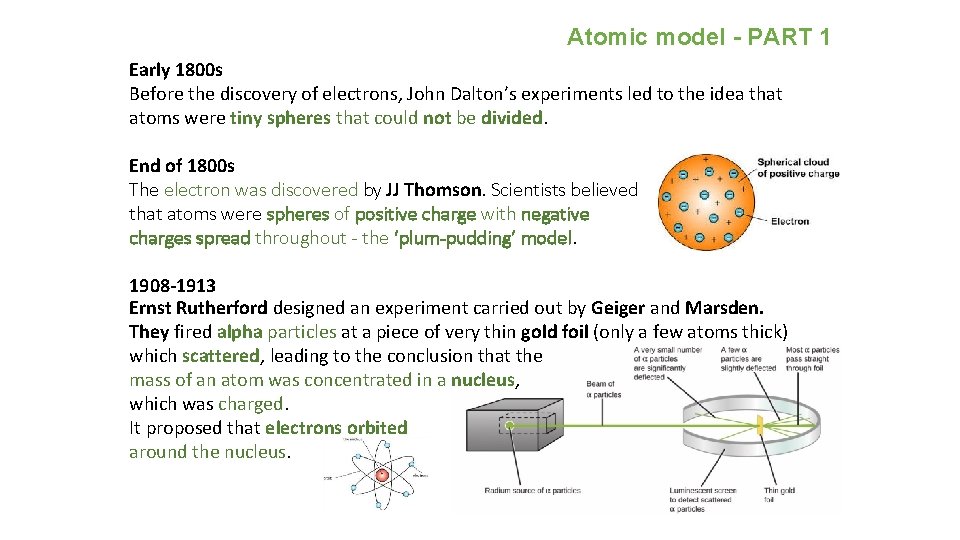
Atomic model - PART 1 Early 1800 s Before the discovery of electrons, John Dalton’s experiments led to the idea that atoms were tiny spheres that could not be divided. End of 1800 s The electron was discovered by JJ Thomson. Scientists believed that atoms were spheres of positive charge with negative charges spread throughout - the ‘plum-pudding’ model. 1908 -1913 Ernst Rutherford designed an experiment carried out by Geiger and Marsden. They fired alpha particles at a piece of very thin gold foil (only a few atoms thick) which scattered, leading to the conclusion that the mass of an atom was concentrated in a nucleus, which was charged. It proposed that electrons orbited around the nucleus.

Atomic model - PART 1 1914 Niels Bohr noticed that the light given out when atoms were heated only had specific amounts of energy and he adapted the nuclear model by suggesting that electrons orbit the nucleus at specific distances in certain fixed energy levels (or shells). The energy must be given out when excited electrons fall from a high to low energy level. Later experiments led to the idea that the positive charge of the nucleus could be subdivided into a whole number of smaller particles, each particle having the same amount of positive charge. The name proton was given to these particles. 1932 James Chadwick bombarded beryllium atoms with alpha particles. An unknown radiation was produced. Chadwick interpreted this radiation as being composed of particles with a neutral electrical charge and the approximate mass of a proton. This particle became known as the neutron.

Atomic model - PART 1 1800 s 1890 s 1908 -1913 1914 • John Dalton – tiny spheres that could not be divided. • JJ Thomson – electron discovered. Plum pudding model - spheres of positive charge with negative charges spread evenly though. • E. Rutherford, Geiger and Marsden - alpha particle scattering experiment. Nuclear model - mass of atom concentrated in a charged nucleus, with orbiting electrons. • Niels Bohr – electrons orbit nucleus at specific distances in fixed energy levels (shells). Energy given out when electrons change level. • Positive charge of nucleus could be subdivided into particles of positive Later… charge – protons. 1932 • James Chadwick – provided evidence for the existence of neutrons within the nucleus.
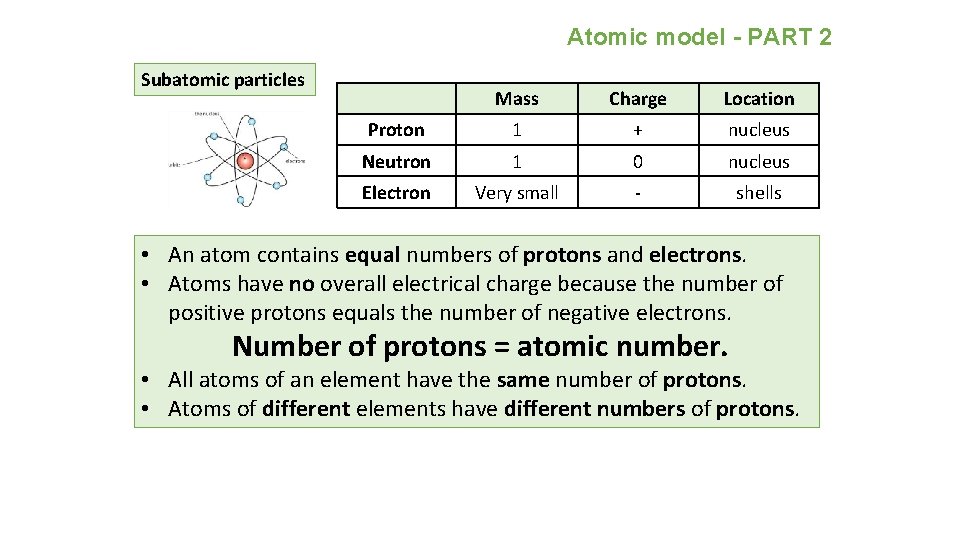
Atomic model - PART 2 Subatomic particles Mass Charge Location Proton 1 + nucleus Neutron 1 0 nucleus Electron Very small - shells • An atom contains equal numbers of protons and electrons. • Atoms have no overall electrical charge because the number of positive protons equals the number of negative electrons. Number of protons = atomic number. • All atoms of an element have the same number of protons. • Atoms of different elements have different numbers of protons.
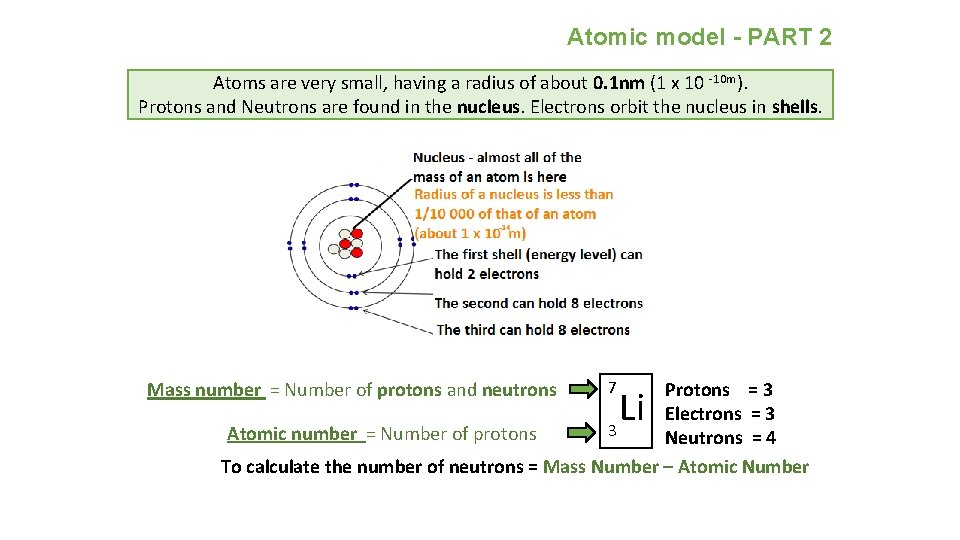
Atomic model - PART 2 Atoms are very small, having a radius of about 0. 1 nm (1 x 10 -10 m). Protons and Neutrons are found in the nucleus. Electrons orbit the nucleus in shells. Mass number = Number of protons and neutrons 7 Protons = 3 Electrons = 3 3 Atomic number = Number of protons Neutrons = 4 To calculate the number of neutrons = Mass Number – Atomic Number Li

Atomic model - PART 2 Atoms sometimes lose or gain electrons (e. g. when a metal reacts with a none metal). When they do this they become a charge atom or an ion. If an atom loses one or more electrons, it gains a positive charge because it has less electrons than protons. If an atom gains one or more electrons, it gains a negative charge because it has more electrons than protons. e. g. If sodium atom loses one electron, it forms a Na+ ion. It has 11 protons, 12 neutrons and 10 electrons. e. g. If an oxygen atom gains two electrons, it forms a O 2 - ion. It has 8 protons, 8 neutrons and 10 electrons.
Atoms are tiny - the radius of a typical atom is one tenth of a billionth of a meter.
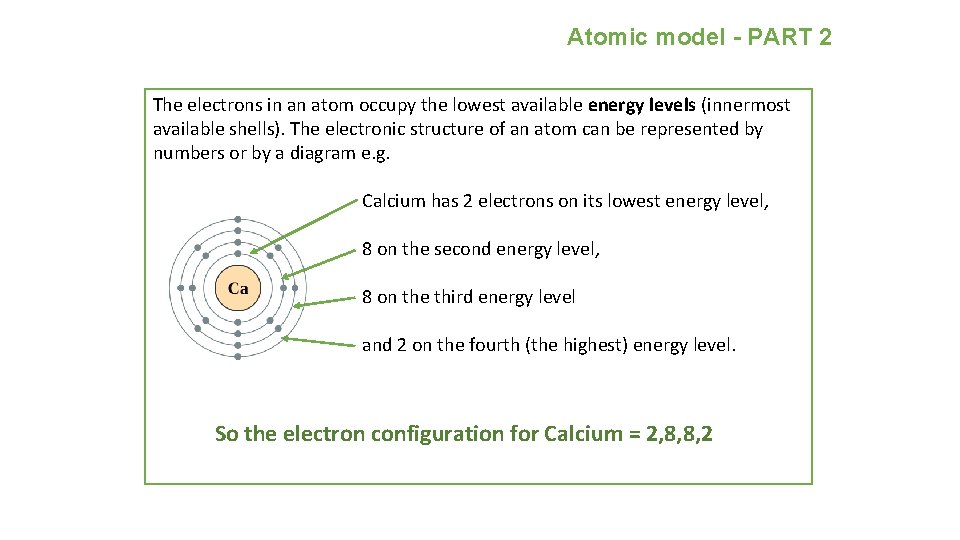
Atomic model - PART 2 The electrons in an atom occupy the lowest available energy levels (innermost available shells). The electronic structure of an atom can be represented by numbers or by a diagram e. g. Calcium has 2 electrons on its lowest energy level, 8 on the second energy level, 8 on the third energy level and 2 on the fourth (the highest) energy level. So the electron configuration for Calcium = 2, 8, 8, 2
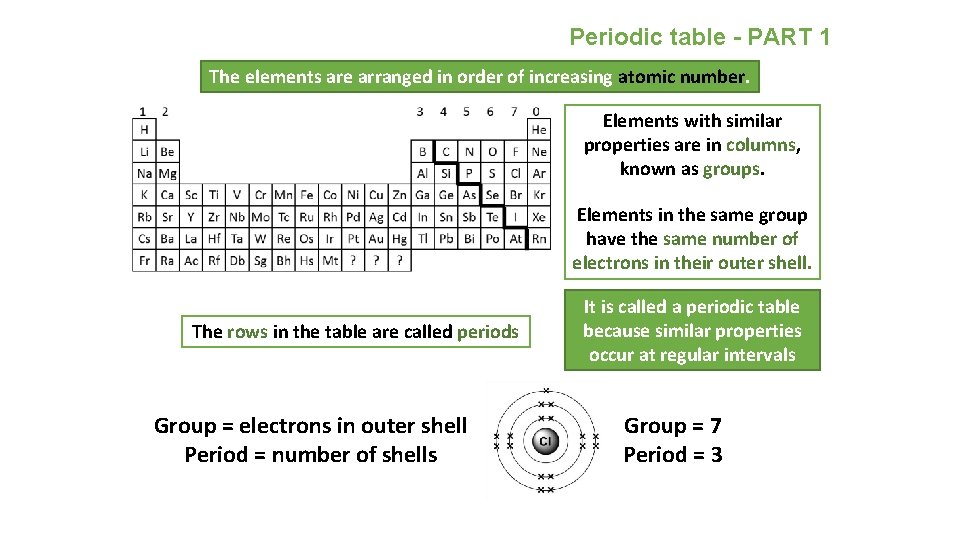
Periodic table - PART 1 The elements are arranged in order of increasing atomic number. Elements with similar properties are in columns, known as groups. Elements in the same group have the same number of electrons in their outer shell. The rows in the table are called periods Group = electrons in outer shell Period = number of shells It is called a periodic table because similar properties occur at regular intervals Group = 7 Period = 3
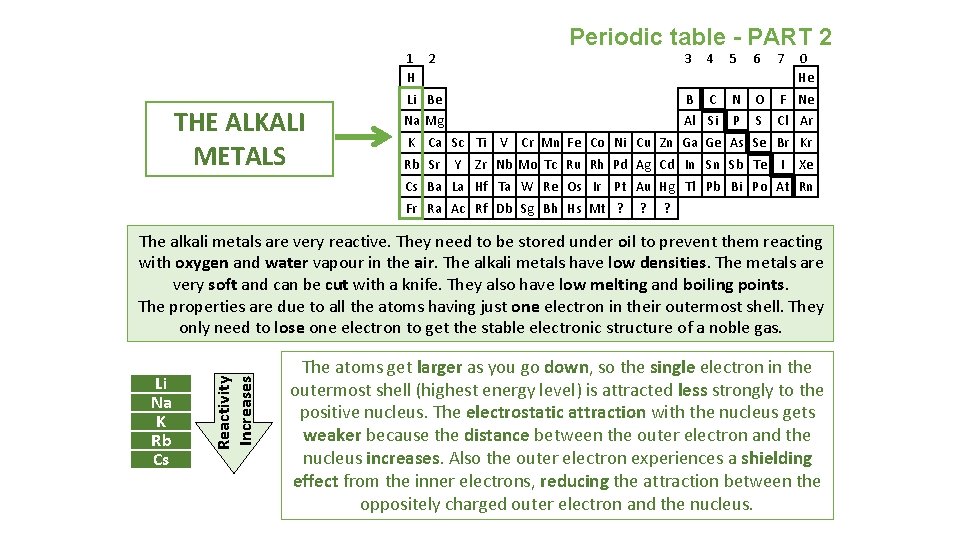
Periodic table - PART 2 1 2 H THE ALKALI METALS 3 4 5 6 7 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti Rb Sr Y 0 He V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt ? ? ? Li Na K Rb Cs Reactivity Increases The alkali metals are very reactive. They need to be stored under oil to prevent them reacting with oxygen and water vapour in the air. The alkali metals have low densities. The metals are very soft and can be cut with a knife. They also have low melting and boiling points. The properties are due to all the atoms having just one electron in their outermost shell. They only need to lose one electron to get the stable electronic structure of a noble gas. The atoms get larger as you go down, so the single electron in the outermost shell (highest energy level) is attracted less strongly to the positive nucleus. The electrostatic attraction with the nucleus gets weaker because the distance between the outer electron and the nucleus increases. Also the outer electron experiences a shielding effect from the inner electrons, reducing the attraction between the oppositely charged outer electron and the nucleus.

Periodic table - PART 2 The alkali metals have a silvery, shiny surface when they are first cut. However, this goes dull very quickly as the metals reacts with the oxygen in the air. e. g. sodium + oxygen sodium oxide 4 Na(s) + O 2(g) 2 Na 2 O(s) shiny metal oxide Lithium, sodium and potassium all react vigorously with water. When you add them to water, the metal floats, moves around and fizzes. e. g. potassium + water potassium hydroxide + hydrogen 2 K(s) + 2 H 2 O(l) 2 KOH(aq) + H 2(g) Potassium ignites with a lilac flame They also react vigorously with non metals, such as group seven. They form 1+ ions in the reactions to make ionic compounds. These are generally white and dissolve in water, giving colourless solutions. e. g. sodium + chlorine sodium chloride 2 Na(s) + Cl 2(g) 2 Na. Cl(s)
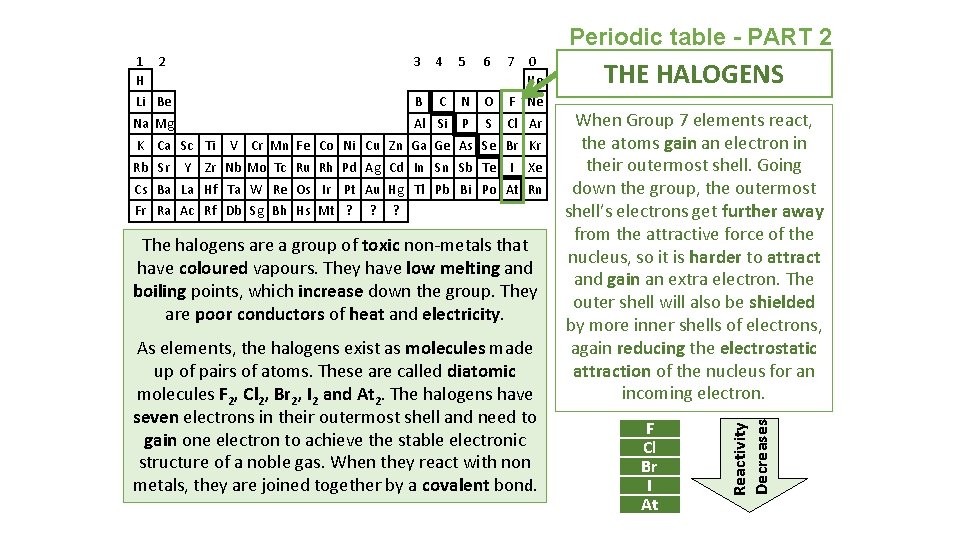
Periodic table - PART 2 3 4 5 6 7 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti Rb Sr Y 0 He V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt ? ? ? The halogens are a group of toxic non-metals that have coloured vapours. They have low melting and boiling points, which increase down the group. They are poor conductors of heat and electricity. As elements, the halogens exist as molecules made up of pairs of atoms. These are called diatomic molecules F 2, Cl 2, Br 2, I 2 and At 2. The halogens have seven electrons in their outermost shell and need to gain one electron to achieve the stable electronic structure of a noble gas. When they react with non metals, they are joined together by a covalent bond. THE HALOGENS When Group 7 elements react, the atoms gain an electron in their outermost shell. Going down the group, the outermost shell’s electrons get further away from the attractive force of the nucleus, so it is harder to attract and gain an extra electron. The outer shell will also be shielded by more inner shells of electrons, again reducing the electrostatic attraction of the nucleus for an incoming electron. F Cl Br I At Reactivity Decreases 1 2 H

Periodic table - PART 2 The halogens react with hydrogen. The reactions with hydrogen become less reactive as you go down the group. e. g. fluorine + hydrogen fluoride F 2(g) + H 2(g) 2 HF(g) The halogens also react with metals. The halogen atoms gain a single electron to give them a stable arrangement of electrons. They form ionic compound. e. g. sodium + chlorine sodium chloride 2 Na(s) + Cl 2(g) 2 Na. Cl(s) A more reactive halogen will also displace a less reactive halogen from solutions of its salts. e. g. chlorine + potassium bromide potassium chloride + bromine Cl 2(g) + 2 KBr(aq) 2 KCl(aq) + Br 2(aq) The colour of the solution after mixing depends on the less reactive pair of halogens. Cl 2(aq) Br 2(aq) I 2(aq)
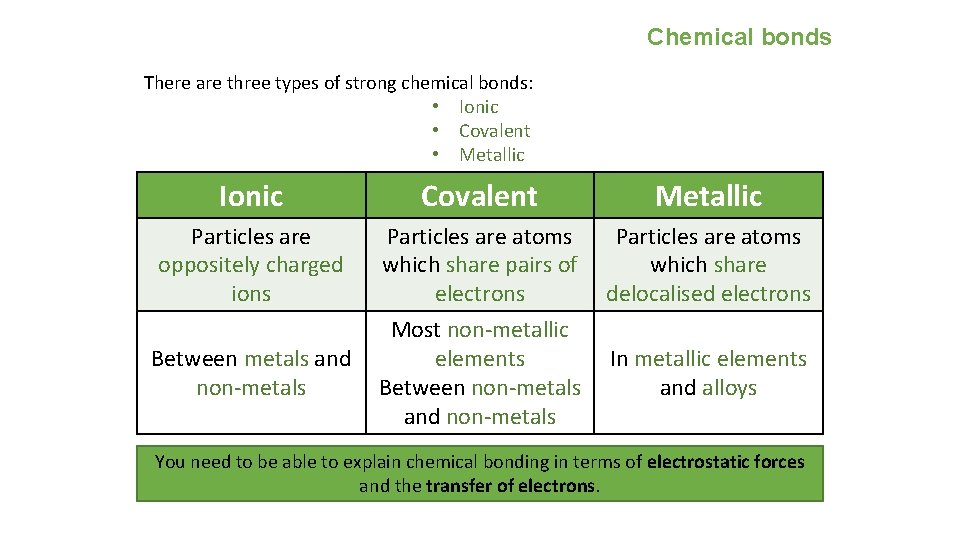
Chemical bonds There are three types of strong chemical bonds: • Ionic • Covalent • Metallic Ionic Particles are oppositely charged ions Covalent Metallic Particles are atoms which share pairs of which share electrons delocalised electrons Most non-metallic Between metals and elements In metallic elements non-metals Between non-metals and alloys and non-metals You need to be able to explain chemical bonding in terms of electrostatic forces and the transfer of electrons.

Ionic bonding Ionic bonds form between metals and non-metals. Ionic bonding involves the transfer of electrons in the outer shells. Metals lose electrons to become positively charged ions and non-metals gain electrons to become negatively charged ions. The elements in Group 1 react with the elements in Group 7. Groups 1 elements can each lose one electron. This electron can be given to an atom from Group 7, they both achieve the stable electronic structure of a noble gas.
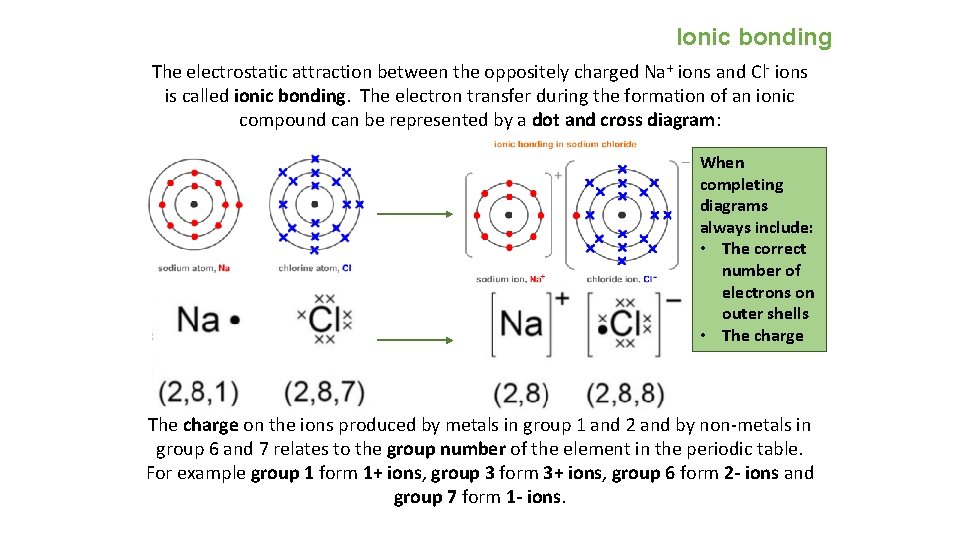
Ionic bonding The electrostatic attraction between the oppositely charged Na + ions and Cl- ions is called ionic bonding. The electron transfer during the formation of an ionic compound can be represented by a dot and cross diagram: When completing diagrams always include: • The correct number of electrons on outer shells • The charge on the ions produced by metals in group 1 and 2 and by non-metals in group 6 and 7 relates to the group number of the element in the periodic table. For example group 1 form 1+ ions, group 3 form 3+ ions, group 6 form 2 - ions and group 7 form 1 - ions.
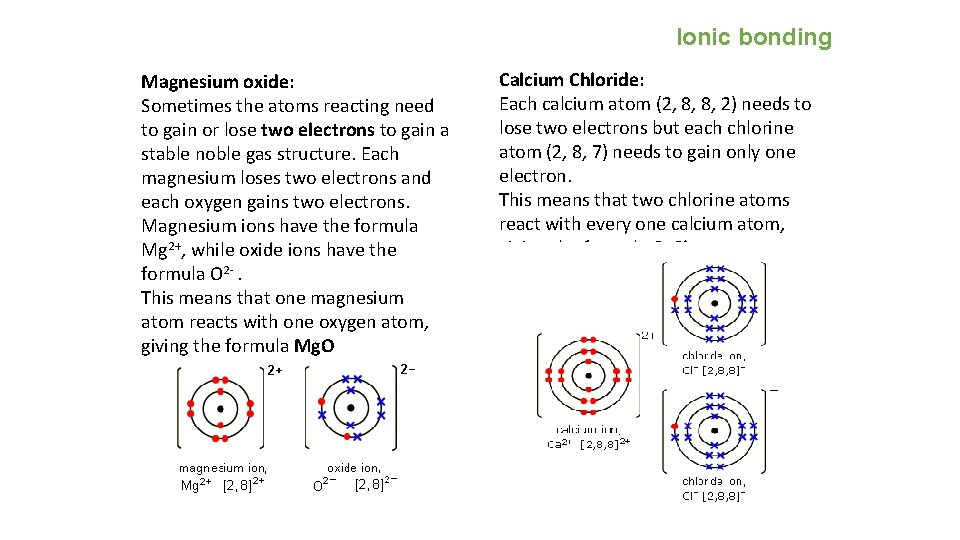
Ionic bonding Magnesium oxide: Sometimes the atoms reacting need to gain or lose two electrons to gain a stable noble gas structure. Each magnesium loses two electrons and each oxygen gains two electrons. Magnesium ions have the formula Mg 2+, while oxide ions have the formula O 2 -. This means that one magnesium atom reacts with one oxygen atom, giving the formula Mg. O Calcium Chloride: Each calcium atom (2, 8, 8, 2) needs to lose two electrons but each chlorine atom (2, 8, 7) needs to gain only one electron. This means that two chlorine atoms react with every one calcium atom, giving the formula Ca. Cl 2
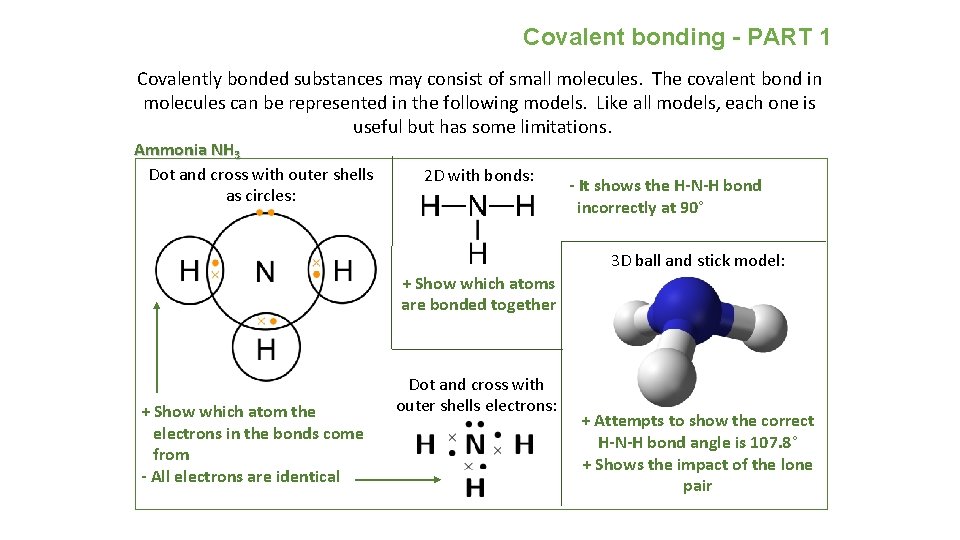
Covalent bonding - PART 1 Covalently bonded substances may consist of small molecules. The covalent bond in molecules can be represented in the following models. Like all models, each one is useful but has some limitations. Ammonia NH 3 Dot and cross with outer shells as circles: 2 D with bonds: - It shows the H-N-H bond incorrectly at 90° 3 D ball and stick model: + Show which atoms are bonded together + Show which atom the electrons in the bonds come from - All electrons are identical Dot and cross with outer shells electrons: + Attempts to show the correct H-N-H bond angle is 107. 8° + Shows the impact of the lone pair

Reactions of acids - PART 1 Acids react with some metals to produce salts and hydrogen. Metal + acid salt + hydrogen Reactions between metals and acids only occur if the metal is more reactive than the hydrogen in the acid. If the metal is too reactive, the reaction with acid is violent. The salt that is made depends on the metal and acid used. Salts made when metals react nitric acid are called nitrates. Zinc + Nitric acid Zinc Nitrate + Hydrogen Salts made when metals react with sulfuric acids are called sulfates. Iron + Sulfuric Acid Iron Sulfate + Hydrogen Salts made when metals react with hydrochloric acid are called chlorides. Magnesium + Hydrochloric acid Magnesium Chloride + Hydrogen

Reactions of acids - PART 1 - HIGHER In the reaction between magnesium and hydrochloric acid, the hydrogen ions are displaced from the solution by magnesium as the magnesium is more reactive than hydrogen. The following ionic equation occurs: Mg(s) + 2 H+(aq) Mg 2+(aq) + H 2(g) The chloride ions are not included as they do not change in the reaction. These are known as spectator ions. The reaction can be further represented by half equations, showing that the reaction between a metal and acid is a redox reaction. Mg 2+ + 2 e – The magnesium atoms lose two electrons, they have been oxidised. 2 H+ + 2 e - H 2 The hydrogen ions have gained electrons, they have been reduced.

Reactions of acids - PART 1 Acids are neutralised by alkalis (eg: soluble metal hydroxides) and bases (eg: insoluble metal hydroxides and metal oxides) to produce salts and water and by metal carbonates to produce salts, water and carbon dioxide. The salt name depends on the acid used and the positive ions in the alkali, base or carbonate. Making Soluble Salts from acids and alkalis Salts can be made by reacting an acid with an alkali. Acid + Alkali Salt + Water Making Soluble Salts from acids and bases Salts can be made by reacting an acid with a insoluble base. Acid + Bases Salt + Water Making Soluble Salts from acids and metal carbonates Salts can be made by reacting an acid with a metal carbonate. Acid + Metal carbonate Salt + Water + Carbon dioxide Salts are made of positive metal ions (or ammonia ions - NH 4+) and a negative ion from the acid. Like all ionic compounds, salts have no overall charge, so once you know the charges on the ions, you can work out the formula. Example: magnesium sulfate is Mg. SO 4 ion formula Group 1 Li+ Na+ K+ Transition metals Cu 2+ Fe 3+ Group 2 Mg 2+ Ca 2+ Group 7 F- Cl- Br- Aluminium Al 3+ Nitrate NO 3 - Sulphate SO 42 - Ammonium NH 4+

Reactions of acids - PART 1 Soluble salts can be made from acids by reacting them with solid insoluble substances, such as metals, metal oxides, hydroxides or carbonates. The solid is added to the acid until no more reacts and the excess solid is filtered off to produce a solution of the salt. Salt solutions can be crystallised to produce solid salts. You will complete this as a required practical. 1. Measure the required volume of acid with a measuring cylinder and add the weighed solid (insoluble metal, oxide, hydroxide or carbonate) in small portions with stirring. 2. Safety goggles required - the mixture may be heated to speed up the reaction. When no more of the solid dissolves it means ALL the acid is neutralised and there should be a little excess solid. You should see a residue of the solid (oxide, hydroxide, carbonate) left at the bottom of the beaker. 3. Filter the solution to remove the excess solid metal/oxide/carbonate, into an evaporating dish. On filtration, only a solution of the salt is left. 4. Then hot concentrated solution is left to cool and crystallise. After crystallisation, you collect and dry the crystals with a filter paper. If the solution is heated, the solvent will evaporate faster. Heating a solution until all the solvent has evaporated is known as heating to dryness.

Exothermic and endothermic reactions part 1 – Exothermic reactions Energy is conserved in chemical reactions. The amount of energy in the Universe at the end of a chemical reaction is the same as before the reaction takes place. H 2 (g) + Cl 2 (g) 2 HCl (g) In the above reaction energy is released, it gets hotter. An exothermic reaction is one that transfers energy to the surroundings so the temperature of the surroundings increases – “it gets hotter”. The two HCl molecules made will not hold as much energy as the H 2 and Cl 2 molecules at the start, so the spare energy is released as heat.

Exothermic and endothermic reactions part 1 – Exothermic reactions There a number of common exothermic reactions, they include: Combustion Know all three of these examples of exothermic reactions Oxidation Everyday uses of exothermic reactions include – Self-heating cans Hand warmers Neutralisation Know both of these uses for exothermic reactions

Exothermic and endothermic reactions part 1 – Endothermic reactions We have already learnt that energy is conserved in chemical reactions. 2 CH 3 COOH(aq) + Na 2 CO 3(s) 2 CH 3 COONa(aq) + CO 2(g) + H 2 O (l) In the above reaction, energy is taken in- it gets colder. An endothermic reaction is one that takes energy from the surroundings so the temperature of the surroundings decreases – “it gets colder”. The sodium ethanoate, carbon dioxide and water molecules made will hold more energy than the ethanoic acid and sodium carbonate molecules at the start, so the energy needed is taken in as heat. Know all three of Other examples of endothermic reactions are these examples of endothermic • Thermal decomposition reactions • Sports injury packs
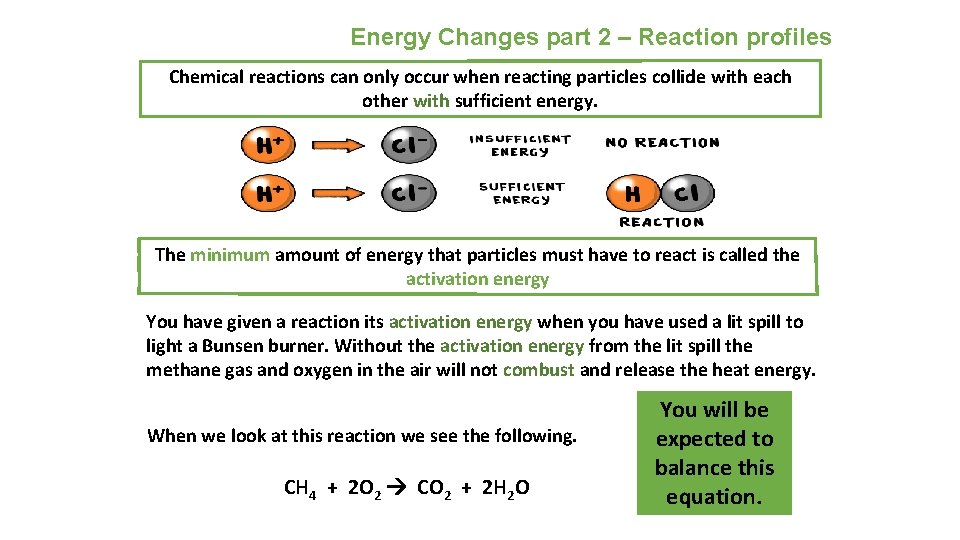
Energy Changes part 2 – Reaction profiles Chemical reactions can only occur when reacting particles collide with each other with sufficient energy. The minimum amount of energy that particles must have to react is called the activation energy You have given a reaction its activation energy when you have used a lit spill to light a Bunsen burner. Without the activation energy from the lit spill the methane gas and oxygen in the air will not combust and release the heat energy. When we look at this reaction we see the following. CH 4 + 2 O 2 CO 2 + 2 H 2 O You will be expected to balance this equation.
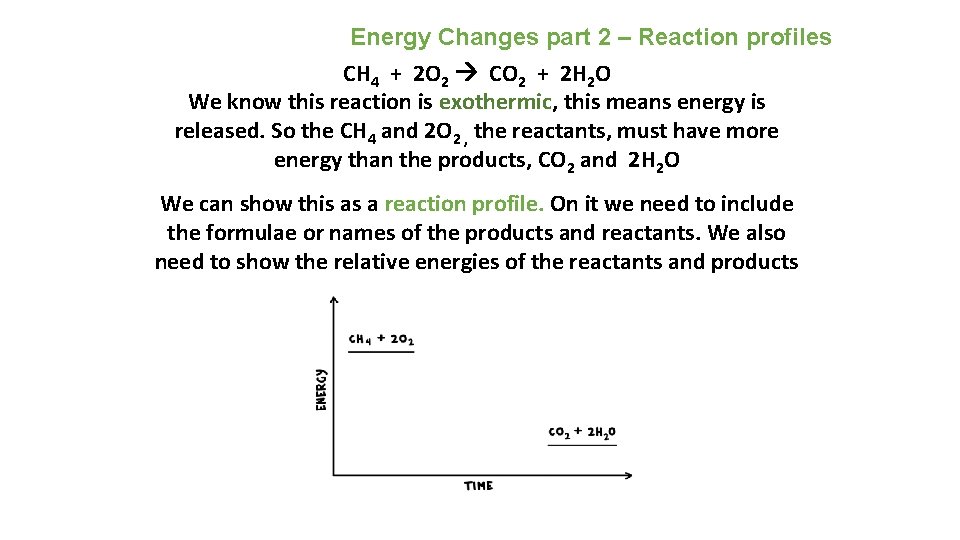
Energy Changes part 2 – Reaction profiles CH 4 + 2 O 2 CO 2 + 2 H 2 O We know this reaction is exothermic, this means energy is released. So the CH 4 and 2 O 2 , the reactants, must have more energy than the products, CO 2 and 2 H 2 O We can show this as a reaction profile. On it we need to include the formulae or names of the products and reactants. We also need to show the relative energies of the reactants and products e. g.
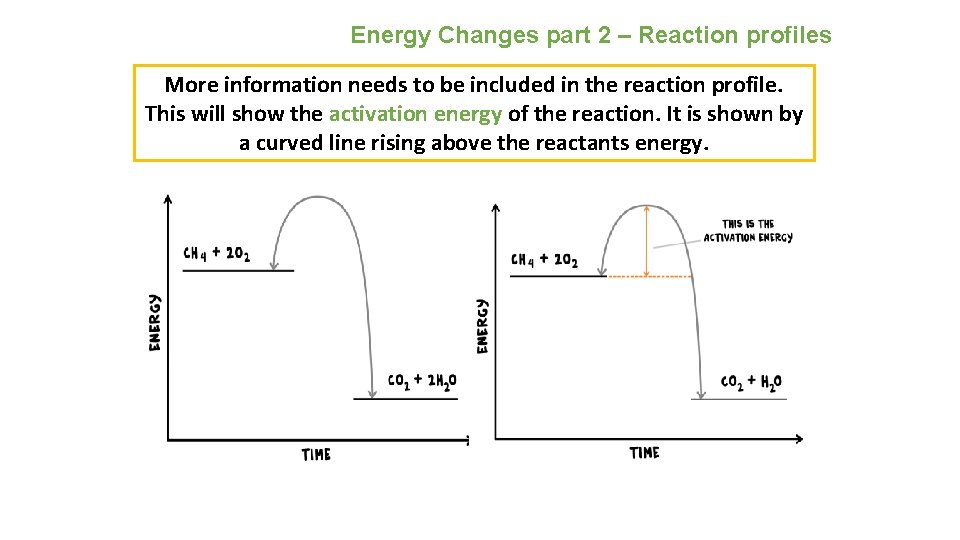
Energy Changes part 2 – Reaction profiles More information needs to be included in the reaction profile. This will show the activation energy of the reaction. It is shown by a curved line rising above the reactants energy.

Energy Changes part 2 – Reaction profiles We can now see the overall change in energy within the reaction. The products have less energy than the reactants. This will have been lost as heat as the reaction is exothermic.
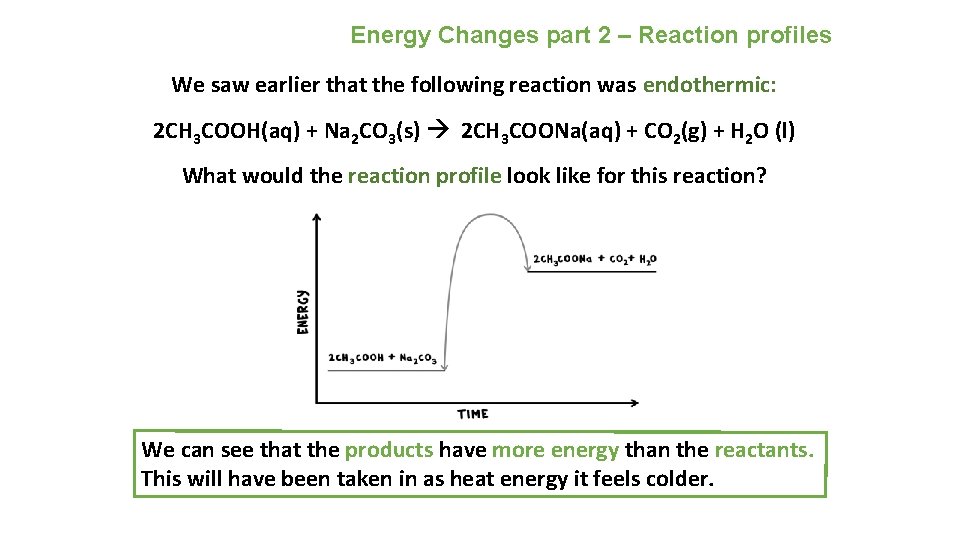
Energy Changes part 2 – Reaction profiles We saw earlier that the following reaction was endothermic: 2 CH 3 COOH(aq) + Na 2 CO 3(s) 2 CH 3 COONa(aq) + CO 2(g) + H 2 O (l) What would the reaction profile look like for this reaction? We can see that the products have more energy than the reactants. This will have been taken in as heat energy it feels colder.

The energy change of reactions (HT only) For the reaction of methane with oxygen we can write out the balanced symbol equation: CH 4 + 2 O 2 CO 2 + 2 H 2 O We can draw out the bonds between the atoms e. g. Each line represents a bond, two lines represent a double bond e. g. in the oxygen molecule.
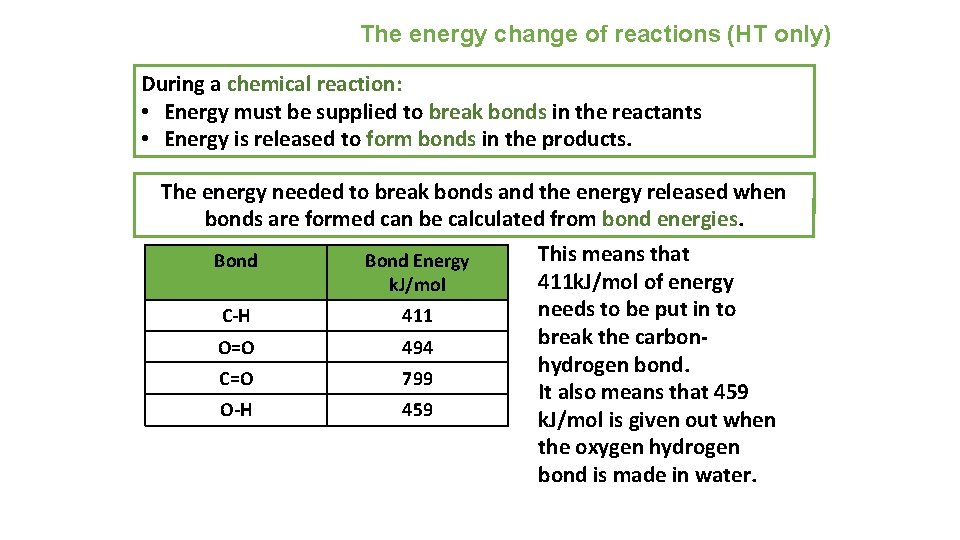
The energy change of reactions (HT only) During a chemical reaction: • Energy must be supplied to break bonds in the reactants • Energy is released to form bonds in the products. The energy needed to break bonds and the energy released when bonds are formed can be calculated from bond energies. Bond Energy k. J/mol C-H 411 O=O 494 C=O 799 O-H 459 This means that 411 k. J/mol of energy needs to be put in to break the carbonhydrogen bond. It also means that 459 k. J/mol is given out when the oxygen hydrogen bond is made in water.

The energy change of reactions (HT only) The difference between the sum of the energy needed to break bonds in the reactants and the sum of the energy released when bonds in the products are formed is the overall energy change of the reaction. Worked example For the reactants For the products There are four C-H bonds so 4 x 411 k. J/mol = 1, 644 k. J/mol There are two C=O bonds so 2 x 799 k. J/mol = 1, 598 k. J/mol There are two O=O bonds so 2 x 494 k. J/mol = 988 k. J/mol There are four O-H bonds so 4 x 459 k. J/mol = 1, 836 k. J/mol The sum of these is the energy supplied to break the bonds in the reactants it is 1, 644 + 988 = 2, 632 k. J/mol The sum of these is the energy released when bonds in the products are formed it is 1, 598 + 1, 836 = 3, 434 k. J/mol

The energy change of reactions (HT only) We already know that the difference between the sum of the energy needed to break bonds in the reactants and the sum of the energy released when bonds in the products are formed is the overall energy change of the reaction. This means that Overall energy change = energy needed to – energy released as break the bonds are made = 2, 632 k. J/mol - 3, 434 k. J/mol Overall energy change = - 802 k. J/mol This is an exothermic reaction, so the sum of the difference between the calculations is negative. For an endothermic reaction it would be positive. Students should be able to calculate the energy transferred in chemical reactions using bond energies supplied.

The energy change of reactions (HT only) Know these two definitions- they are often asked for in the exam. In an exothermic reaction, the energy released from forming new bonds is greater than the energy needed to break existing bonds In an endothermic reaction, the energy needed to break existing bonds is greater then the energy released from forming new bonds

Chemical measurements - PART 2 Relative formula mass (Mr) of a compound is the sum of the relative atomic masses of the atoms in the numbers shown in the formula. The relative atomic masses can be found in the periodic table Na. Cl H 2 O (1 x Na + 1 x Cl) Ar: Na (23) Cl (35. 5) (2 x H + 1 x O) Ar: H (1) O (16) Mr = 23 + 35. 5 = 58. 5 Mr = (1 x 2) + 16 = 18 H 2 SO 4 (2 x H + 1 x S + 4 x O) Ar: H (1) S (32) O (16) Al 2(SO 4)3 (2 x Al + 3 x S + 12 x O) Ar: Al (27) S (32) O (16) Mr = (1 x 2) + 32 + (16 x 4) = 98 Mr = (27 x 2) + (32 x 3) + (16 x 12) = 342 In a balanced chemical equation, the sum of the relative formula masses of the reactants equals the sum of the relative formula masses of the products. For example: 2 Mg + O 2 2 Mg. O (2 x 24) + (2 x 16) 2 x (24+16) 80
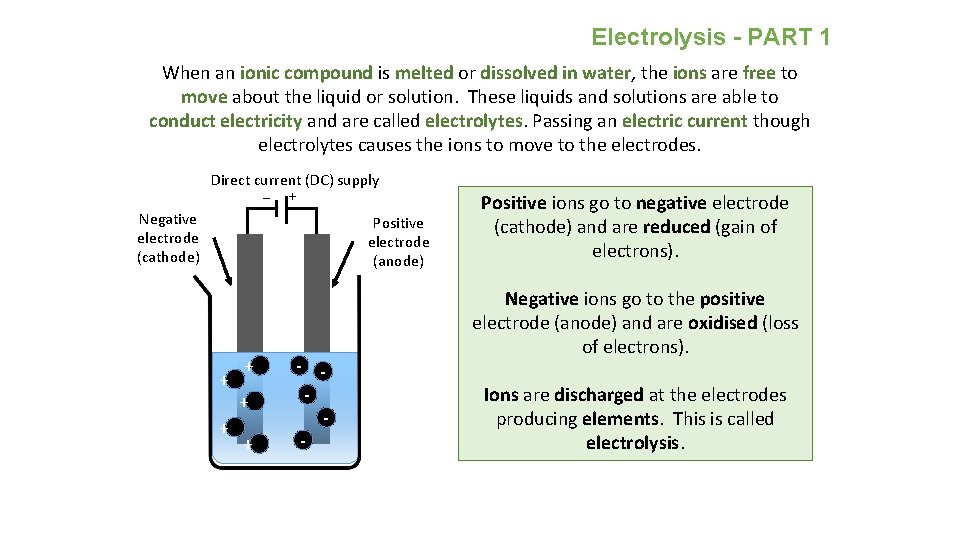
Electrolysis - PART 1 When an ionic compound is melted or dissolved in water, the ions are free to move about the liquid or solution. These liquids and solutions are able to conduct electricity and are called electrolytes. Passing an electric current though electrolytes causes the ions to move to the electrodes. Direct current (DC) supply _ + Negative electrode (cathode) Positive electrode (anode) + + + - - Positive ions go to negative electrode (cathode) and are reduced (gain of electrons). Negative ions go to the positive electrode (anode) and are oxidised (loss of electrons). Ions are discharged at the electrodes producing elements. This is called electrolysis.
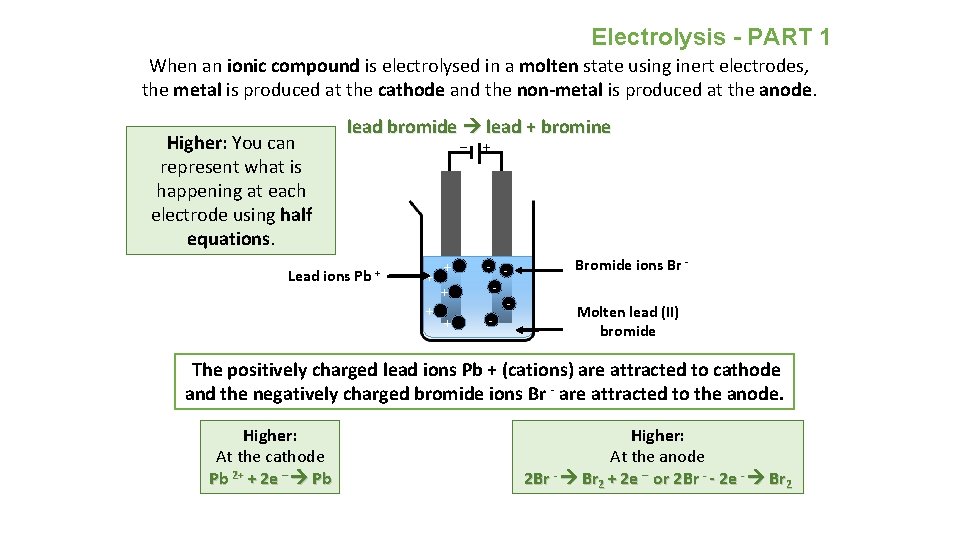
Electrolysis - PART 1 When an ionic compound is electrolysed in a molten state using inert electrodes, the metal is produced at the cathode and the non-metal is produced at the anode. Higher: You can represent what is happening at each electrode using half equations. lead bromide lead + bromine Lead ions Pb + _ + + + - - Bromide ions Br Molten lead (II) bromide The positively charged lead ions Pb + (cations) are attracted to cathode and the negatively charged bromide ions Br - are attracted to the anode. Higher: At the cathode Pb 2+ + 2 e – Pb Higher: At the anode 2 Br - Br 2 + 2 e – or 2 Br - - 2 e - Br 2
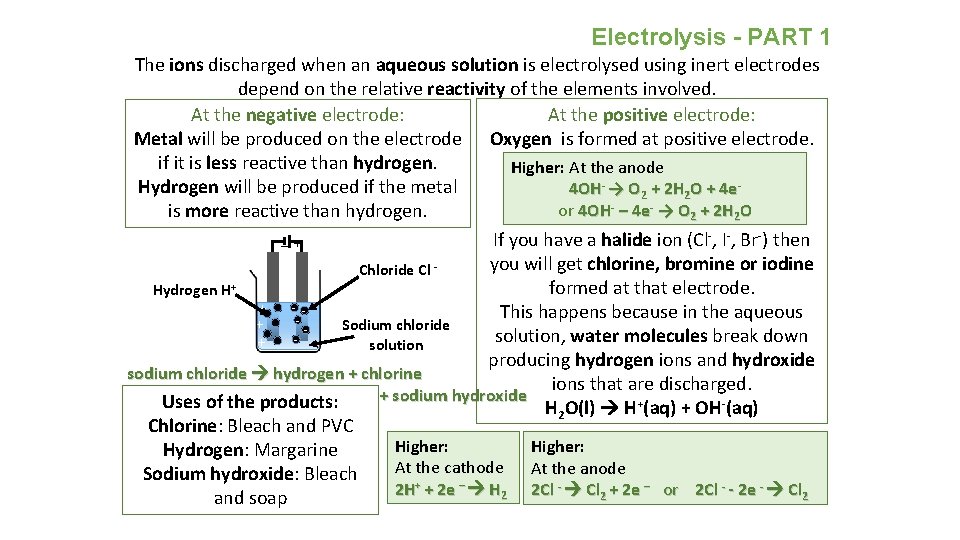
Electrolysis - PART 1 The ions discharged when an aqueous solution is electrolysed using inert electrodes depend on the relative reactivity of the elements involved. At the positive electrode: At the negative electrode: Metal will be produced on the electrode Oxygen is formed at positive electrode. if it is less reactive than hydrogen. Higher: At the anode Hydrogen will be produced if the metal 4 OH- → O 2 + 2 H 2 O + 4 eor 4 OH- – 4 e- → O 2 + 2 H 2 O is more reactive than hydrogen. _ + If you have a halide ion (Cl-, I-, Br-) then you will get chlorine, bromine or iodine Chloride Cl formed at that electrode. Hydrogen H+ - This happens because in the aqueous ++ + Sodium chloride ++ solution, water molecules break down solution producing hydrogen ions and hydroxide sodium chloride hydrogen + chlorine ions that are discharged. + sodium hydroxide Uses of the products: H 2 O(l) H+(aq) + OH-(aq) Chlorine: Bleach and PVC Higher: Hydrogen: Margarine At the cathode At the anode Sodium hydroxide: Bleach 2 H+ + 2 e – H 2 2 Cl - Cl 2 + 2 e – or 2 Cl - - 2 e - Cl 2 and soap
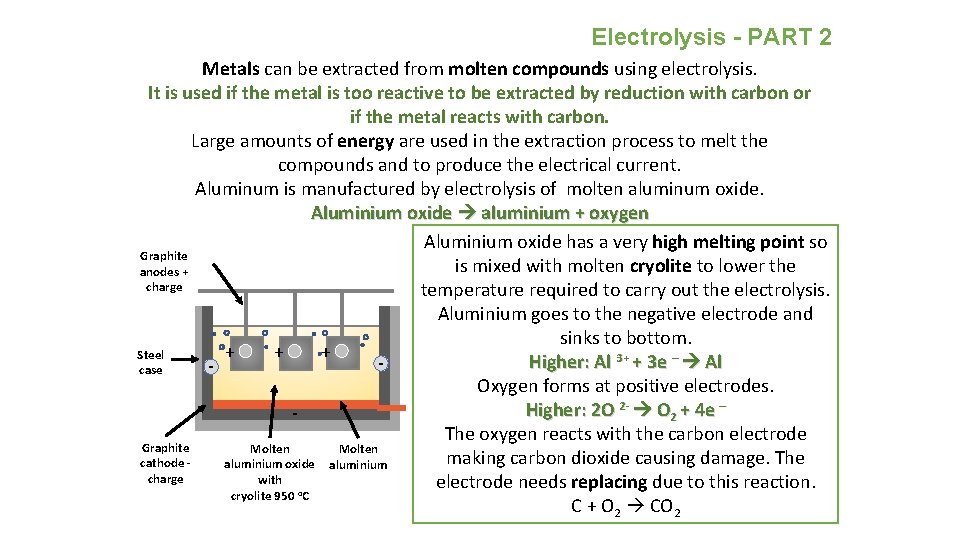
Electrolysis - PART 2 Metals can be extracted from molten compounds using electrolysis. It is used if the metal is too reactive to be extracted by reduction with carbon or if the metal reacts with carbon. Large amounts of energy are used in the extraction process to melt the compounds and to produce the electrical current. Aluminum is manufactured by electrolysis of molten aluminum oxide. Aluminium oxide aluminium + oxygen Aluminium oxide has a very high melting point so Graphite is mixed with molten cryolite to lower the anodes + charge temperature required to carry out the electrolysis. Aluminium goes to the negative electrode and sinks to bottom. + + + Steel Higher: Al 3+ + 3 e – Al case Oxygen forms at positive electrodes. Higher: 2 O 2 - O 2 + 4 e – The oxygen reacts with the carbon electrode Graphite Molten making carbon dioxide causing damage. The cathode - aluminium oxide aluminium charge with electrode needs replacing due to this reaction. cryolite 950 o. C C + O 2 CO 2 -
- Slides: 46