Whiteboarding Ideal Gases Five points representing five different

+ Whiteboarding! Ideal Gases Five points representing five different states of one mole of an ideal gas are labeled on the pressure—volume graph below. Rank the temperatures of the ideal gas in the five labeled states. Explain your reasoning.

+ Whiteboarding! Ideal Gases Five points representing five different states of one mole of an ideal gas are labeled on the pressure—volume graph below. Rank the temperatures of the ideal gas in the five labeled states. Explain your reasoning. Answer: B > C > A > E > D. As prescribed in the Ideal Gas Law the temperature in each state is proportional to the product of the pressure and volume

+ Whiteboarding! Ideal Gases A cylinder contains two samples of different ideal gases. A piston separating the two gases is free to move without friction. Each gas occupies half of the cylinder, but there are twice as many molecules on the left side of the piston as on the right, and the absolute temperature is twice as large for the gas on the right side as for the gas on the left. Is the pressure in the gas on the left side greater than, less than, or equal to the pressure in the gas on the right side? Explain.

+ Whiteboarding! Ideal Gases A cylinder contains two samples of different ideal gases. A piston separating the two gases is free to move without friction. Each gas occupies half of the cylinder, but there are twice as many molecules on the left side of the piston as on the right, and the absolute temperature is twice as large for the gas on the right side as for the gas on the left. Is the pressure in the gas on the left side greater than, less than, or equal to the pressure in the gas on the right side? Explain. Answer: They are equal. The volumes of the two gases are the same and the products of the number of moles and the absolute temperatures of the gases are also the same.
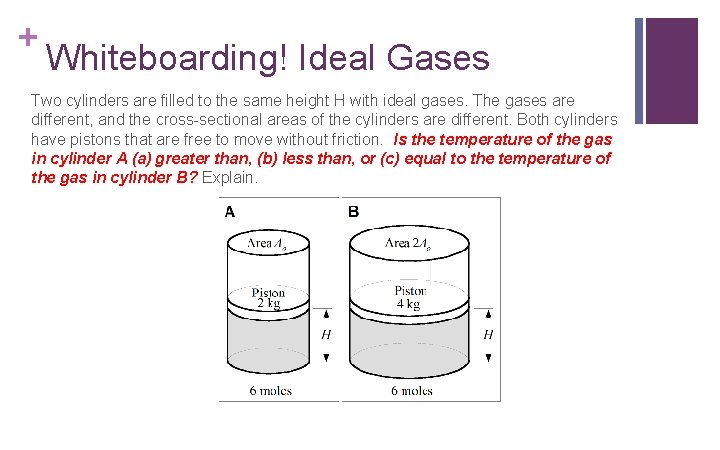
+ Whiteboarding! Ideal Gases Two cylinders are filled to the same height H with ideal gases. The gases are different, and the cross-sectional areas of the cylinders are different. Both cylinders have pistons that are free to move without friction. Is the temperature of the gas in cylinder A (a) greater than, (b) less than, or (c) equal to the temperature of the gas in cylinder B? Explain.

+ Whiteboarding! Ideal Gases Two cylinders are filled to the same height H with ideal gases. The gases are different, and the cross-sectional areas of the cylinders are different. Both cylinders have pistons that are free to move without friction. Is the temperature of the gas in cylinder A (a) greater than, (b) less than, or (c) equal to the temperature of the gas in cylinder B? Explain. Less than. The volume of the gas in case B is twice the volume of the gas in case A, and the pressures are the same (WHAT? ? ? WHY? ? ? ), as are the number of moles. From the ideal gas law, then, the absolute temperature in case B will be double the absolute temperature in case A.

+ AP Physics 2 – Class Starter Quiz 4: Ideal Gases 1. Clear everything from your desks and take out your quiz notebooks! 2. Collect the Data Sheets from the front of the room 3. Set up dividers to separate yourself from your neighbours!
+ AP Physics 2 – Ideal Gases Please take out your notes to section 5 a – Ideal Gas Processes

+ Reading n Reading - 1 st Law Thermodynamic Processes
- Slides: 9