Which Patients Should Be Treated with Monotherapy and

Which Patients Should Be Treated with Monotherapy and Is There Any Role for Long-Term ASPIRIN? Roxana Mehran, MD, FESC, FACC, FAHA, MSCAI Mount Sinai Professor in Cardiovascular Clinical Research and Outcomes Director of Interventional Cardiovascular Research and Clinical Trials, Icahn School of Medicine at Mount Sinai, New York, NY, USA

Disclosures Affiliation/Financial Relationship Company Consultant/Advisory/Speaking Engagements Boston Scientific, Medscape, Siemens Medical Solutions, Roivant Sciences Inc, Sanofi, Janssen, Abbott Laboratories, Medtelligence (Janssen Scientific Affairs), Idorsia Pharmaceuticals Ltd. (no fee), Regeneron Pharmaceuticals (no fee), Abiomed (spouse), The Medicines Company (spouse), Abbott Laboratories (to institution), Spectranetics/Philips/Volcano Corp (to institution) Research Funding to Institution Abbott Laboratories, Astra Zeneca, Bayer, Beth Israel Deaconess, BMS, CSL Behring, DSI, Medtronic, Novartis, Orbus. Neich Scientific Advisory Board PLx Opco Inc/dba PLx Pharma Inc, Medtelligence (Janssen Scientific Affairs), Bristol. Myers Squibb (to institute), Equity, <1% Claret Medical, Elixir Medical DSMB membership paid to the institution Watermark Research Partners Associate Editor ACC, AMA
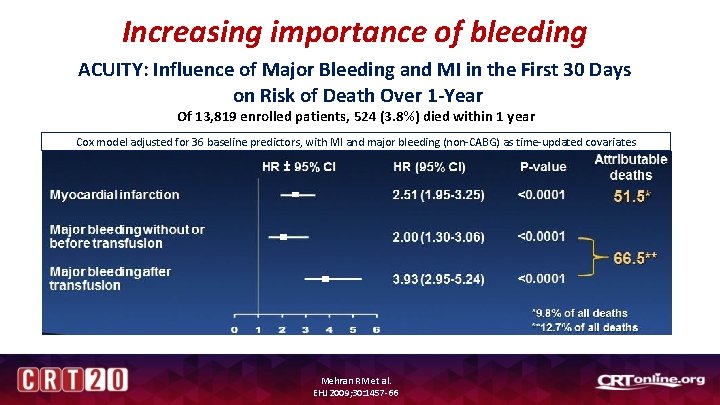
Increasing importance of bleeding ACUITY: Influence of Major Bleeding and MI in the First 30 Days on Risk of Death Over 1 -Year Of 13, 819 enrolled patients, 524 (3. 8%) died within 1 year Cox model adjusted for 36 baseline predictors, with MI and major bleeding (non-CABG) as time-updated covariates Mehran RM et al. EHJ 2009; 30: 1457 -66

Increasing importance of bleeding TRACER: Influence of Major Bleeding and MI after the First 30 Days on Risk of Death Over 1 Year 12, 944 patients with non-ST elevation ACS from the TRACER trial Valgimigli M, et al. Eur Heart J. 2017 Mar 14; 38(11): 804 -810
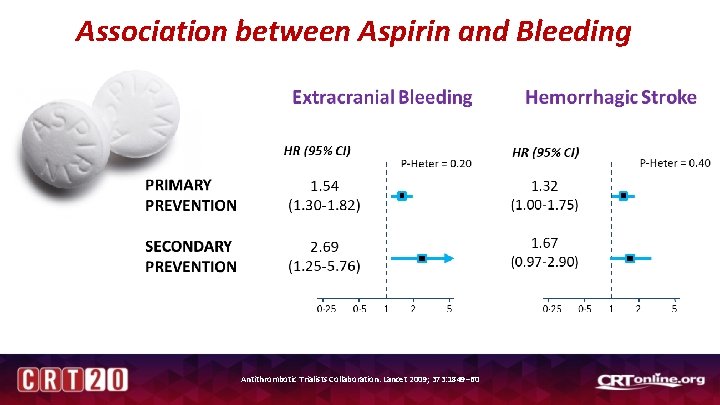
Association between Aspirin and Bleeding Antithrombotic Trialists Collaboration. Lancet 2009; 373: 1849– 60
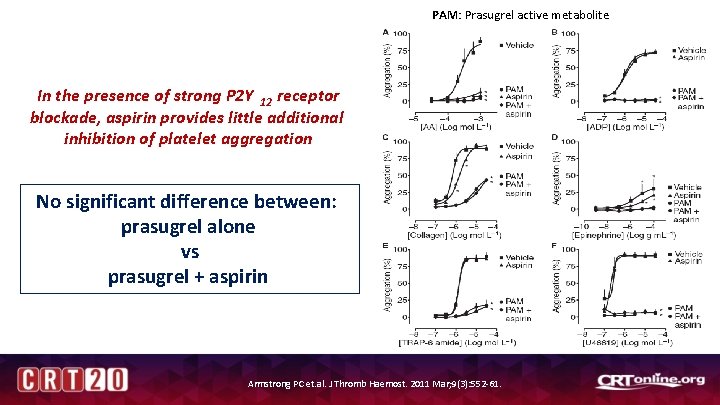
PAM: Prasugrel active metabolite In the presence of strong P 2 Y 12 receptor blockade, aspirin provides little additional inhibition of platelet aggregation No significant difference between: prasugrel alone vs prasugrel + aspirin Armstrong PC et. al. J Thromb Haemost. 2011 Mar; 9(3): 552 -61.
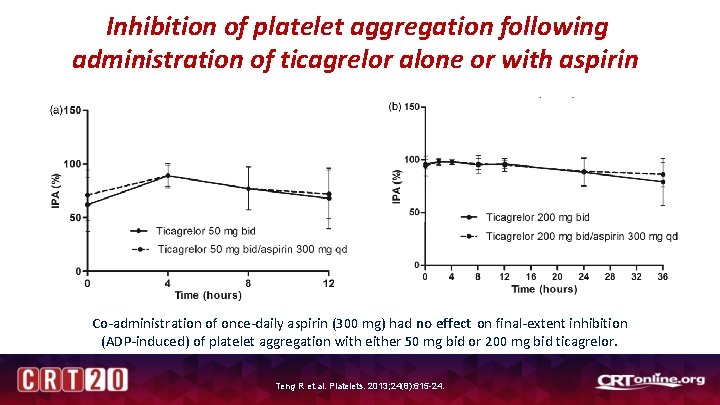
Inhibition of platelet aggregation following administration of ticagrelor alone or with aspirin Co-administration of once-daily aspirin (300 mg) had no effect on final-extent inhibition (ADP-induced) of platelet aggregation with either 50 mg bid or 200 mg bid ticagrelor. Teng R et. al. Platelets. 2013; 24(8): 615 -24.
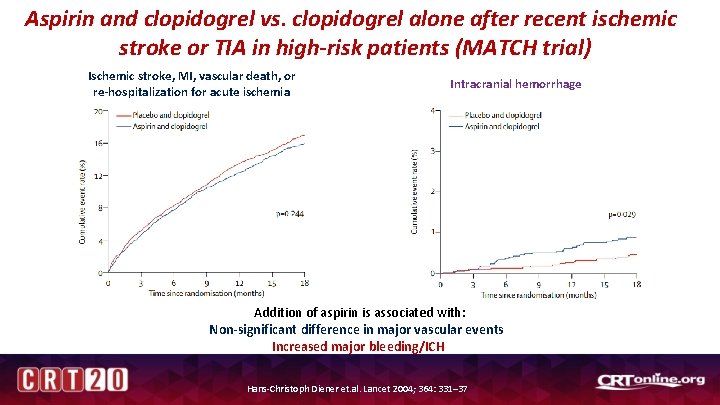
Aspirin and clopidogrel vs. clopidogrel alone after recent ischemic stroke or TIA in high-risk patients (MATCH trial) Ischemic stroke, MI, vascular death, or re-hospitalization for acute ischemia Intracranial hemorrhage Addition of aspirin is associated with: Non-significant difference in major vascular events Increased major bleeding/ICH Hans-Christoph Diener et. al. Lancet 2004; 364: 331– 37
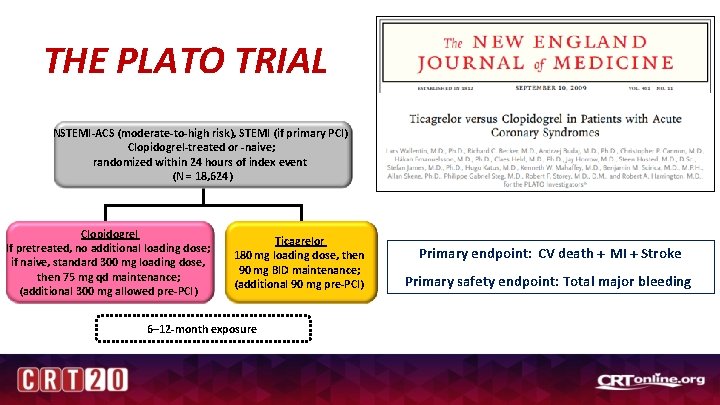
THE PLATO TRIAL NSTEMI-ACS (moderate-to-high risk), STEMI (if primary PCI) Clopidogrel-treated or -naive; randomized within 24 hours of index event (N = 18, 624) Clopidogrel If pretreated, no additional loading dose; if naive, standard 300 mg loading dose, then 75 mg qd maintenance; (additional 300 mg allowed pre-PCI) Ticagrelor 180 mg loading dose, then 90 mg BID maintenance; (additional 90 mg pre-PCI) 6– 12 -month exposure Primary endpoint: CV death + MI + Stroke Primary safety endpoint: Total major bleeding
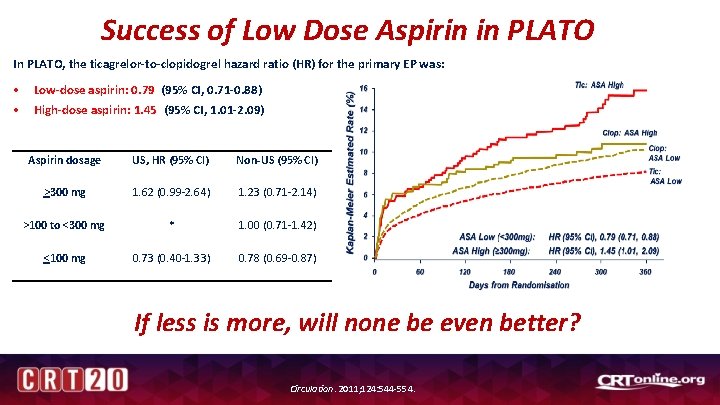
Success of Low Dose Aspirin in PLATO In PLATO, the ticagrelor-to-clopidogrel hazard ratio (HR) for the primary EP was: • Low-dose aspirin: 0. 79 (95% CI, 0. 71 -0. 88) • High-dose aspirin: 1. 45 (95% CI, 1. 01 -2. 09) Aspirin dosage US, HR (95% CI) Non-US (95% CI) >300 mg 1. 62 (0. 99 -2. 64) 1. 23 (0. 71 -2. 14) >100 to <300 mg * 1. 00 (0. 71 -1. 42) <100 mg 0. 73 (0. 40 -1. 33) 0. 78 (0. 69 -0. 87) If less is more, will none be even better? Circulation. 2011; 124: 544 -554.

Stacking: An Unappreciated Enemy Chronic oral anticoagulation Routine post-PCI LMWH GPIIb/IIIa inhibitors UFH → Bivalirudin Clopidogrel → Prasugrel / Ticagrelor Aspirin (81 mg – 325 mg) Courtesy: Gregg W. Stone

Recently, there has been more recognition for trials that withdraw rather than add to current treatments “…we need clinical trials that can investigate the withdrawal of certain established medications to see whether such withdrawal induces patient benefit, harm, or no difference compared with continued medication. ” - Stuart Pocock, Ph. D & Bernard Gersh, MB, Ch. B, DPhil Pocock, S. J. and B. J. Gersh (2014). "Do Current Clinical Trials Meet Society’s Needs? " J Am Coll Cardiol 64(15): 1615 -1628.
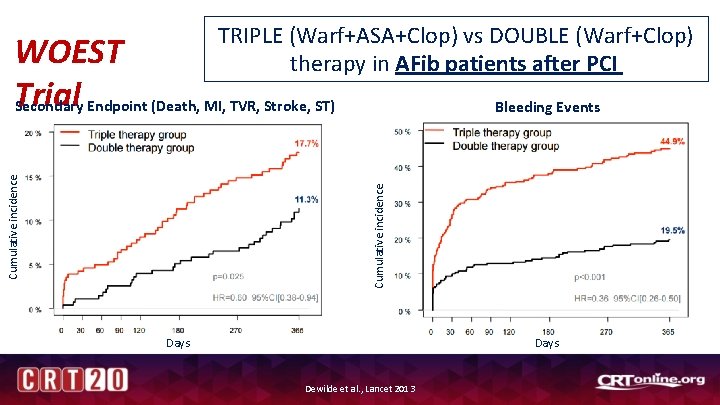
TRIPLE (Warf+ASA+Clop) vs DOUBLE (Warf+Clop) therapy in AFib patients after PCI Bleeding Events Cumulative incidence WOEST Trial Secondary Endpoint (Death, MI, TVR, Stroke, ST) Days Dewilde et al. , Lancet 2013
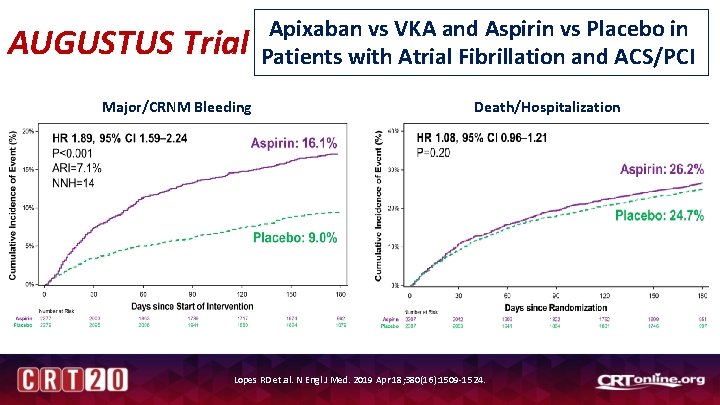
AUGUSTUS Trial Major/CRNM Bleeding Apixaban vs VKA and Aspirin vs Placebo in Patients with Atrial Fibrillation and ACS/PCI Death/Hospitalization Lopes RD et. al. N Engl J Med. 2019 Apr 18; 380(16): 1509 -1524.
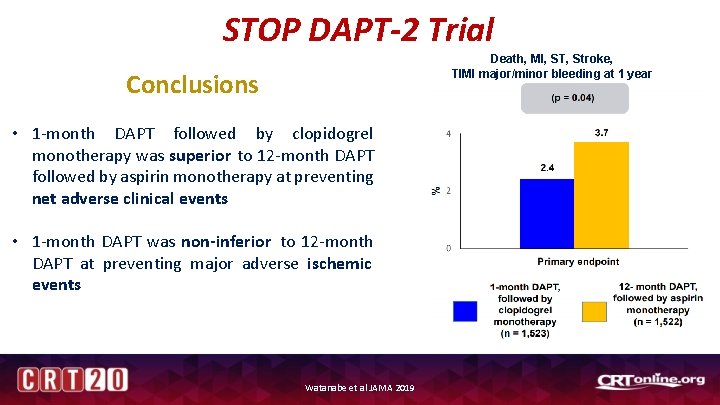
STOP DAPT-2 Trial Death, MI, ST, Stroke, TIMI major/minor bleeding at 1 year Conclusions • 1 -month DAPT followed by clopidogrel monotherapy was superior to 12 -month DAPT followed by aspirin monotherapy at preventing net adverse clinical events • 1 -month DAPT was non-inferior to 12 -month DAPT at preventing major adverse ischemic events Watanabe et al JAMA 2019
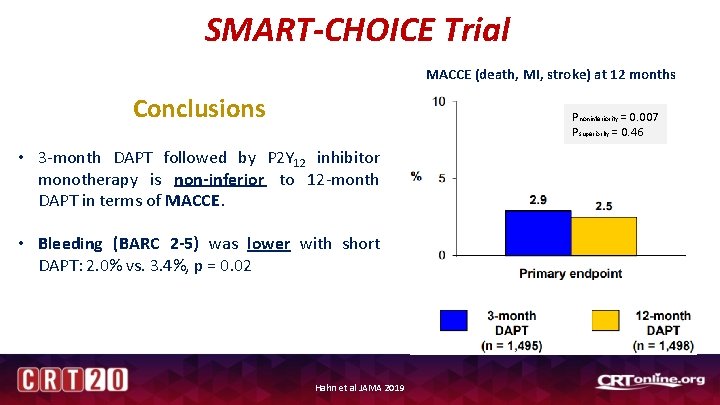
SMART-CHOICE Trial MACCE (death, MI, stroke) at 12 months Conclusions Pnoninferiority = 0. 007 Psuperiority = 0. 46 • 3 -month DAPT followed by P 2 Y 12 inhibitor monotherapy is non-inferior to 12 -month DAPT in terms of MACCE. • Bleeding (BARC 2 -5) was lower with short DAPT: 2. 0% vs. 3. 4%, p = 0. 02 Hahn et al JAMA 2019

Can we safely withdraw aspirin, when novel, more potent P 2 Y 12 inhibitors are used?
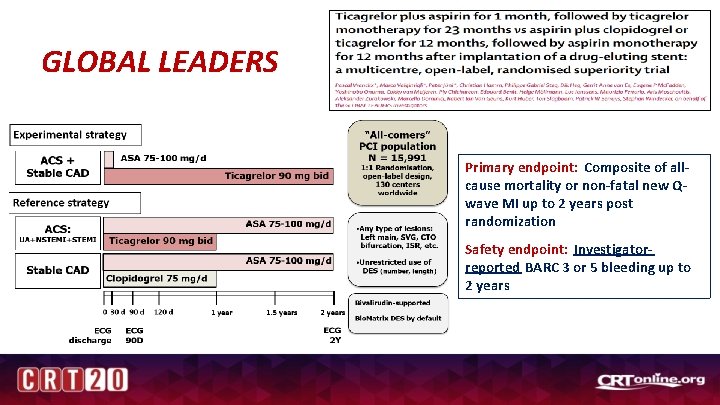
GLOBAL LEADERS Primary endpoint: Composite of allcause mortality or non-fatal new Qwave MI up to 2 years post randomization Safety endpoint: Investigatorreported BARC 3 or 5 bleeding up to 2 years
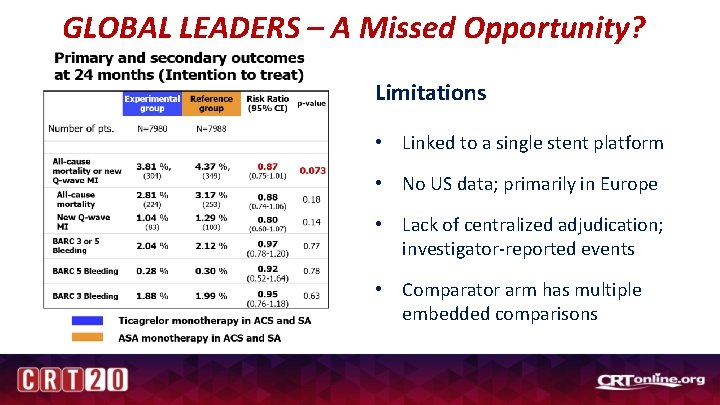
GLOBAL LEADERS – A Missed Opportunity? Limitations • Linked to a single stent platform • No US data; primarily in Europe • Lack of centralized adjudication; investigator-reported events • Comparator arm has multiple embedded comparisons

Advent of potent P 2 Y 12 inhibitors Unanswered questions after PLATO Increased focus on bleeding Concern about antiplatelet stacking Missed opportunity with GLOBAL LEADERS? Aspirin-related bleeding: lack of benefit in primary prevention The perfect storm to test the need for ASA

TWILIGHT Trial Objectives Primary Objective: To determine the impact of SAPT (ticagrelor monotherapy) versus DAPT (ticagrelor plus aspirin) for 12 months in reducing clinically relevant bleeding (BARC 2, 3 or 5) among high-risk patients who have undergone successful PCI. Secondary Objective: To determine the impact of SAPT (ticagrelor monotherapy) versus DAPT (ticagrelor plus aspirin) for 12 months on major ischemic adverse events (all-cause death, non-fatal MI or stroke) among high-risk patients who have undergone successful PCI.

Methods TWILIGHT was a randomized, double-blinded, placebo-controlled trial conducted in 187 sites across 11 countries. Patients undergoing successful PCI with at least 1 locally-approved DES whom the treating clinician intended to discharge on ticagrelor plus aspirin were eligible to participate. Trial inclusion required the presence of at least 1 additional clinical and angiographic feature associated with a high risk of ischemic or bleeding events.

TWILIGHT Inclusion Criteria Clinical criteria Angiographic criteria Age ≥ 65 years Multivessel CAD Female gender Target lesion requiring total stent length >30 mm Troponin positive ACS Thrombotic target lesion Established vascular disease (previous MI, documented PAD or CAD/PAD revasc) Bifurcation lesion(s) with Medina X, 1, 1 classification requiring ≥ 2 stents DM treated with medications or insulin Left main (≥ 50%) or proximal LAD (≥ 70%) lesions CKD (e. GFR <60 ml/min/1. 73 m 2 or Cr. Cl <60 ml/min) Calcified target lesion(s) requiring atherectomy

TWILIGHT Exclusion Criteria x Under 18 years of age x Liver cirrhosis x Contraindication to aspirin or ticagrelor x Life expectancy <1 year x Planned surgery within 90 days x Unable or unwilling to provide informed consent x Planned coronary revascularization (surgical or percutaneous) within 90 days x Need for chronic oral anticoagulation x Prior stroke x Dialysis-dependent renal failure x Active bleeding or extreme-risk for major bleeding x Salvage PCI or STEMI presentation x Women of child bearing potential x Fibrinolytic therapy within 24 hours of index PCI x Concomitant therapy with a strong cytochrome P-450 3 A inhibitor or inducer x Platelet count <100, 000 mm 3 x Requiring ongoing treatment with aspirin ≥ 325 mg daily #AHA 19
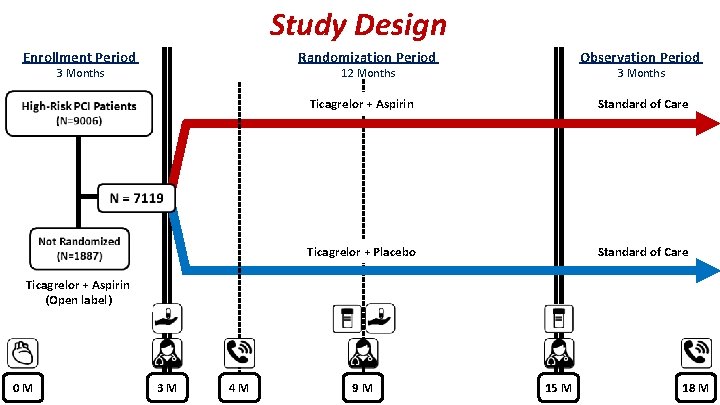
Study Design Enrollment Period Randomization Period 3 Months Observation Period 12 Months 3 Months Ticagrelor + Aspirin Standard of Care Ticagrelor + Placebo Standard of Care Ticagrelor + Aspirin (Open label) 0 M 3 M 4 M 9 M 15 M 18 M
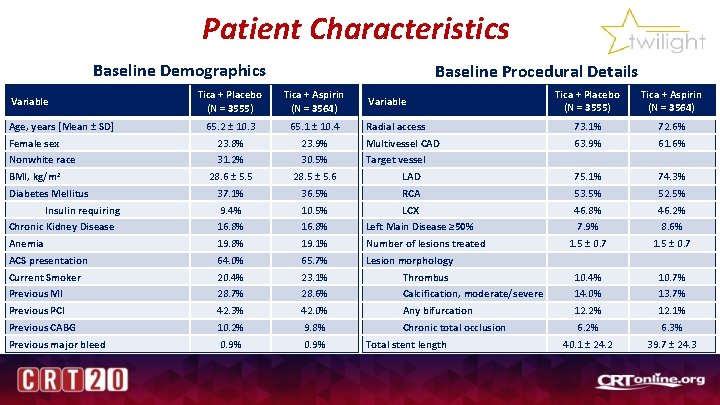
Patient Characteristics Baseline Demographics Baseline Procedural Details Tica + Placebo (N = 3555) Tica + Aspirin (N = 3564) Radial access 73. 1% 72. 6% Multivessel CAD 63. 9% 61. 6% LAD 75. 1% 74. 3% 36. 5% RCA 53. 5% 52. 5% 9. 4% 10. 5% LCX 46. 8% 46. 2% Chronic Kidney Disease 16. 8% Left Main Disease ≥ 50% 7. 9% 8. 6% Anemia 19. 8% 19. 1% Number of lesions treated 1. 5 ± 0. 7 ACS presentation 64. 0% 65. 7% Lesion morphology Current Smoker 20. 4% 23. 1% Thrombus 10. 4% 10. 7% Previous MI 28. 7% 28. 6% Calcification, moderate/severe 14. 0% 13. 7% Previous PCI 42. 3% 42. 0% Any bifurcation 12. 2% 12. 1% Previous CABG 10. 2% 9. 8% Chronic total occlusion 6. 2% 6. 3% Previous major bleed 0. 9% 40. 1 ± 24. 2 39. 7 ± 24. 3 Variable Age, years [Mean ± SD] Female sex Nonwhite race BMI, kg/m 2 Diabetes Mellitus Insulin requiring Tica + Placebo (N = 3555) Tica + Aspirin (N = 3564) 65. 2 ± 10. 3 23. 8% 65. 1 ± 10. 4 23. 9% 31. 2% 30. 5% 28. 6 ± 5. 5 28. 5 ± 5. 6 37. 1% Variable Target vessel Total stent length
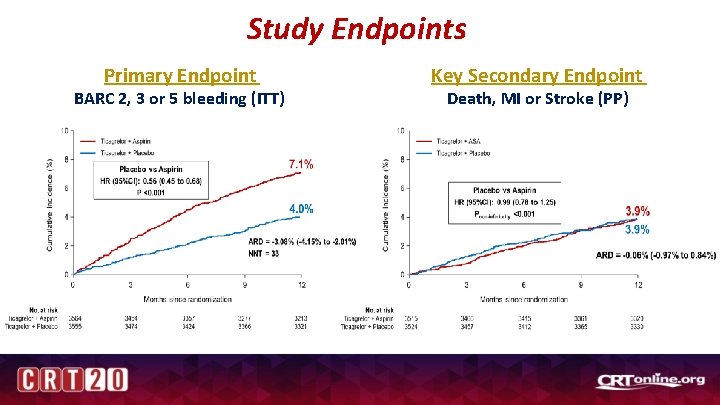
Study Endpoints Primary Endpoint BARC 2, 3 or 5 bleeding (ITT) Key Secondary Endpoint Death, MI or Stroke (PP)

Conclusions In high-risk patients who underwent PCI and were treated with ticagrelor and aspirin for 3 months without any major adverse (bleeding or ischemic) events, an antiplatelet strategy of continuing ticagrelor monotherapy resulted in: • substantially less bleeding than ticagrelor plus aspirin • without increasing ischemic events over a period of 1 year

TICO: The Next Ticagrelor Monotherapy Trial Results Next Month at ACC 2020! Kim et al. Am Heart J, 2019; 212, 45 -52. #AHA 19

The contemporary role of Aspirin Summary • Modest additional antiplatelet inhibition on top of potent P 2 Y 12 inhibitors • Major role in the occurrence of bleeding complications during DAPT and triple therapy • Improved safety of new generation DES may allow for less intensive antiplatelet regimen

…I’m sorry to say but it does seem like the TWILIGHT of Aspirin Therapy.

Thank you!
- Slides: 32