Which of the following best describes an Arrhenius

Which of the following best describes an Arrhenius base? a) b) c) d) e) proton donor proton acceptor produces H+ in water produces OH− in water electron donor © 2014 Pearson Education, Inc.

Which of the following best describes an Arrhenius base? a) b) c) d) e) proton donor proton acceptor produces H+ in water produces OH− in water electron donor © 2014 Pearson Education, Inc.
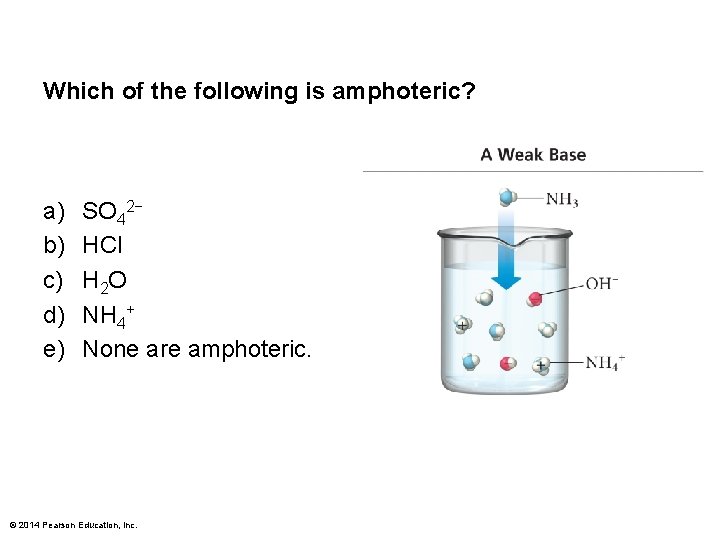
Which of the following is amphoteric? a) b) c) d) e) SO 42− HCl H 2 O NH 4+ None are amphoteric. © 2014 Pearson Education, Inc.
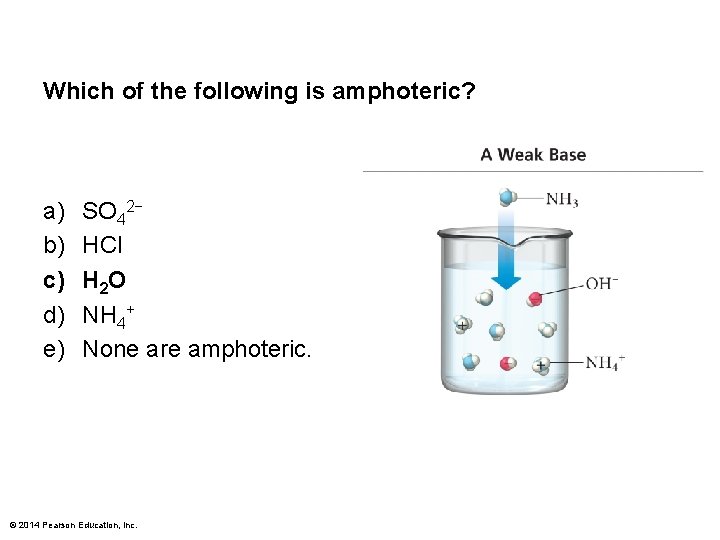
Which of the following is amphoteric? a) b) c) d) e) SO 42− HCl H 2 O NH 4+ None are amphoteric. © 2014 Pearson Education, Inc.

The p. H of a 0. 200 M weak acid is found to be 3. 5, what is the Ka of the acid? HA ⇌ H+ + A− a) b) c) d) e) 5. 00 × 10– 7 61. 3 1. 58 × 10– 4 1. 58 × 10– 15 1. 63 × 10– 2 © 2014 Pearson Education, Inc.

The p. H of a 0. 200 M weak acid is found to be 3. 5, what is the Ka of the acid? HA ⇌ H+ + A− a) b) c) d) e) 5. 00 × 10– 7 61. 3 1. 58 × 10– 4 1. 58 × 10– 15 1. 63 × 10– 2 © 2014 Pearson Education, Inc.
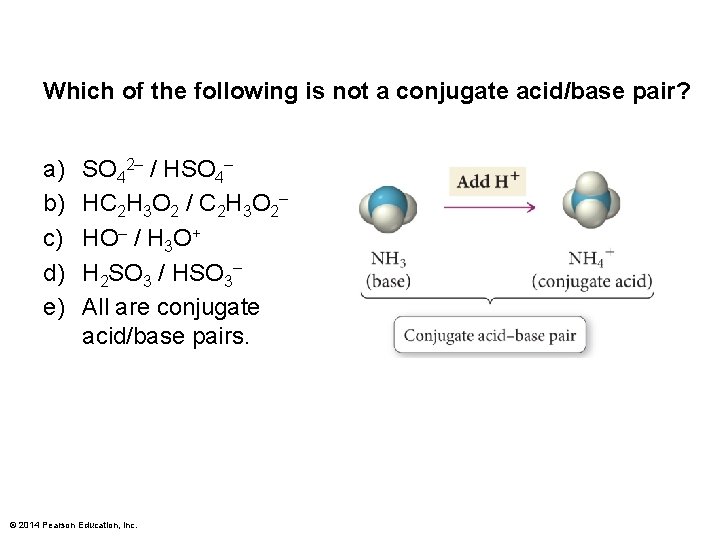
Which of the following is not a conjugate acid/base pair? a) b) c) d) e) SO 42– / HSO 4– HC 2 H 3 O 2 / C 2 H 3 O 2– HO– / H 3 O+ H 2 SO 3 / HSO 3– All are conjugate acid/base pairs. © 2014 Pearson Education, Inc.
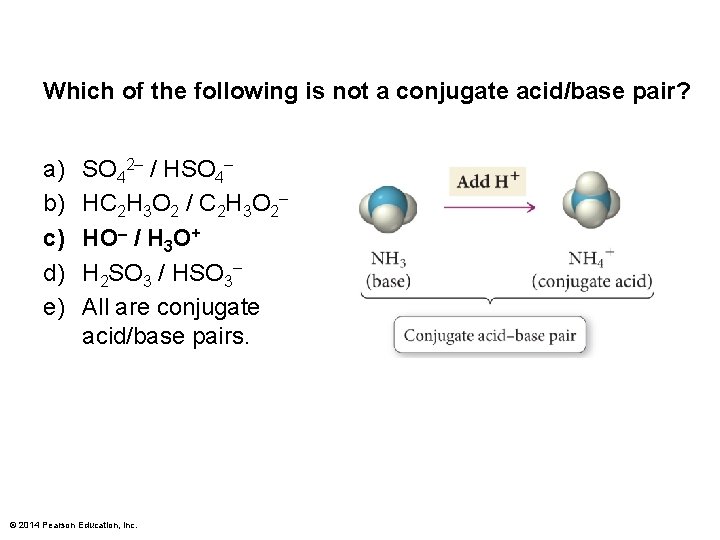
Which of the following is not a conjugate acid/base pair? a) b) c) d) e) SO 42– / HSO 4– HC 2 H 3 O 2 / C 2 H 3 O 2– HO– / H 3 O+ H 2 SO 3 / HSO 3– All are conjugate acid/base pairs. © 2014 Pearson Education, Inc.
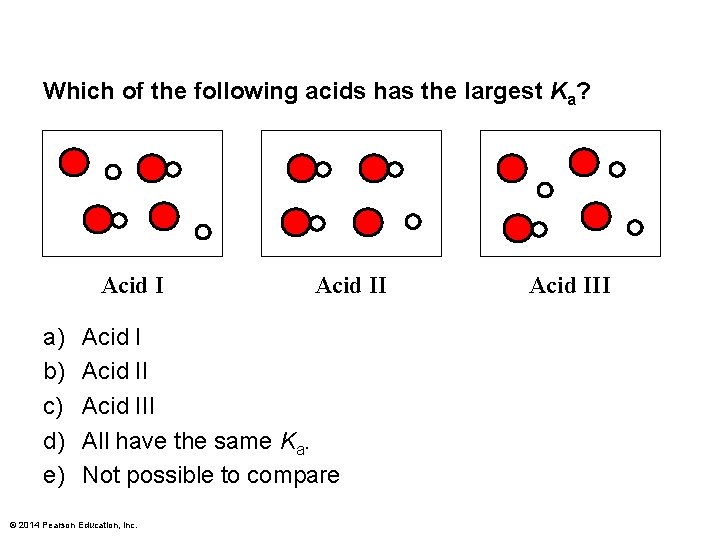
Which of the following acids has the largest Ka? Acid I a) b) c) d) e) Acid III All have the same Ka. Not possible to compare © 2014 Pearson Education, Inc. Acid III
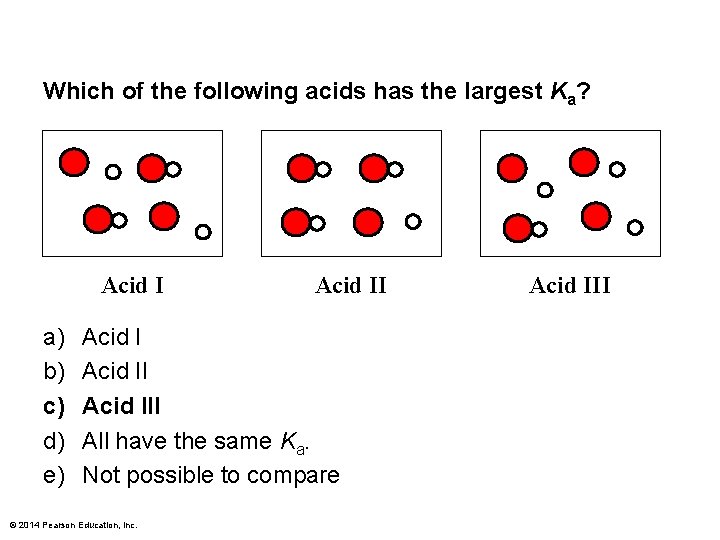
Which of the following acids has the largest Ka? Acid I a) b) c) d) e) Acid III All have the same Ka. Not possible to compare © 2014 Pearson Education, Inc. Acid III
Which of the following is a weak acid? a) b) c) d) e) H 2 SO 4 HCl HBr HF All are strong acids. © 2014 Pearson Education, Inc.
Which of the following is a weak acid? a) b) c) d) e) H 2 SO 4 HCl HBr HF All are strong acids. © 2014 Pearson Education, Inc.

What is the Kw of pure water at 50. 0°C if the p. H is 6. 630? H 2 O (l) + H 2 O(l) ⇌ H 3 O+(aq) + OH−(aq) a) b) c) d) e) 1. 0 × 10– 14 2. 34 × 10– 7 5. 50 × 10– 14 2. 13 × 10– 14 There is not enough information to solve. © 2014 Pearson Education, Inc.

What is the Kw of pure water at 50. 0°C if the p. H is 6. 630? H 2 O (l) + H 2 O(l) ⇌ H 3 O+(aq) + OH−(aq) a) b) c) d) e) 1. 0 × 10– 14 2. 34 × 10– 7 5. 50 × 10– 14 2. 13 × 10– 14 There is not enough information to solve. © 2014 Pearson Education, Inc.
Aspirin is a weak acid with a Ka = 3. 16 × 10– 4. What is the p. H of a solution that is 0. 25 M in aspirin? a) b) c) d) e) 0. 60 11. 94 3. 50 2. 05 2. 58 © 2014 Pearson Education, Inc.
Aspirin is a weak acid with a Ka = 3. 16 × 10– 4. What is the p. H of a solution that is 0. 25 M in aspirin? a) b) c) d) e) 0. 60 11. 94 3. 50 2. 05 2. 58 © 2014 Pearson Education, Inc.

What is the percent ionization of a 0. 250 M acetic acid solution? Ka = 1. 75 × 10– 5 a) b) c) d) e) 0. 209 % 0. 837 % 1. 67 % 7. 00 × 10– 3 % 123 × 10– 7 % © 2014 Pearson Education, Inc.

What is the percent ionization of a 0. 250 M acetic acid solution? Ka = 1. 75 × 10– 5 a) b) c) d) e) 0. 209 % 0. 837 % 1. 67 % 7. 00 × 10– 3 % 123 × 10– 7 % © 2014 Pearson Education, Inc.

What is the p. H of a 0. 116 M solution of Ba(OH)2? a) b) c) d) e) 8. 62 13. 06 13. 37 0. 63 12. 56 © 2014 Pearson Education, Inc.

What is the p. H of a 0. 116 M solution of Ba(OH)2? a) b) c) d) e) 8. 62 13. 06 13. 37 0. 63 12. 56 © 2014 Pearson Education, Inc.

Sodium benzoate is a common food preservative. What is the p. H of a 0. 25 M solution of sodium benzoate? p. Ka (benzoic acid) = 4. 19 a) b) c) d) e) 5. 21 8. 79 2. 40 11. 60 13. 40 © 2014 Pearson Education, Inc.

Sodium benzoate is a common food preservative. What is the p. H of a 0. 25 M solution of sodium benzoate? p. Ka (benzoic acid) = 4. 19 a) b) c) d) e) 5. 21 8. 79 2. 40 11. 60 13. 40 © 2014 Pearson Education, Inc.

Which of the following is a basic salt? a) b) c) d) e) Na. Cl NH 4 Cl KF a, b, and c are all acidic. a, b, and c are all basic. © 2014 Pearson Education, Inc.

Which of the following is a basic salt? a) b) c) d) e) Na. Cl NH 4 Cl KF a, b, and c are all acidic. a, b, and c are all basic. © 2014 Pearson Education, Inc.

Which of the following is true regarding the Ka values of a diprotic acid? a) b) c) d) Ka 1 < Ka 2 Ka 1 = Ka 2 Ka 1 > Ka 2 The relative values of Ka 1 and Ka 2 cannot be predicted. © 2014 Pearson Education, Inc.

Which of the following is true regarding the Ka values of a diprotic acid? a) b) c) d) Ka 1 < Ka 2 Ka 1 = Ka 2 Ka 1 > Ka 2 The relative values of Ka 1 and Ka 2 cannot be predicted. © 2014 Pearson Education, Inc.
A 0. 10 M solution of which of the following would have the lowest p. H? a) b) c) d) e) IO– IO 2– IO 3– All have the same p. H. Not possible to determine © 2014 Pearson Education, Inc.
A 0. 10 M solution of which of the following would have the lowest p. H? a) b) c) d) e) IO– IO 2– IO 3– All have the same p. H. Not possible to determine © 2014 Pearson Education, Inc.
- Slides: 28