Which block on the periodic table contains the


Which block on the periodic table contains the actinide series of elements?

Which is the most important characteristic in determining an element’s chemical properties? a. the number of protons and neutrons in its nucleus b. which period it is found in c. the number of valence electrons it contains d. its outermost energy level

Which region contains elements with two valence electrons?
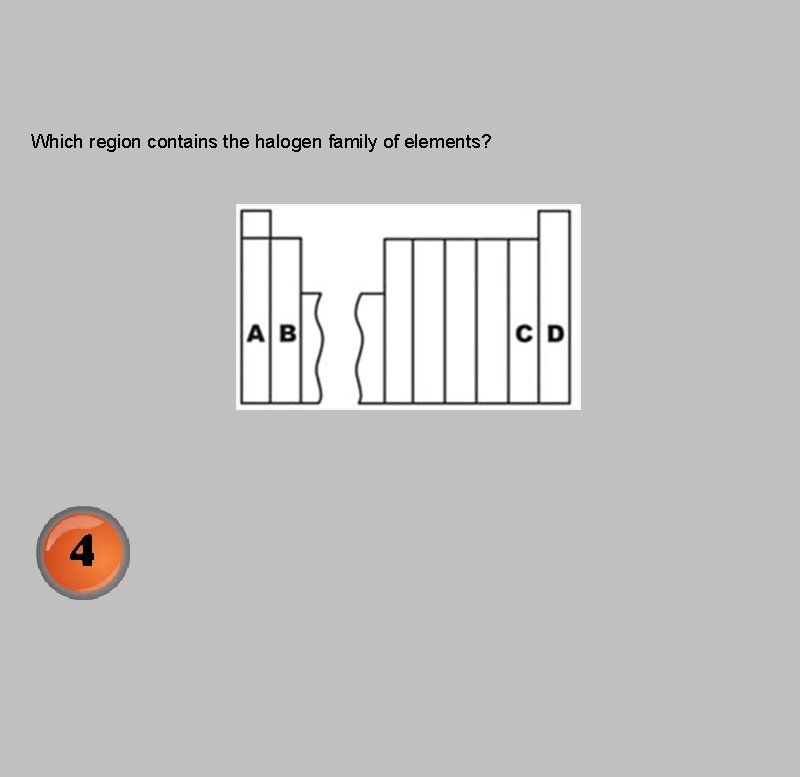
Which region contains the halogen family of elements?

Which of the following elements is a metal? a. boron b. nitrogen c. magnesium d. carbon

In which pair is the on the left larger than the on the right? a. Na, Rb b. S, Mg c. Br, Brd. K, K+

Which is the energy level that is being filled by electrons in the transition elements of period 5?

What is the energy level that is being filled by electrons in the transition elements of period 5?

Which is a transition element with five d-block electrons in energy level 4? a. niobium (Nb) b. rhenium (Re) c. manganese (Mn) d. technetium (Tc)
![Identify the period and group numbers of the element with this electron configuration. [Ne]3 Identify the period and group numbers of the element with this electron configuration. [Ne]3](http://slidetodoc.com/presentation_image_h/dfb07d748b87dac4bb486db830eefbe6/image-11.jpg)
Identify the period and group numbers of the element with this electron configuration. [Ne]3 s 23 p 4

Which element is the most metallic?

According to ____ periodic table, the physical and chemical properties of elements are periodic functions of their atomic weights. a. Dmitri Mendeleev’s b. John Newlands’ c. Henry Moseley’s d. Lothar Meyer’s

Which metalloid is in the fourth period and the same group as Carbon?

What is defined as the energy required to remove an electron from an atom of an element in the gaseous state?

List the metalloids.

What element has the highest electronegativity?

Identify the element having the largest size. 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 1 s 2 2 p 2

Write the electron dot structure of the element of group 6 A and the second period.

Identify the element having the highest electronegativity. 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 1 s 2 2 p 2

What are the names of the second halogen and the first noble gas?
- Slides: 21