Wheres Waldo Mrs Burts Physical Science Class MVCA

Where’s Waldo? ?

Mrs. Burt’s Physical Science Class MVCA Mission & Vision Unit 3: Chemistry Lesson 6: Chemical Equations Lesson 8: Lab: Dissolving Materials Lesson 9: Mixtures

MVCA Mission The mission of the Michigan Virtual Charter Academy is to provide an innovative, intensive academic preparation that inspires and educates students to achieve the highest levels of academic knowledge and skills. Michigan Virtual Charter Academy embraces a collaborative partnership between teachers and parents in order to empower students to reach extraordinary heights. Extraordinary results require extraordinary efforts! Through commitment, hard work, consistency, and responsibility, every student will meet the challenge of mastering high expectations

Lesson 6: Chemical Equations In this lesson you will learn how to balance chemical equations, showing that nothing is gained or lost in a chemical reaction. Explain "conservation of mass" in chemical reactions. Describe a chemical reaction using a chemical equation.

Writing an Equation Let’s take a look at a simple household item you see all the time and write the chemical equation that represents the reaction to create it: Aluminum Foil. The equation tells you the basic facts of the reaction. But as written, this reaction violates a basic law of nature. What is wrong?

The equation has Al. Cl 3 on the right side and KCl on the left side. Two atoms of Cl seem to be lost. Antoine Lavoisier (let’s just say a French guy ) discovered that the mass of the substances used in the reactions was the same after the reactions occurred as before. This called Law of Conservation of Mass. Total mass remains the same during a chemical reaction. We are now going to watch a BBC video explaining what this awesome French Guy was able to accomplish! http: //www. youtube. com/watch? v=x 9 i. Zq 3 Zxb. O 8

Balancing an Equation Let’s rewrite that equation now that we know why it was wrong! It needs to be balanced so both sides are equivalent (equal). In a balanced equation, the number of each type of atom on the reactant side equals the number if each type of atom on the product side.

Four steps to balancing an equation: 1) Write the chemical equation with the chemical symbols. 2)Count the number of atoms of each element on both sides of the equation.
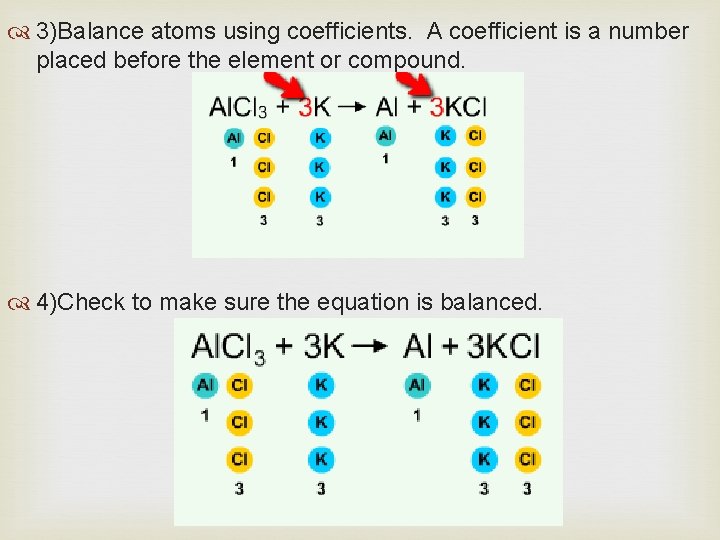
3)Balance atoms using coefficients. A coefficient is a number placed before the element or compound. 4)Check to make sure the equation is balanced.

Balancing an Explosive Reaction Let’s balance an explosive reaction. Sulfuric acid + sodium hydroxide reacts explosively to give us sodium sulfate. Let’s balance it! What’s the first thing we do?
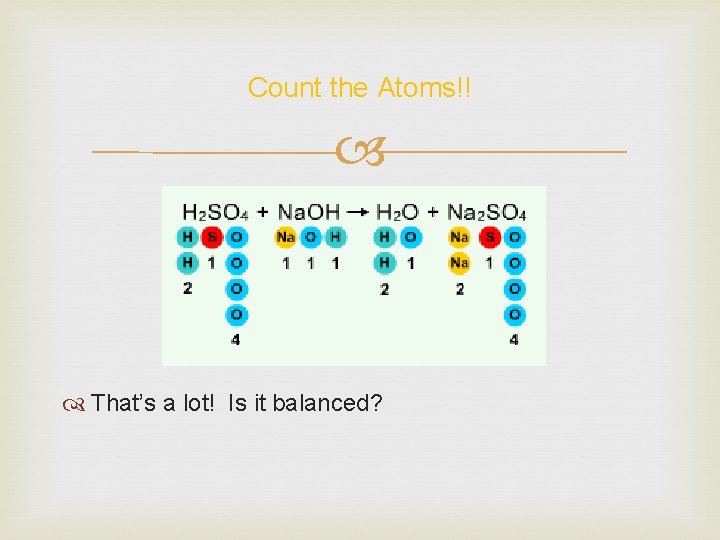
Count the Atoms!! That’s a lot! Is it balanced?

Add coefficients! Balancing equations sometimes takes some guessing. Begin where there is a shortage. Work your way from there.
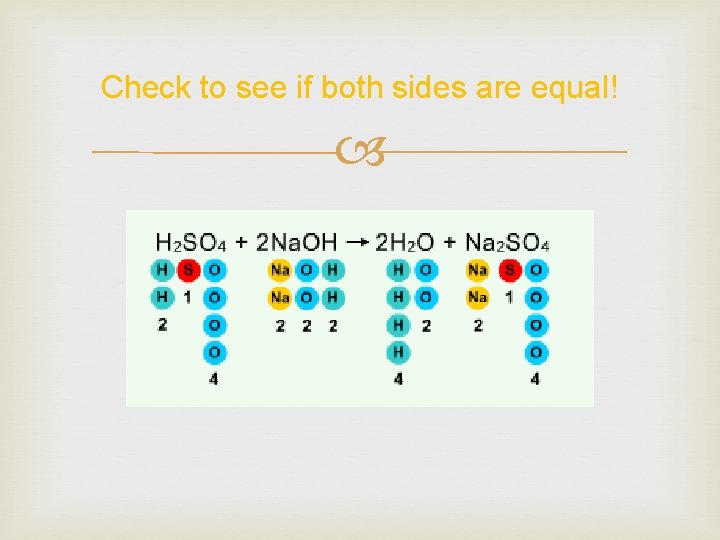
Check to see if both sides are equal!

Atomic Mass of Elements and Moles The atomic mass of an element can be found by using the periodic table. Here is what Nitrogen looks like on the periodic table. It has an atomic mass of 14. 01. 1 mole of Nitrogen is 14. 01 grams of Nitrogen.

Let’s look at a compound Ag. NO 3 Ag Atomic Mass= N Atomic Mass= 03 Atomic Mass= Atomic Mass of Ag. NO 3 is: 1 Mole is:

Lesson 9: Mixtures Explain that the properties of a substance or mixture depend on the properties, motions, and interactions of its molecules. Given a list of common substances and chemical formulas, classify matter as elements, compounds, or mixtures. Define homogeneous mixtures and heterogeneous mixtures, and recognize examples of each.

Mixtures A combination of materials that retain their initial identity!! When I say go I want you to type what you think an example of a mixture would be in the chat box. Fire Hydrant! Jing it!! Share!!

Student Ideas of a Mixture

Element, Compound, Mixture Oh MY! An element is a substance that cannot be broken down or separated into simpler substances. A compound is a substance made up of two or more elements that can only be separated by chemical means. Compounds and mixtures are made of elements. But often, the elements in a mixture are parts of compounds. Mixtures can be made of solids, liquids, or gases or combinations of them.

3 Things That Make a Mixture Different From a Compound: 1) The components of a mixture keep their unique properties and identities. The components are not in fixed ratios. The components of a mixture can be separated physically.


Mixture? Why?

Mixture? Why? Unlike the elements in a compound, the components of a mixture do not have fixed ratios. All ocean is salty. Water in some parts of an ocean has more salt than in other parts. There is no single ratio of salt to water in all the world’s oceans.

Heterogeneous Mixtures A heterogeneous mixture has components spread evenly throughout the mixture.

Homogeneous Mixture When all parts of a mixture have the same amount of each ingredient, the mixture is called homogeneous. A mixture with one or more of the substances dissolved in another is called a solution.
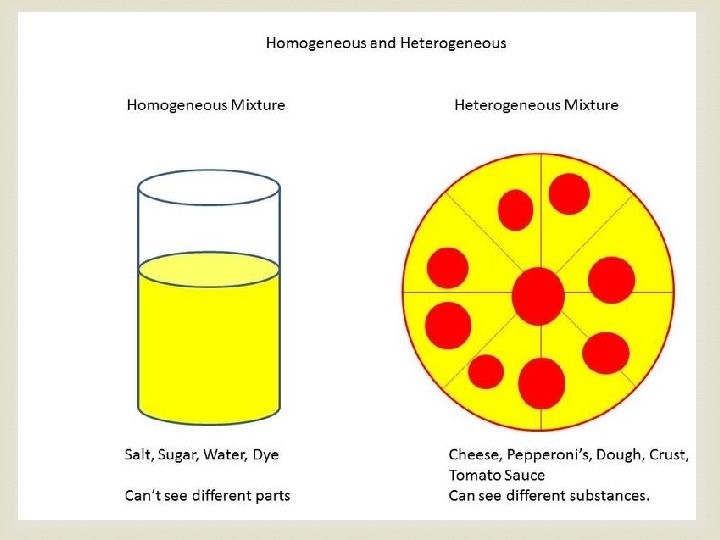

Let’s go back to our list of mixtures. Can we separate those into homogeneous and heterogeneous mixtures?

Any Questions? Remember to keep your progress percent in mind. The end of the quarter was last week. Are you on progress? On track for the end of the year? Office Hours Are Wednesday at 2 pm. Have A Fantabulous Week! You guys are the best!!
- Slides: 28