Where weve been Where were going Water cycle

Where we’ve been, Where we’re going • Water cycle • Rivers • Ground water • Hurricanes • El Nino/La Nina • Water Quality/Chemistry • Water’s physical properties • Ocean geography : ocean basins, plate tectonics • Ocean water : waves, tides & currents

The Oceans (the big blue)

Properties of sea water: 1. 2. 3. Salinity – why is it salty? What kind of salt is it? Density of sea water & it’s effect

Important vocabulary: • Salinity = Saltiness • Salinity = how much salt is in the water • Salt = another name for IONIC COMPOUND Na. Cl Mg. Cl 2 Cu. SO 4 - all “salts” What is the salinity of the oceans? ” on average it is ~33 ppt (parts per thousands) ~33 g/kg Or, - each kg of ocean water has 33 grams of salt in it.

Some vocabulary to know: Fresh water <0. 5 Brackish water 0. 5– 30 Salt water 30– 50 Brine >50

So, why is the ocean salty?

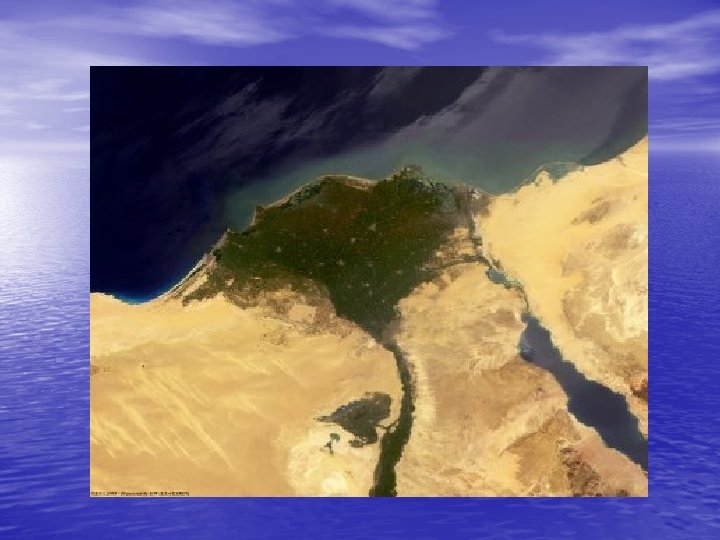


River water dissolves “salts” from the land carries them to the ocean Rivers are about 0. 1 ppt salinity • Bicarbonate HCO 3158. 4% • Calcium Ca 2+ 15% • Sulfate SO 4211. 2% • Chloride Cl 17. 8% • Sodium Na 1+ 6. 3%
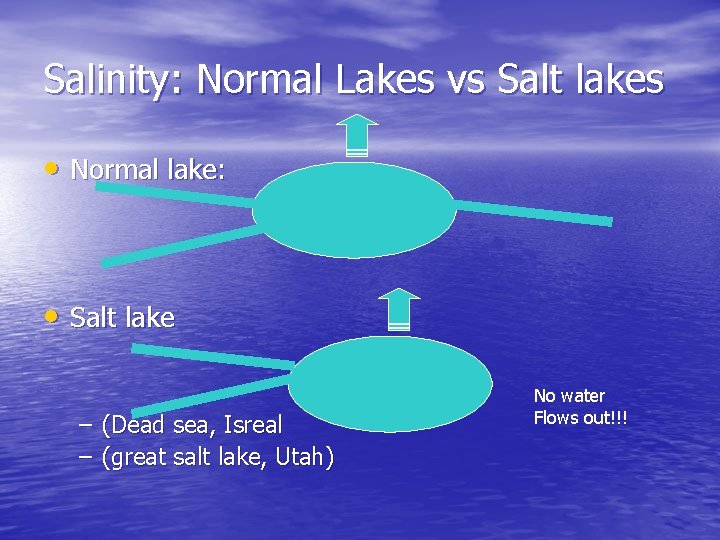
Salinity: Normal Lakes vs Salt lakes • Normal lake: • Salt lake – (Dead sea, Isreal – (great salt lake, Utah) No water Flows out!!!

Salinity: Why is the ocean salty? If you had a glass, and you put slightly muddy water in it, then let it all evaporate… then refilled it with more muddy water, and let it evaporate, …. What would happen? If you had another glass and you put tap water in it, (is tap water pure? ) and let it evaporate, then refilled, and let it evaporate, and you did this 10000 times…. What would happen?


So why is the ocean salty? • All the dissolved stuff from rivers accumulated over time. • But…. If you look at the ocean there has to be more to it!

What type of salt is it? Composition of sea salt compared to rivers – NOT just concentrated rivers salts • Rivers • 0. 1 ppt Bicarbonate Calcium Sulfate Chloride Sodium vs. 58. 4% 15% 11. 2% 7. 8% 6. 3% Ocean 32 ppt Chloride 55% Sodium 30. 6% Sulfate 7. 68% Magn. 3. 69% Calcium 1. 16% Potassium 1. 10%

So, why is the ocean’s salt different than just “concentrated river” salt • Things use the calcium & carbonate to make shells of calcium carbonate • Also, Many algae, (Diatoms) make their shells from Silica (glass/sand). • Short answer: Some of the dissolved stuff is useful, and so is taken out.

So, what must not be very usefull? • Rivers • 0. 1 ppt vs. • Bicarbonate • Calcium 58. 4% 15% • Sulfate • Chloride • Sodium 11. 2 7. 8% 6. 3% Ocean 32 ppt Chloride 55% Sodium 30. 6% Sulfate 7. 68% Magn. 3. 69% Calcium 1. 16% Potassium 1. 10%

Sea Water’s Density Which is denser Salt water or fresh water? Which is denser cold water or hot water? So what are the two things that can change water’s density?
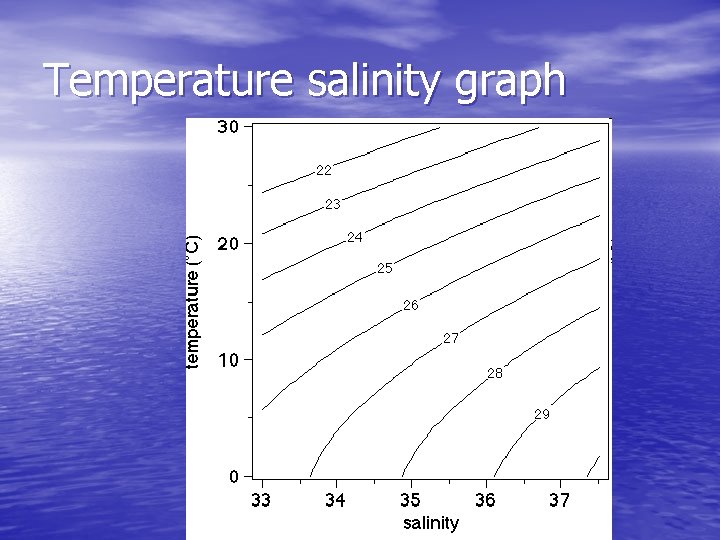
Temperature salinity graph
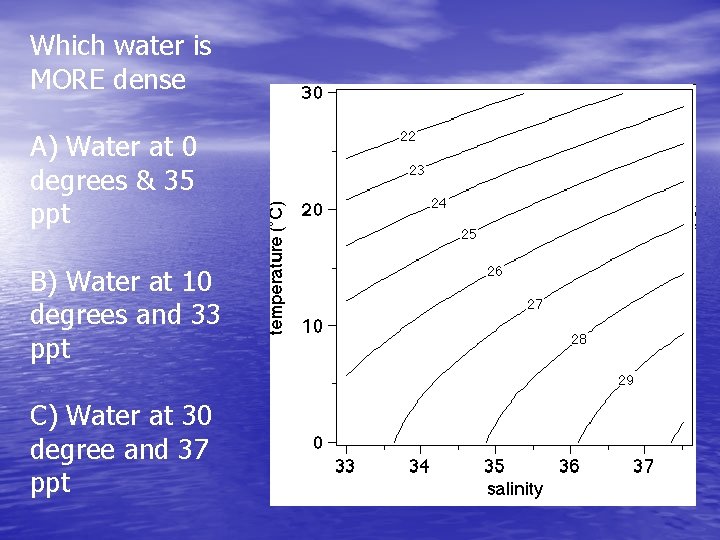
Which water is MORE dense A) Water at 0 degrees & 35 ppt B) Water at 10 degrees and 33 ppt C) Water at 30 degree and 37 ppt
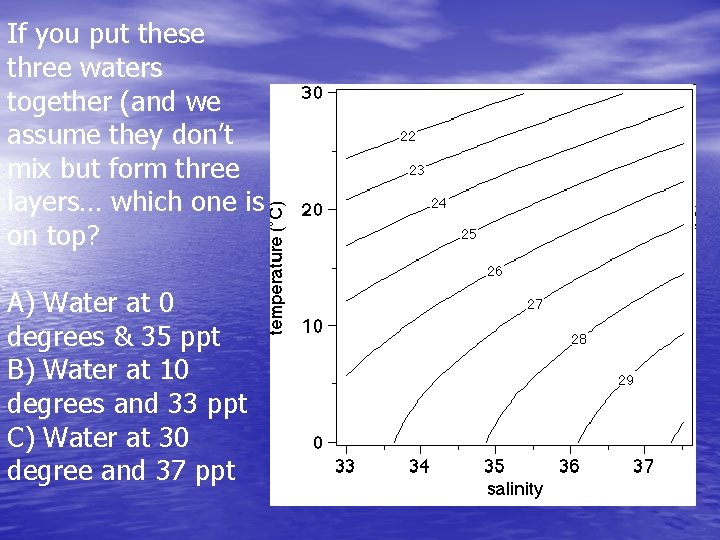
If you put these three waters together (and we assume they don’t mix but form three layers… which one is on top? A) Water at 0 degrees & 35 ppt B) Water at 10 degrees and 33 ppt C) Water at 30 degree and 37 ppt
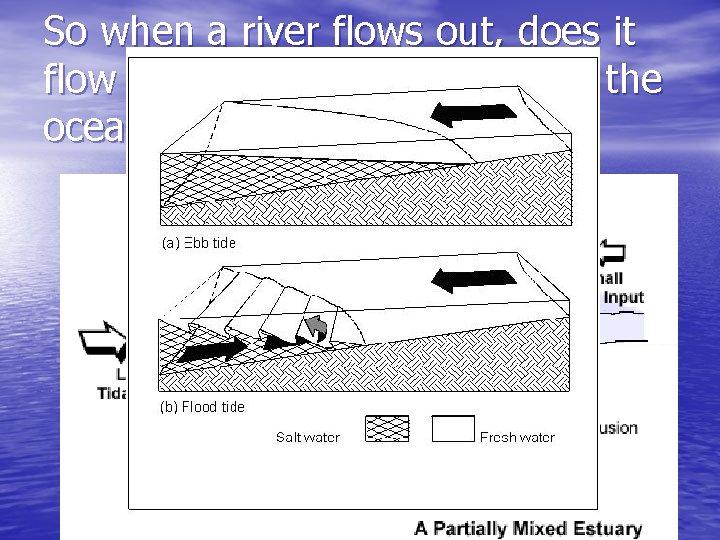
So when a river flows out, does it flow on top or to the bottom of the ocean?

2 nd place density takes a big effect 1 st – is estuaries (rivers meat ocean) fresh water vs salt water 2 nd – is the cold north and south poles salt water vs cold saltier water

Evaporating and freezing “purifies” water. What color is water and ice? What happens to salt and other stuff when water freezes? Does it stay or get pushed out? Anything dissolved gets pushed out, Salt, dissolved gas, anything Ice is clear, impurities make it cloudy
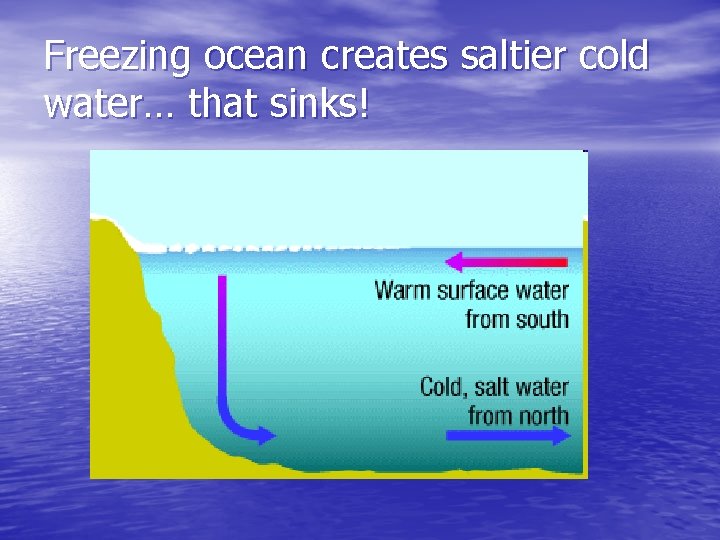
Freezing ocean creates saltier cold water… that sinks!
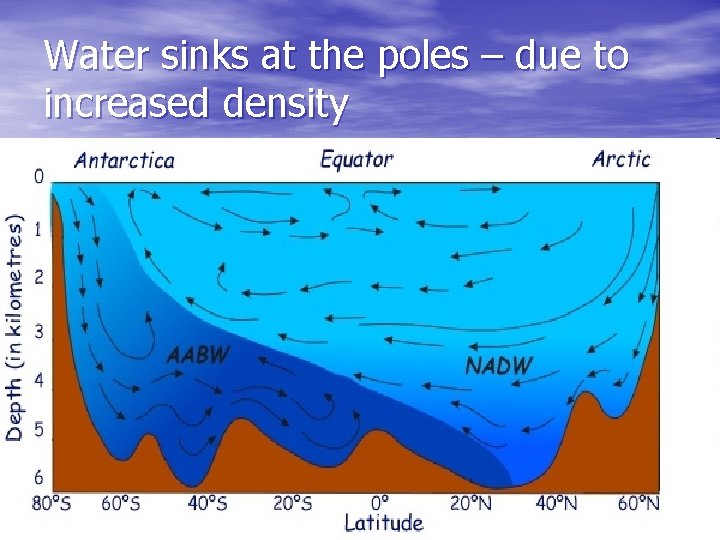
Water sinks at the poles – due to increased density

Summary 1. Oceans are salty due to river imput of 2. 3. 4. thousands of years of dissolved minerals Ocean salt is not the same as river salt because of biology (uses some of the salts) Density of sea water is controlled by salt and temperature Sea water tends to sink at the poles due to increased salinity and decrease in temperature.
- Slides: 28