When atoms lose or gain electrons they become


When atoms lose or gain electrons, they become ions. Cations are positive and are formed by elements on the left side of the periodic chart. Anions are negative and are formed by elements on the right side of the periodic chart.
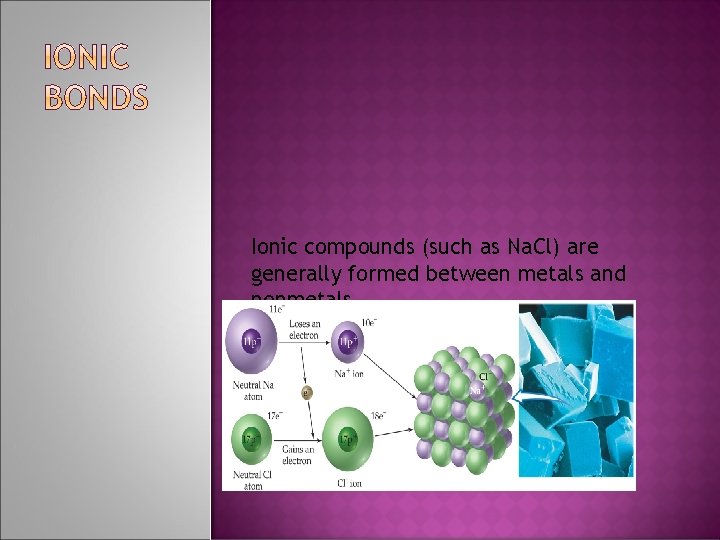
Ionic compounds (such as Na. Cl) are generally formed between metals and nonmetals.

The less electronegative atom is usually listed first. A prefix is used to denote the number of atoms of each element in the compound (mono- is not used on the first element listed, however. )

The ending on the more electronegative element is changed to -ide. CO 2: carbon dioxide CCl 4: carbon tetrachloride

If the prefix ends with a or o and the name of the element begins with a vowel, the two successive vowels are often elided into one: N 2 O 5: dinitrogen pentoxide

Oxidation numbers = charge of an atom With variable metals, the charge must be determined by anion knowing the compound’s net charge = 0 Ni. SO 4 _____ + -2 = 0 Nickel (II) sulfate Pb 3 N 4 3(___) + 4(-3) Lead (IV) nitride = 0

Depends upon anion with hydrogen Ionic names ending in –ide HF hydrogen fluoride hydrofluoric acid Ionic names ending in –ate H 3 PO 4 Hydrogen phosphate Phosphoric acid Ionic names ending in -ite HNO 2 Hydrogen nitrite Nitrous acid
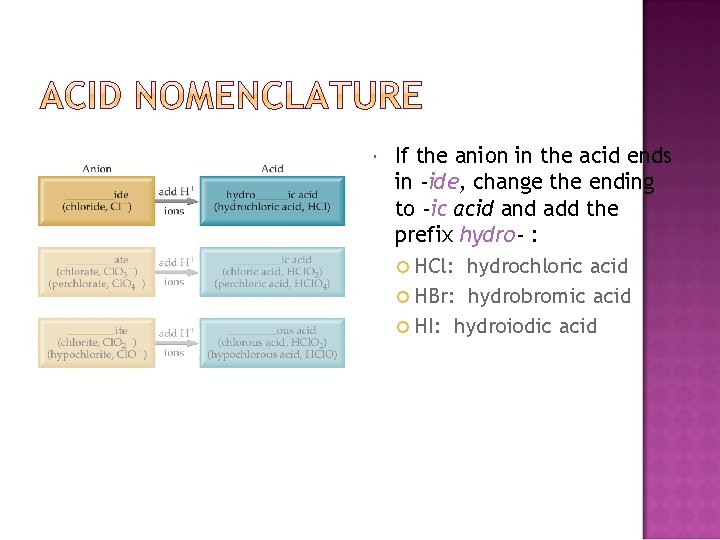
If the anion in the acid ends in -ide, change the ending to -ic acid and add the prefix hydro- : HCl: hydrochloric acid HBr: hydrobromic acid HI: hydroiodic acid
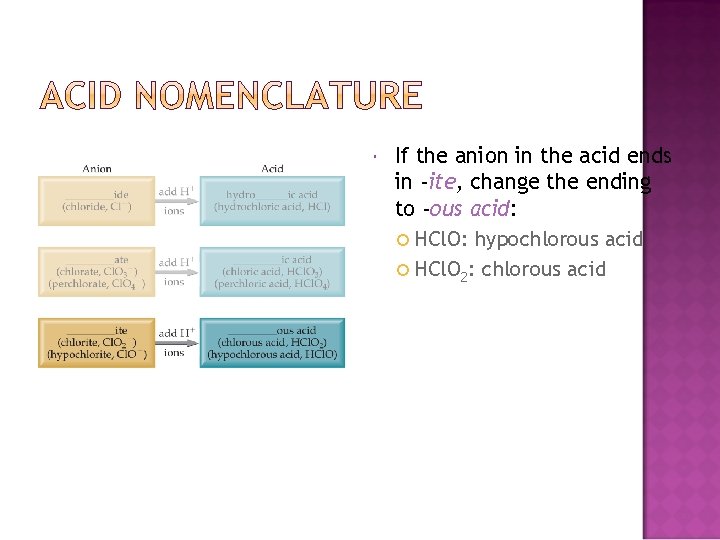
If the anion in the acid ends in -ite, change the ending to -ous acid: HCl. O: hypochlorous acid HCl. O 2: chlorous acid
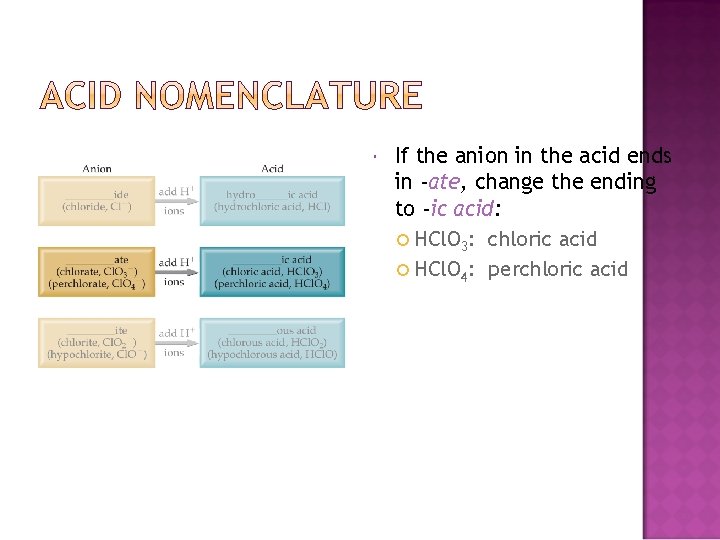
If the anion in the acid ends in -ate, change the ending to -ic acid: HCl. O 3: chloric acid HCl. O 4: perchloric acid

Know the prefixes or make it neutral

Because compounds are electrically neutral, one can determine the formula of a compound this way: The charge on the cation becomes the subscript on the anion. The charge on the anion becomes the subscript on the cation. If these subscripts are not in the lowest whole-number ratio, divide them by the greatest common factor.
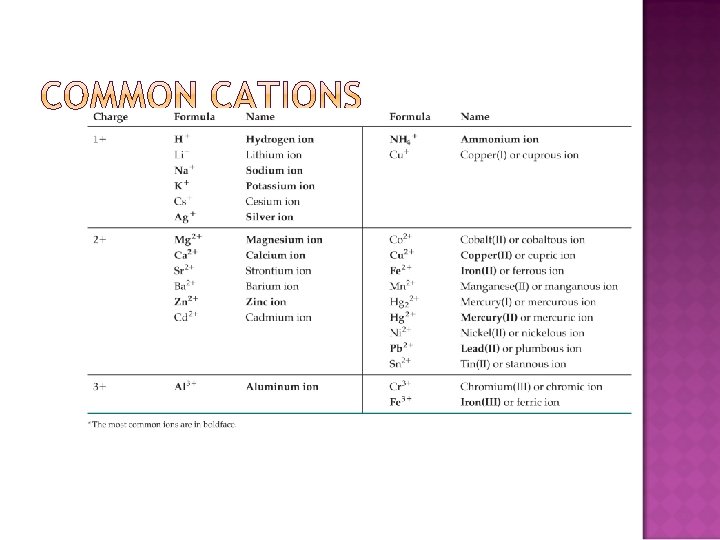
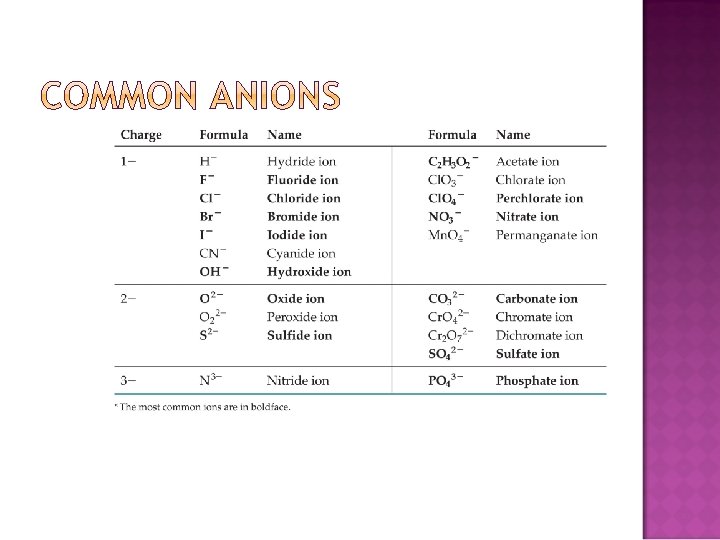

When there are two oxyanions involving the same element: The one with fewer oxygens ends in -ite NO 2− : nitrite; SO 32− : sulfite The one with more oxygens ends in -ate NO 3− : nitrate; SO 42− : sulfate
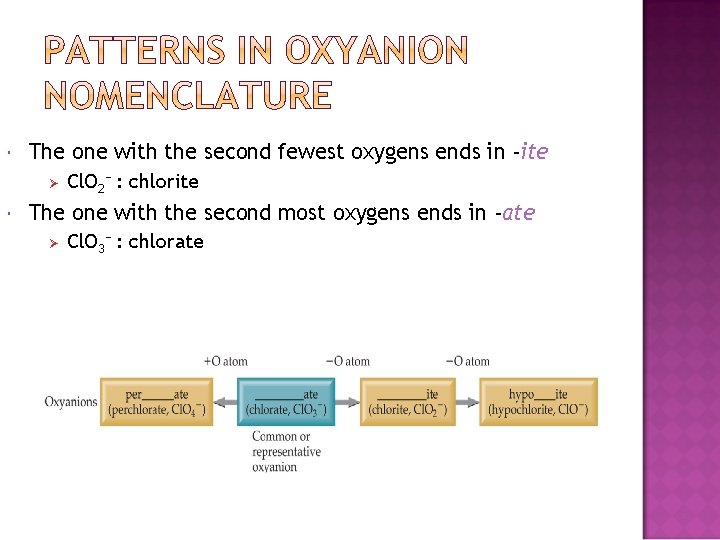
The one with the second fewest oxygens ends in -ite Ø Cl. O 2− : chlorite The one with the second most oxygens ends in -ate Ø Cl. O 3− : chlorate
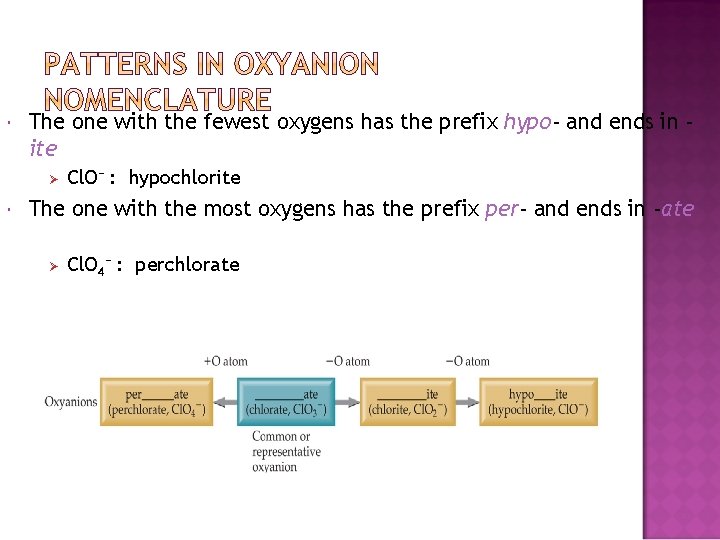
The one with the fewest oxygens has the prefix hypo- and ends in ite Ø Cl. O− : hypochlorite The one with the most oxygens has the prefix per- and ends in -ate Ø Cl. O 4− : perchlorate

Ionic-must know formulas and charges Overall compound is neutral Toughest task is knowing polyatomic ions ionic Covalent-if you know your prefixes, it’s easy Many large covalent molecules have common names such as sugar C 12 H 22 O 11 covalent
- Slides: 19