WHATS THE MATTER WITH MATTER Thinking about Matter

WHAT’S THE MATTER WITH MATTER?

Thinking about Matter Sort the following into two categories, matter and not matter. ○ Peanut butter, water, fish, garbage, time, motion, the human brain, carbon dioxide gas, yourself, an idea, tree, energy. How did you distinguish between the categories?

What is Matter? Matter is anything that has mass and volume

What forms does matter come in? Solid Liquid Gas Plasma

States of Matter Solid has definite shape and definite volume. The particles of matter in a solid are very tightly packed Solid - Ice

States of Matter Liquid has undefined shape but defined volume The particles in a liquid are not rigidly held in place and are less closely packed than are the particles in a solid. Liquid - Water

States of Matter Gas has undefined shape and undefined volume. Compared to solids and liquids, the particles of gases are very far apart. gases are easily compressed. Gas - Steam

States Of Matter Plasma very hot gasses undefined shape and volume only exist at high temperatures (i. e. the sun)

Physical Properties Characteristics of a substance that can be observed without altering the identity of the substance. State, density, color, odor, melting point, boiling point, luster, conductivity, brittleness, malleability.

Chemical Properties Characteristics of a substance that cannot be observed without altering the identity of the substance. Reactivity, flammability, reaction types ○ Rusting is an example of a chemical property of iron • The inability of a substance to change into another substance • Resistance of iron to undergo change in the presence of nitrogen.

Properties of Matter Video http: //www. youtube. com/watch? v=Ypwtjp 0 FSE

Practice Draw pictures that represents a solid, a liquid, and a gas. Explain each drawing.

Physical and Chemical Changes Physical Changes Do not alter the identity of a substance ○ Crushing, tearing, changes of state (solid to liquid to gas) Chemical Changes Alter the identity or chemistry of a substance ○ Burning, cooking, rusting Physical Versus Chemical Changes Video http: //www. youtube. com/watch? v=qqqm. FFCwd 7 k &feature=related

Physical and Chemical Properties Consider a copper coin. Make a list of its chemical and physical properties. Consider these photos for clues.
Practice Physical vs. Chemical
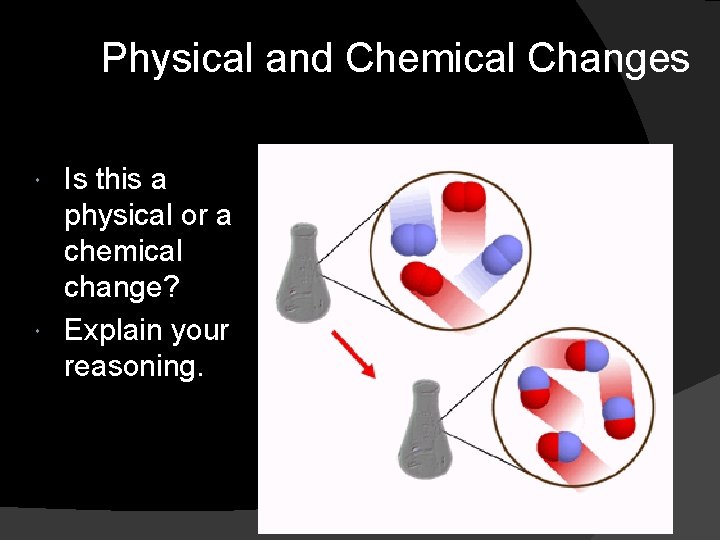
Physical and Chemical Changes Is this a physical or a chemical change? Explain your reasoning.
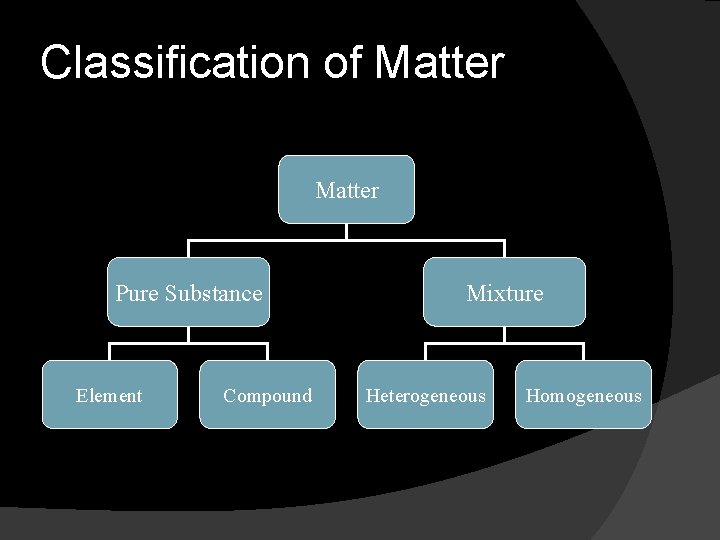
Classification of Matter Pure Substance Element Compound Mixture Heterogeneous Homogeneous
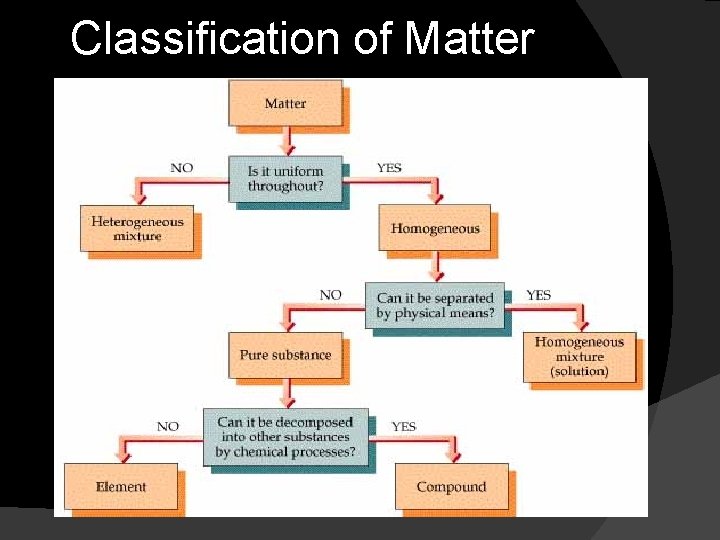
Classification of Matter

Pure Substance Elements Matter that can not be broken down into simpler substances under normal lab conditions Contains only one kind of atom Atom Molecule Elements (symbols) Na, Au, C Where can you find a list of all the elements?
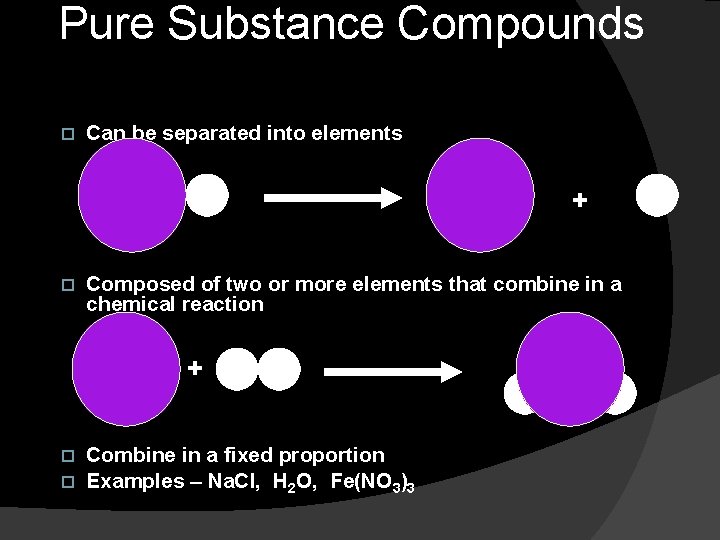
Pure Substance Compounds Can be separated into elements + Composed of two or more elements that combine in a chemical reaction + Combine in a fixed proportion Examples – Na. Cl, H 2 O, Fe(NO 3)3
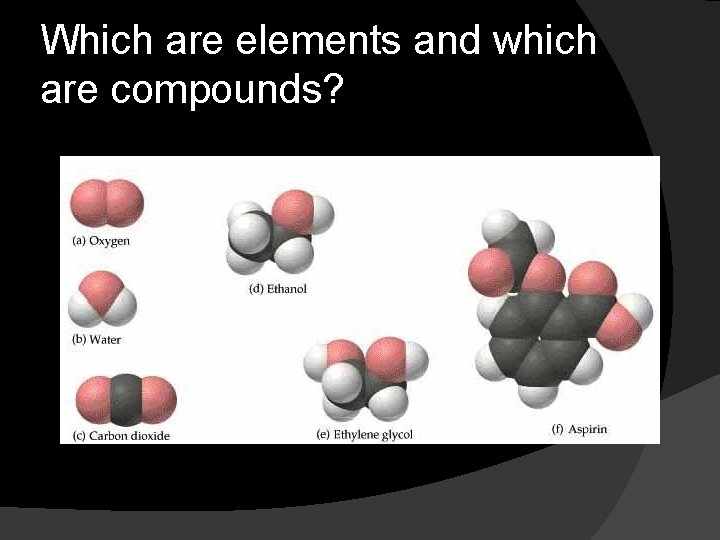
Which are elements and which are compounds?

Mixtures A blend of two or more pure substances Not chemically combined Composition of mixtures is variable Granite

Mixtures Heterogeneous Mixture with visibly different parts. A heterogeneous mixture is one that does not blend smoothly throughout and in which the individual substances remain distinct. Sand + water Salt + Pepper M & M’s

Mixtures Homogeneous Mixture with no visibly different parts. Sea water H 2 O + Na. Cl Air N 2 + O 2 + CO 2

Practice Classifying Matter Make a list of 3 things (and their parts) found in the classroom. Classify these as solid, liquid, or gas pure substances and mixtures homogeneous or heterogeneous elements or compounds Be prepared to share your classifications?

Classify mixtures pure substances elements compounds Draw a picture that represents a pure compound.

Classify each of the following as, a homogenous or heterogeneous mixture, pure substance, element or compound, atom or molecule. 1. CH 4 2. KMn. O 4 3. Co 4. Al + H 2 O Draw atomic level pictures of each substance.

Review Questions Identify each of the following as an example of a homogeneous mixture or a heterogeneous mixture. A. a pile of rusty iron filings B. 70% isopropyl rubbing alcohol C. Saltwater D. Gasoline

Review Questions Identify each of the following as an example of an element or a compound. A. sucrose (table sugar) B. the helium in a balloon C. baking soda D. a diamond

Closure Draw an atomic level picture of a mixture of compounds.

SEPARATION OF MIXTURES

Separation of Mixtures No Chemical Change Heterogeneous Filtration Seperate by size or state of matter

Separation of Mixtures Homogeneous Distillation Using Different Boiling points Crystallization Evaporation or cooling with a solid left behind Chromatography Solution flows along a stationary substance

Filtration §Filtration is a technique that uses a porous barrier to separate a solid from a liquid.
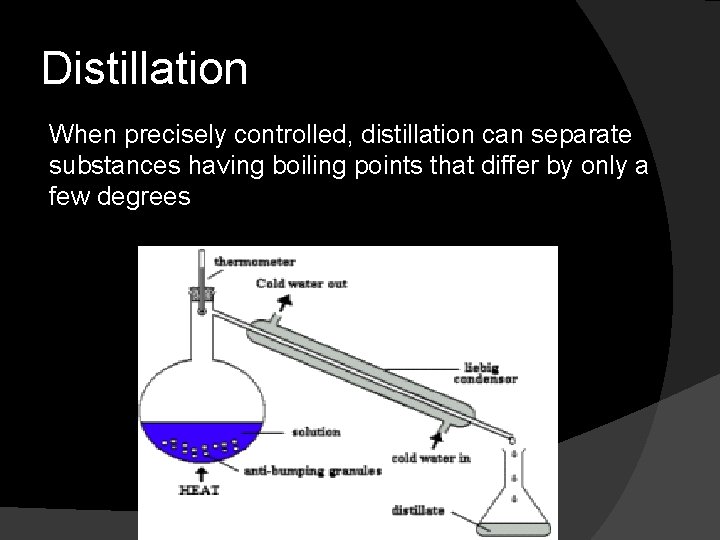
Distillation When precisely controlled, distillation can separate substances having boiling points that differ by only a few degrees

Evaporation/Crystallization • Crystallization is a separation technique that results in the formation of pure solid particles of a substance from a solution containing the dissolved substance.

Chromatography

Chromatography • The separation occurs because the various components of the ink spread through the paper at different rates.

Practice Propose a way to separate this mixture: Styrofoam balls, sand, salt and Iron fillings.
- Slides: 39