Whats Important About the Nutrition Label What Why

• What’s Important About the Nutrition Label? • What • Why does the label tell you? is this important?

• Unit 3: The Chemistry of Life • Unit Objective: • To identify the carbon-based organic molecules of life and understand the roles they play in life processes. • Lesson Objective: • To know how an atom is constructed and how each element’s properties determines how the formation of compounds (bonding) takes place. Only copy down underlined material. Everything else is there for support.

• First Question… • Why do we study CHEMISTRY in BIOLOGY class… • https: //youtu. be/fg. QLyq. Wa. Cb. A • So. Why do we? . . .

• What Are Organisms Made Of? • Every organism is different depending upon it’s role or needs but there are basic elements that are universal to life on Earth.

• The Basic Compounds of Life. • Regardless of the amount, most organisms use elements to form these Macromolecules of Life. 1. 2. 3. 4. • These are the fundamental molecules of life that you will be required to know. • • Fats Proteins Sugars Nucleic Acids. These carbon-based molecules are known as Organic Compounds. Another molecule you will learn about in this mini-unit is water.

• Essential Questions • • How does hydrogen, carbon, and oxygen combine to form molecules that participate in living systems? What do these molecules do to support life? • Objectives • • • To review/identify what makes up matter. To know the elements that form most organic molecules. To review/understand why atoms form bonds. To understand how and why these bonds are essential for life by forming the carbon-based organic molecules of life. This information will help you soon be able to explain how these molecules participate in important processes in living things.

• Vocabulary • Atoms • Element • Valence electrons • Compound • Molecule • Ion

• Before You Learn About the Organic Macromolecules… • You must understand that all bigger molecules are built of smaller pieces. Everything of atoms… • & Atoms arranged into basic molecules. • • These basic molecules are called… • These will sometimes be called… (a. k. a. ) SUBUNITS BUILDING BLOCKS or MONOMERS • What • is a subunit? Something that is combined with other subunits to build something bigger; a macromolecule or polymer.
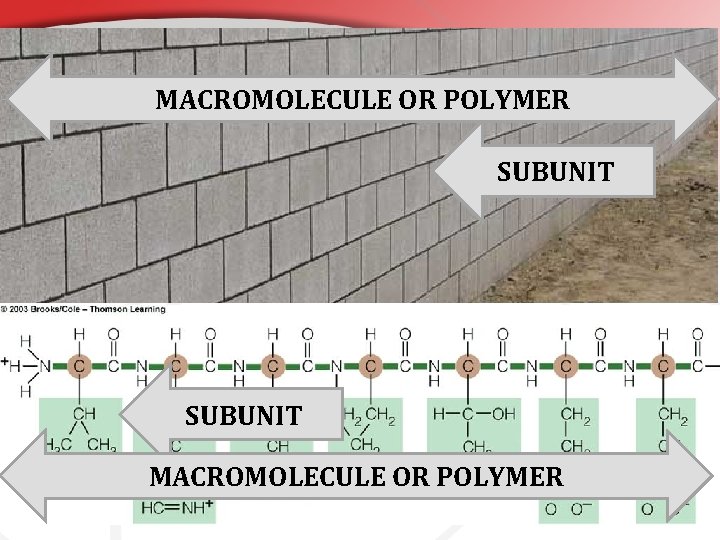
MACROMOLECULE OR POLYMER SUBUNIT MACROMOLECULE OR POLYMER

• I. What’s an Atom? • Every living and nonliving thing is made of matter. • Matter is anything that has mass and takes up space. All matter is made of very small particles called atoms. • An atom is the smallest unit of matter that cannot be broken down by chemical means. • • This means that there are smaller parts, but these smaller parts are not true matter.

• Atoms • The atom is composed of three main types of smaller “subatomic” particles, each with unique properties. • Protons: positively charged particles. • Neutrons: particles with no charge. • Electrons: negatively charged particles.
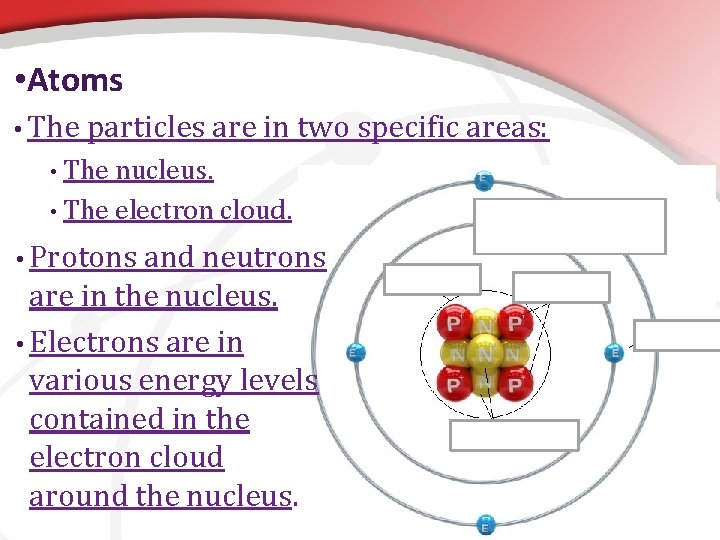
• Atoms • The particles are in two specific areas: The nucleus. • The electron cloud. • • Protons and neutrons are in the nucleus. • Electrons are in various energy levels contained in the electron cloud around the nucleus. Energy Levels of the Electron Cloud
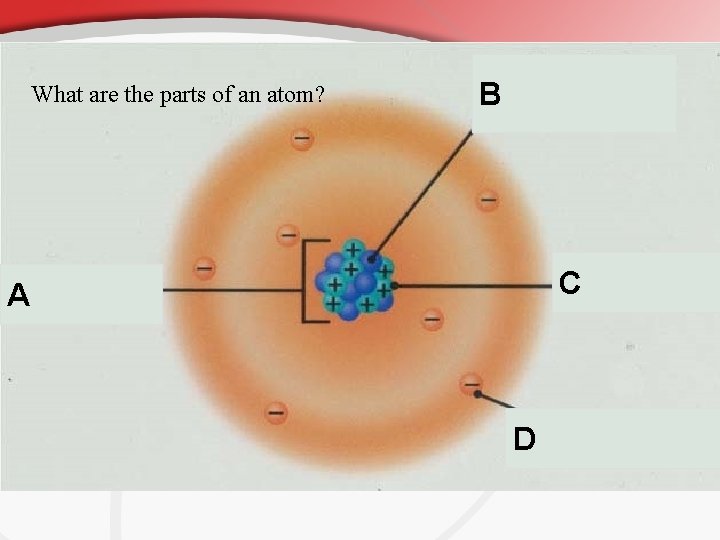
What are the parts of an atom? B C A D


• II. Getting Orientated With Atoms & Elements Find carbon on the periodic table. • This is the information for the element CARBON • Notice its row and column. • • Every box represents a different element. III. An element is any quantity of a substance that is 100% the same type of atom. • Being the same type means they have the same # of protons. • • Ex: If I asked you for a sample of the element carbon you could give me a diamond because every atom in it has 6 protons.

• CHNOPS: What are the Most Abundant Elements in Biology? • • III. a. The six highlighted below are the most abundant elements in the bodies of most biological organisms. Take out the periodic tables & Mark these in your Periodic Table.
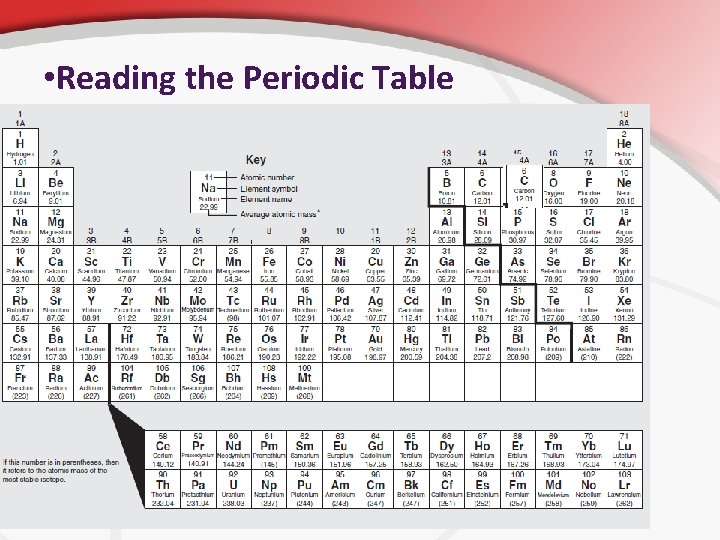
• Reading the Periodic Table • Atomic Number • Symbol • Name • Atomic Mass

• Reading a Periodic Table: Atomic Numbers, Mass, Electrons… • The boxes give a lot of information. • The boxes on the periodic table give basic properties of the elements to help determine things like: • Atomic # • # electrons • # neutrons = # protons = atomic mass - # protons.

• Ions. What Happens When an Atom Gains or loses an Electron. • Usually, atoms by themselves if stable will have equal numbers of protons & electrons. • • • Stable means the +/- charges cancel and they are neutral. Electrons are moving at high speeds and can migrate from atom to atom. Sometimes electrons can jump. If an atom gains or loses electrons they become ions. V. Ions are atoms that have a charge because the protonelectron numbers don’t match. • How are they formed? = by electrons ‘jumping’ from one atom to another… causing a different number of positive and negative charges in the same atom. • • This causes a charge to form on the atom that gives it different reactivity (much like a magnet)
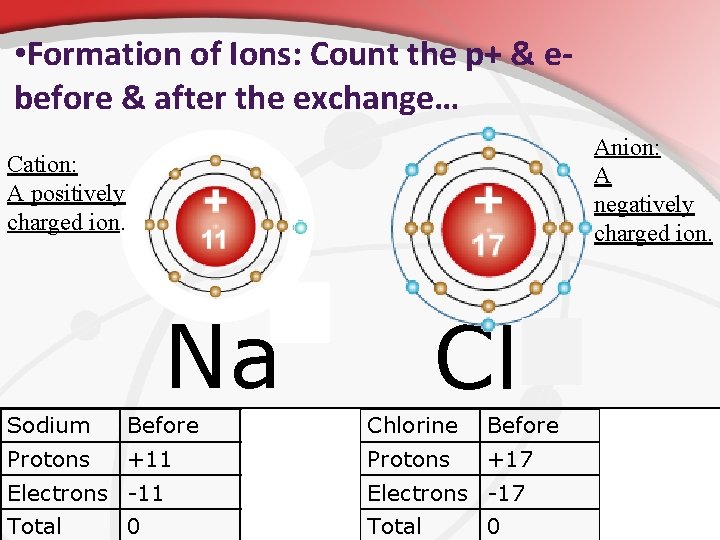
• Formation of Ions: Count the p+ & ebefore & after the exchange… Anion: A negatively charged ion. Cation: A positively charged ion. + Na Cl Sodium Before After Chlorine Before After Protons +11 Protons +17 Electrons -11 -10 Electrons -17 -18 Total +1 Total -1 0 0

• Special Circumstance: Isotopes Elements • Recall, elements are all the same type of atom because every atom has the same number of protons. • • • For example, every atom for any amount of the element carbon has six protons. There are several types of carbon though. In general, atoms of an element can have different numbers of neutrons sometimes. • VI. Isotopes are atoms of elements that have a different number of neutrons. • Isotopes create challenges and benefits because bonds form differently and are usually radioactive to a degree.
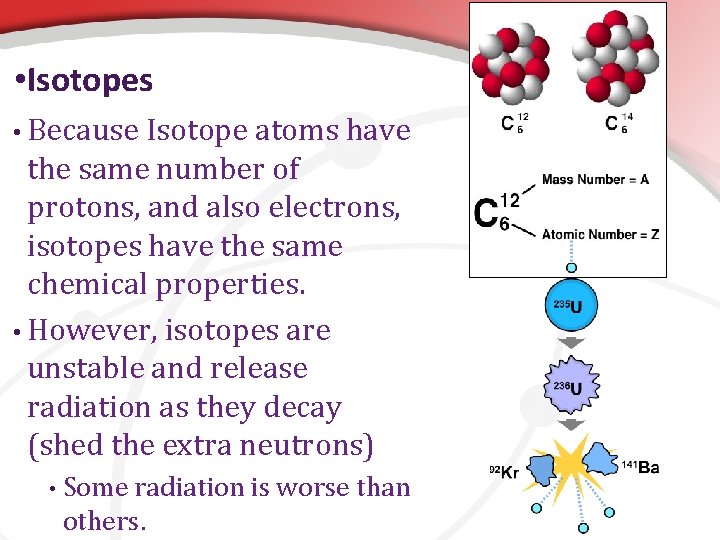
• Isotopes • Because Isotope atoms have the same number of protons, and also electrons, isotopes have the same chemical properties. • However, isotopes are unstable and release radiation as they decay (shed the extra neutrons) • Some radiation is worse than others.

• The Numbers Really Matter • The numbers represent an atom’s proton, neutron, and electron number when it is unbounded and electrically neutral. • Atoms are rarely like this, stable, in nature. • Mainly because the electrons are always flying around. electron

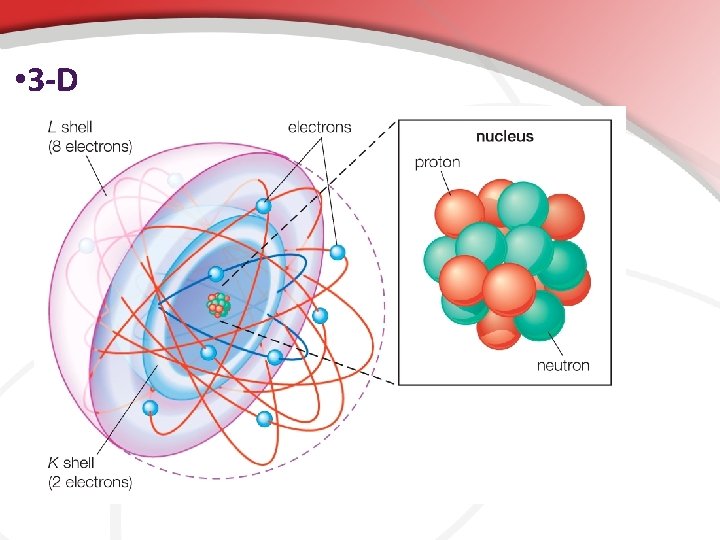
• III. Reading a Periodic table to Determine the Construction and Electron Configuration in an Atoms are usually represented as a nucleus surrounded by rings. (called the Solar System Model) • The rings are energy levels. • The rings exist because e- can’t occupy random or the same space at the same time. • There are rules to how the e- are distributed. • The row (going from side to side) tells you how many rings the atom has. • • • Hydrogen is in the 1 st row = one ring Nitrogen is in the 2 nd row = two rings • Shown • • to the right. How many rings does iodine have? These are the atom’s energy levels.
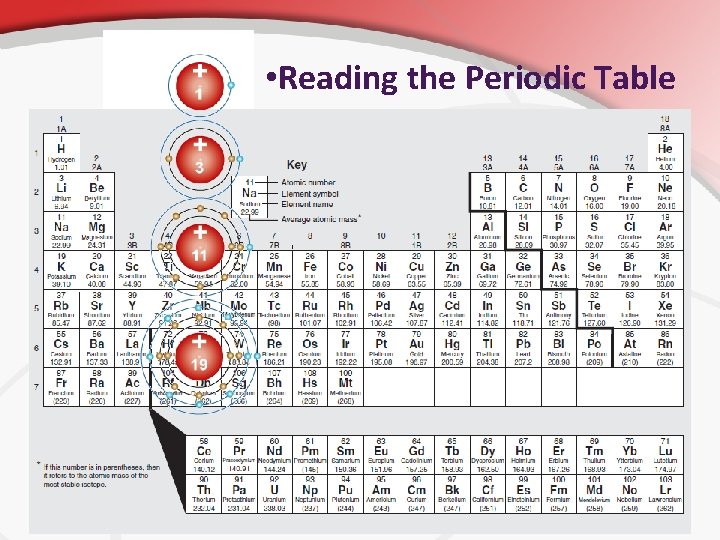
• Reading the Periodic Table • The Column tells you how many electrons in the outer shell (valence electrons). • The Row tells you how many rings (Energy levels)

• The Outer Ring… The Valence Shell Remember, electrons are orbiting the nucleus in the region called the electron cloud. . . In different energy levels (orbitals). • The outer ring is called the valence shell; it is the important one. • • This shows you the valence shell electrons. • Abbreviated ve • In the valence shell bonding happens.

• The Outer Ring The outer most edges of this cloud is called the valence shell. • There is a strict rule for how many electrons are in this valence shell. • For most groups of atoms, you can determine the # of valence shell electrons from the column it’s in. • • The column determines the valence shell electrons. Group 1 A (hydrogen, etc. ) has one valence electron. Group 2 A (beryllium, etc. ) has two. This rule only applies to A groups though. How many valence electrons does phosphorus have? A: 5

Take 3 minutes and copy When done, take 2 minutes and determine which element is which • Copy Into the Supplemental Handout + Electrons orbit in levels. Each level has a max number of e- it can hold. 1 st ring = 2 2 nd ring = 8 3 rd ring = 18 4 th ring = 32 - NOT A PRIORITY. YOU HAVE THIS ALRE

• Part I. Which Atom is Which? • What are the 2 clues?

• Concept Check. Part II. • On your handouts Look for “Part II. Atomic Models”, complete the blank atom for the element carbon. • Draw the: • Correct # of electrons • Correct # of electron in the valence shell • Correct # of protons • Correct # of neutrons • Put them in the correct places.
• Representing the Atom • Lots of Atom Models • Which one’s right? • Depends on the application

• How Do You represent an Atom? • Bohr Model: depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in particular circular orbits around the nucleus.
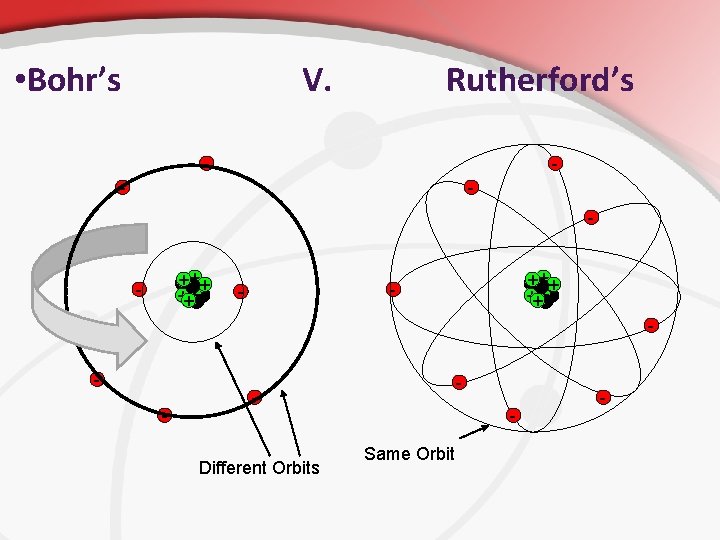
• Bohr’s V. Rutherford’s - - - +++ +++ - - - Different Orbits Same Orbit -
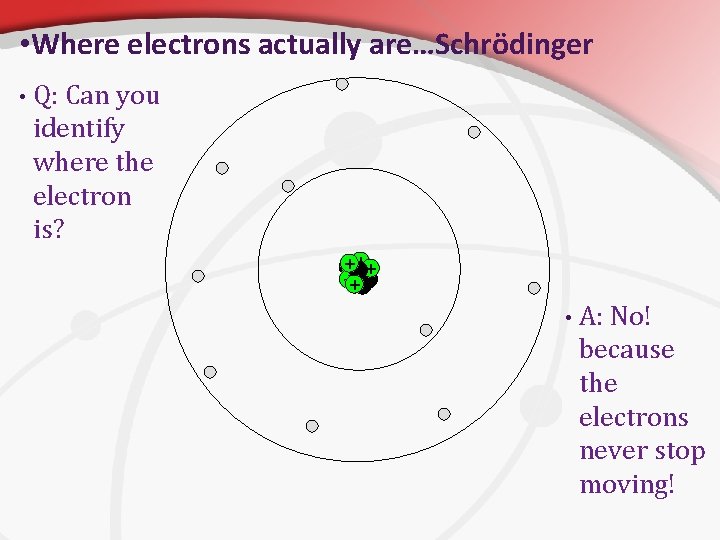
• Where electrons actually are…Schrödinger • Q: Can you identify where the electron is? +++ • A: No! because the electrons never stop moving!
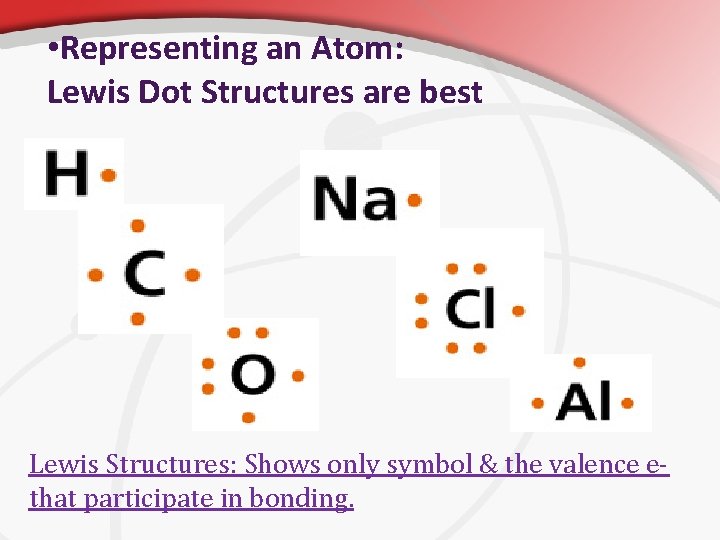
• Representing an Atom: Lewis Dot Structures are best Lewis Structures: Shows only symbol & the valence ethat participate in bonding.
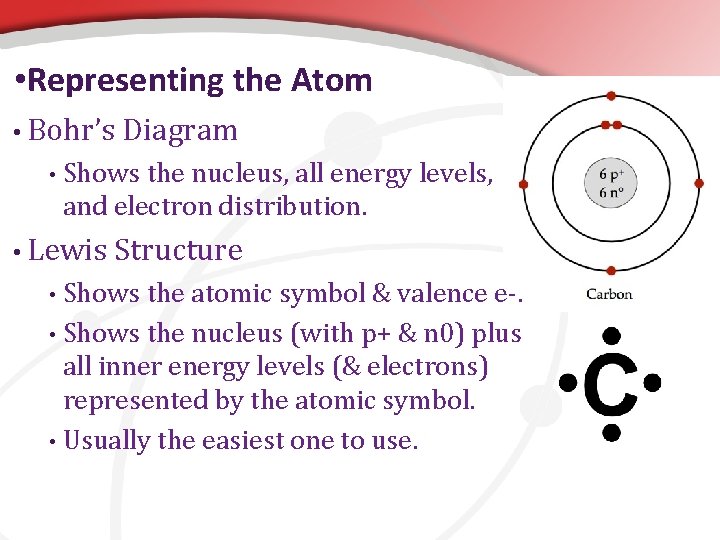
• Representing the Atom • Bohr’s • Diagram Shows the nucleus, all energy levels, and electron distribution. • Lewis Structure Shows the atomic symbol & valence e-. • Shows the nucleus (with p+ & n 0) plus all inner energy levels (& electrons) represented by the atomic symbol. • Usually the easiest one to use. •

• Complete Part II. Practice. Create the Bohr & Lewis Structure for… 5 minutes. • You did the Bohr model for Carbon. • Now complete the Lewis Structure for Carbon. • Also Complete: Use only the number of rings that you need. • Magnesium • Chlorine

• Magnesium

• Chlorine

• Part 2. • Learn the types of chemical bonds. • Practice showing bonding/ predicting how atoms will combine to form larger molecules, compounds, and others. • Become acquainted with the rules that cause atoms to bond as well as determine the result of the new structures. Molecules • Compounds • Ions • Polarity •

• Chemical Bonds Atoms by themselves can have balanced charges (p+/e -), but are often unstable so join, or bond with other atoms to achieve stability. • Chemical bonds: the force that holds atoms together. • Remember Valence Shells… Electrons in the valence shell are called valence electrons. • • The number 8 is special. • • This determines what atoms will bond with what. *OCTET RULE* (*Generally speaking) Atoms tend to combine with each other such that eight electrons will end up in the valence shell. • The reason is stability! • *The exceptions are Hydrogen (can only have 2) and Helium (doesn’t bond)

• Types • There are three types of bonds that you need to know. • • • Ionic bonds = When metal and non-metal atoms SWAP electrons creating ions of opposite charges that are attracted to each other. Covalent bonds = When any two or more atoms SHARE electrons to create molecules. Hydrogen bonds = When separate polar molecules interact with one another. • There is an order of strength… • Covalent > Ionic > Hydrogen. • (covalent is the strongest, hydrogen the weakest)
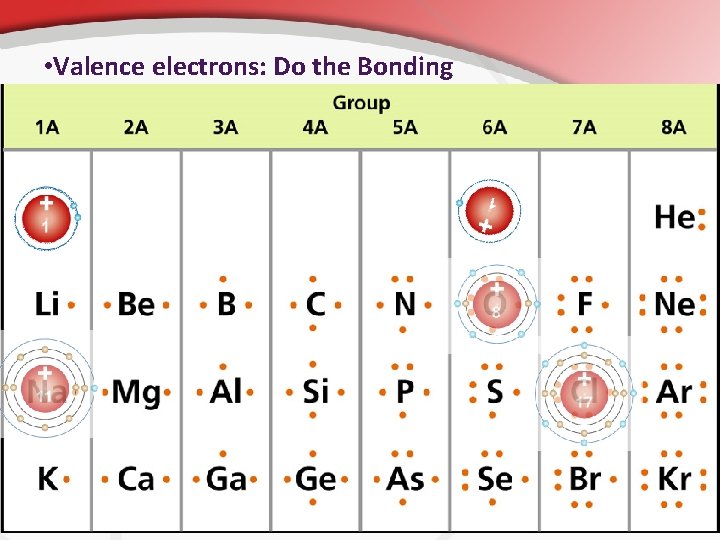
• Valence electrons: Do the Bonding

• Special Circumstance: Noble Gasses • Some atoms don’t generally bond with other elements because they already have 8 valence electrons. • These are called Nobel Gasses. • These are group 8 A (all the way on the right).

• What is Formed When 2 DIFFERENT Elements Bond? • Every other element will bond with other elements to get to 8 valence electrons. • Chemical bonds form between groups of atoms because atoms become stable when they have eight electrons in the valence shell. • When atoms of different elements combine, a compound forms. • A compound is a substance made of the bonded atoms of two or more elements.
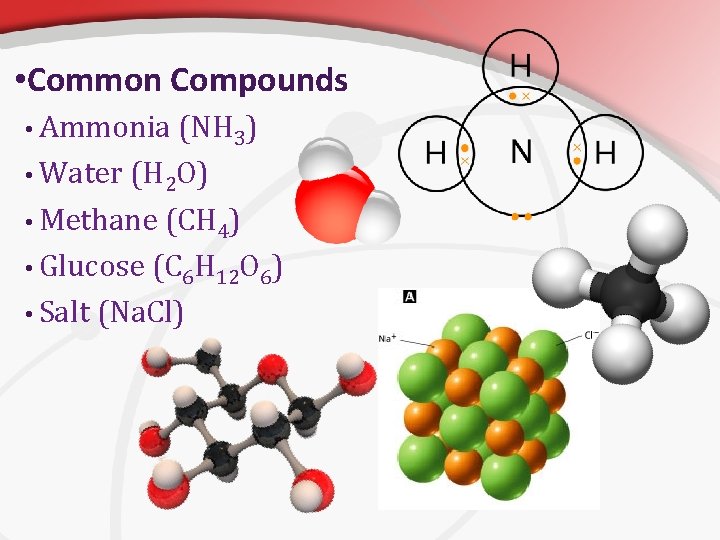
• Common Compounds • Ammonia (NH 3) • Water (H 2 O) • Methane (CH 4) • Glucose (C 6 H 12 O 6) • Salt (Na. Cl)

• Ionic Compounds Ionic Bonding • Atoms can sometimes achieve a stable valence shell (have 8) by losing or gaining electrons. • When this happens, the charge of the atom changes slightly and an ion is formed. • Recall: [An ion is an atom or group of atoms that has an electric charge because it has gained or lost electrons. ] Opposite charges attract. • The attractive force between oppositely charged ions is an ionic bond. •

• How Do You Know How Many Electrons Get Swapped? • It is all based upon the number of valence electrons for the element in its un-bonded form and how close to 8 they exist naturally. • The metals on the left usually give electrons because they are closer to 8 if they lose a few. • The non-metals on the right usually gain electrons because they will achieve 8 if they gain just a few.
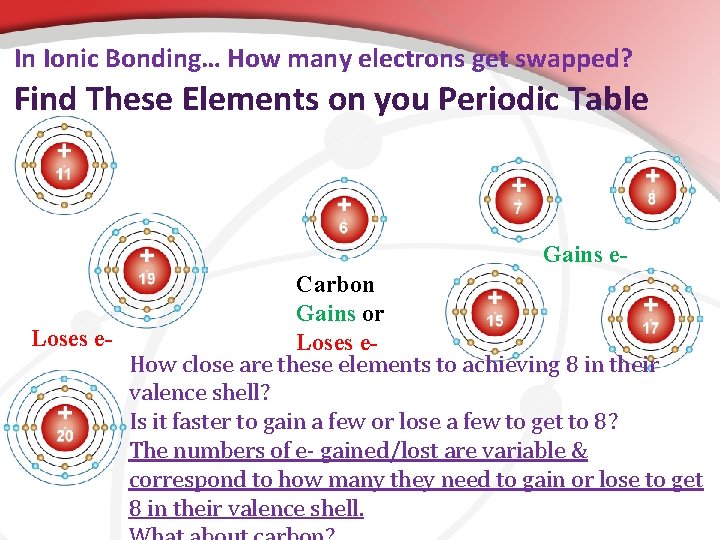
In Ionic Bonding… How many electrons get swapped? Find These Elements on you Periodic Table Gains e. Carbon Gains or Loses e. How close are these elements to achieving 8 in their valence shell? Is it faster to gain a few or lose a few to get to 8? The numbers of e- gained/lost are variable & correspond to how many they need to gain or lose to get 8 in their valence shell.
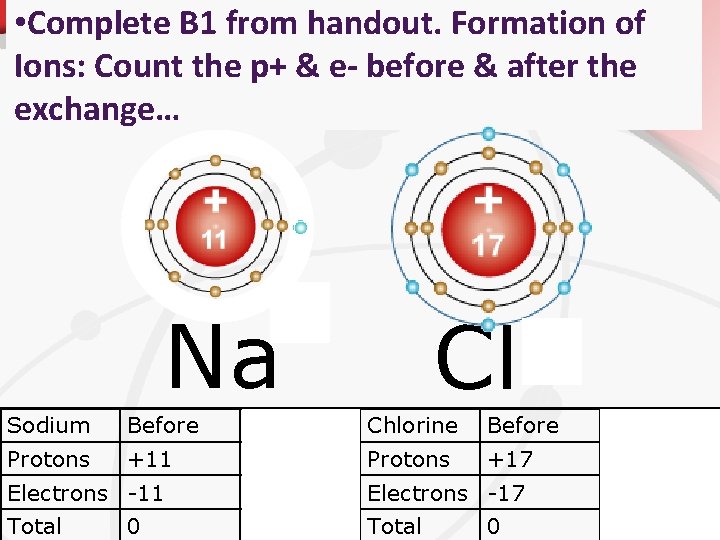
• Complete B 1 from handout. Formation of Ions: Count the p+ & e- before & after the exchange… + Na Cl Sodium Before After Chlorine Before After Protons +11 Protons +17 Electrons -11 -10 Electrons -17 -18 Total +1 Total -1 0 0
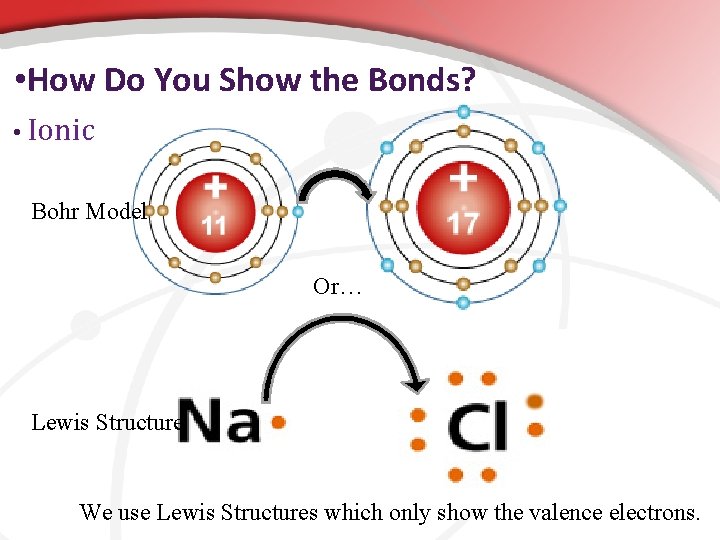
• How Do You Show the Bonds? • Ionic Bohr Model Or… Lewis Structure We use Lewis Structures which only show the valence electrons.

• Show the bonding of Na with Chlorine • Figure out how many electrons sodium loses. • How many does chlorine need? • Find these and fill these out on your sheet. -1 for sodiums • +1 for chlorine • • Draw the arrow showing the electron moving. Lewis Structure
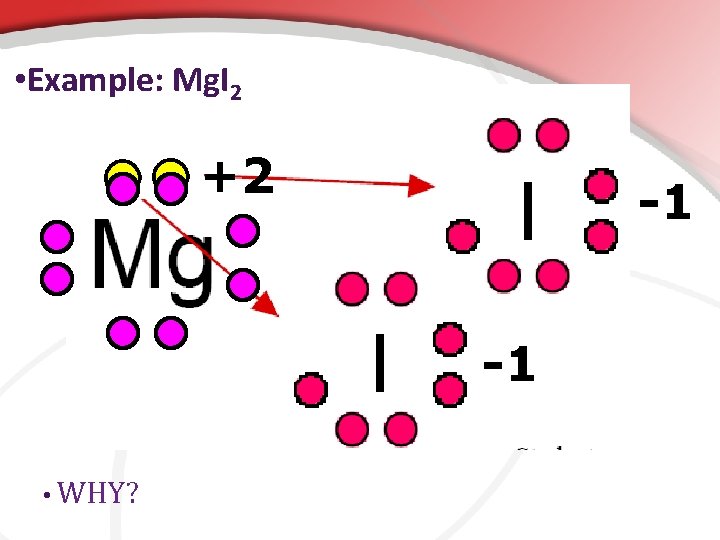
• Example: Mg. I 2 +2 I I • WHY? -1 -1
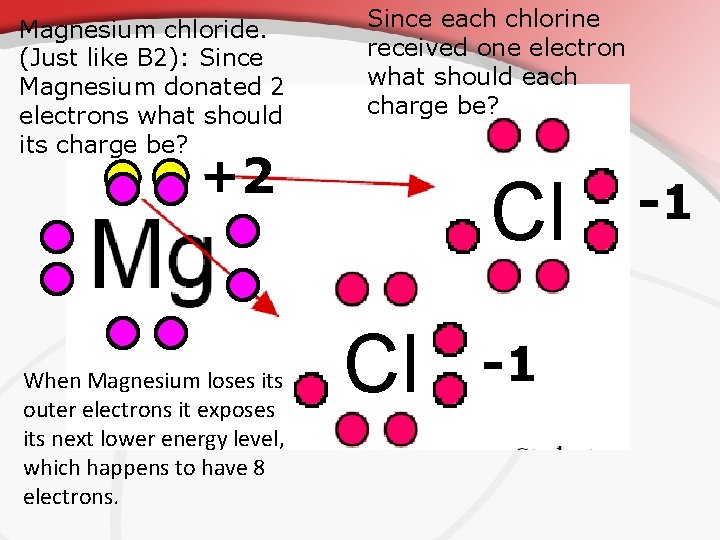
Magnesium chloride. (Just like B 2): Since Magnesium donated 2 electrons what should its charge be? Since each chlorine received one electron what should each charge be? +2 When Magnesium loses its outer electrons it exposes its next lower energy level, which happens to have 8 electrons. Cl Cl -1 -1

• Chemical Bonds, continued Covalent Bonding • Another way that atoms bond is by sharing valence electrons to form a strong covalent bond. • Covalent bonds form molecules: • A molecule is a group of atoms (the same or different) held together by covalent bonds. Remember, the goal is to get 8 valence electrons… • A water molecule, H 2 O, forms when an oxygen atom forms covalent bonds with two hydrogen atoms. •

• Poor Oxygen… • • • Ah, I’m sad because my valence shell isn’t filled. If there was only some way to get more. Wait. Hey Buddy. If we shared some electrons, then we could both have 8…kinda. Now we both can have eight! Sometimes… and that’s enough to form a covalent bond.

• Polarity In some covalent molecules, the electrons are shared equally between the atoms in the molecule. • In some covalent bonds, the shared electrons are attracted more strongly to one atom than to the other. • • • It’s due to electronegativity (something you don’t need to know yet). As a result, one end of the molecule has a partial negative charge, while the opposite end has a partial positive charge. Polar Molecules , like H 2 O, share electrons unequally. • They result when the resulting molecule has partial charges on opposite ends because of electron motion and average location. • In non-polar molecules, like CO 2, electrons are distributed symmetrically along all molecule axes. •
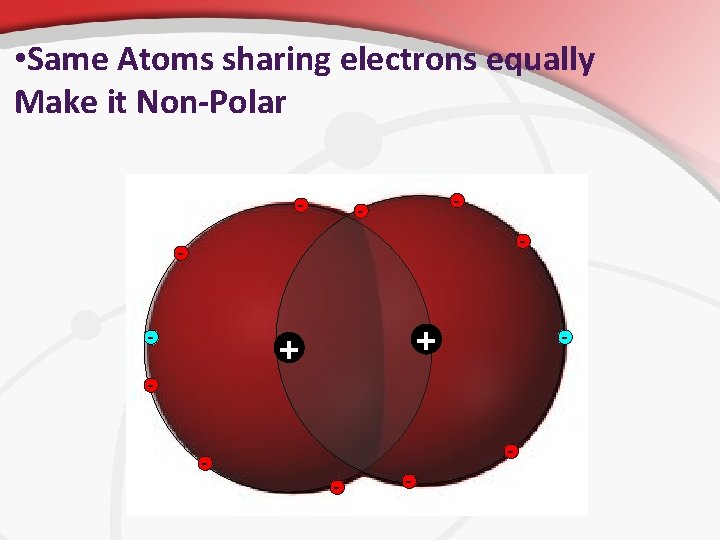
• Same Atoms sharing electrons equally Make it Non-Polar - - - + + - -
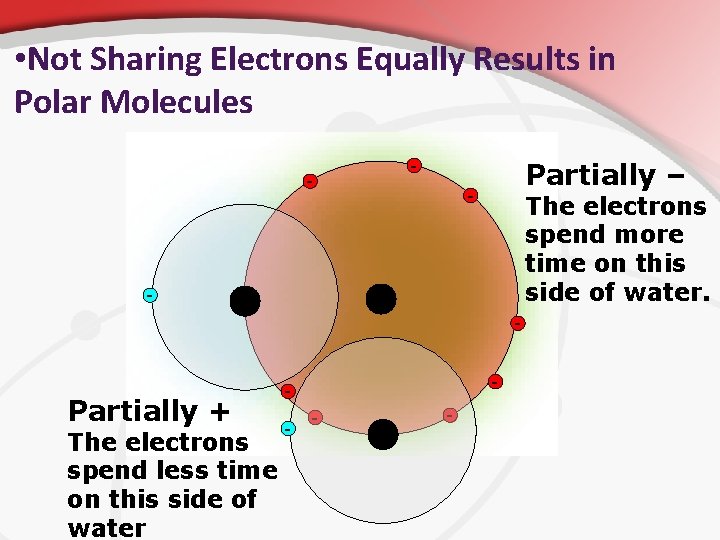
• Not Sharing Electrons Equally Results in Polar Molecules - - Partially – - The electrons spend more time on this side of water. - Partially + The electrons spend less time on this side of water - -
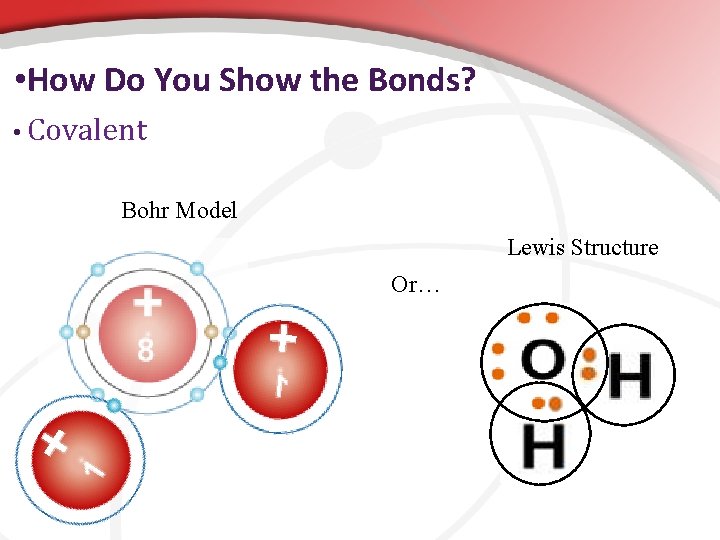
• How Do You Show the Bonds? • Covalent Bohr Model Lewis Structure Or…
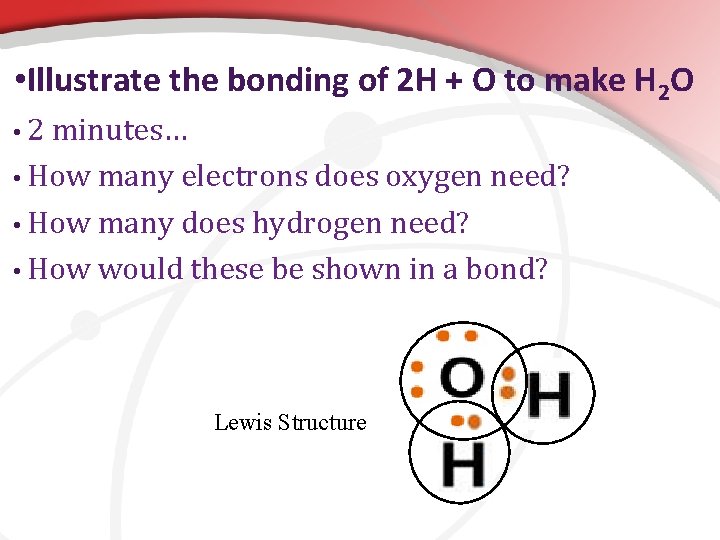
• Illustrate the bonding of 2 H + O to make H 2 O • 2 minutes… • How many electrons does oxygen need? • How many does hydrogen need? • How would these be shown in a bond? Lewis Structure
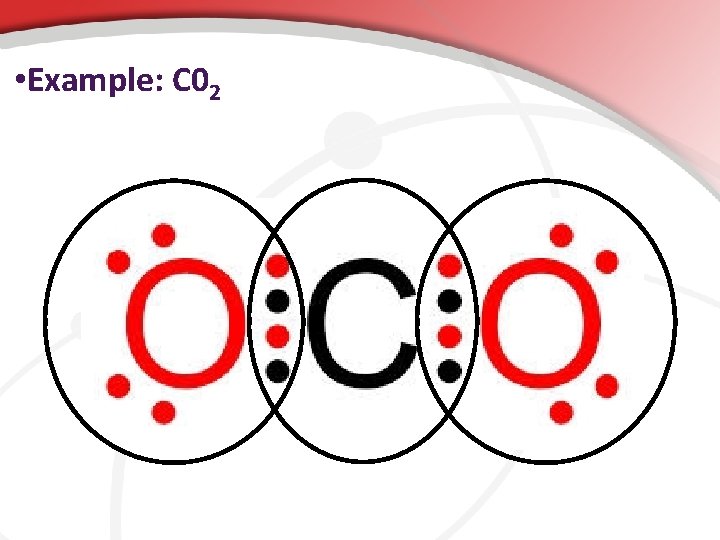
• Example: C 02
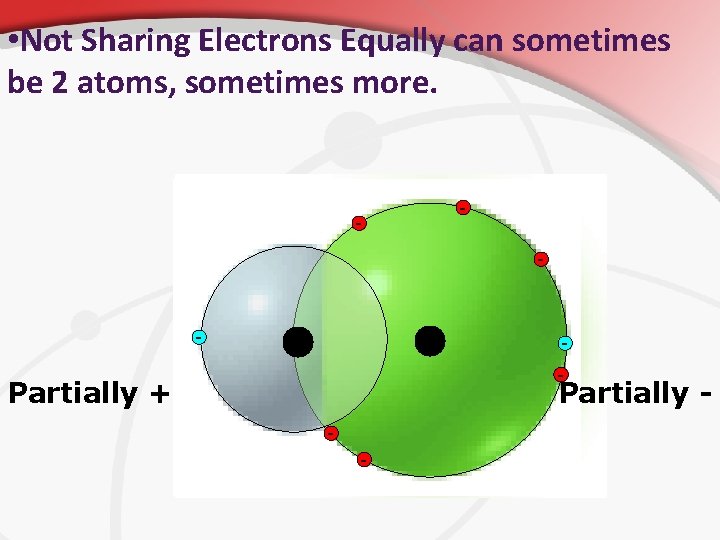
• Not Sharing Electrons Equally can sometimes be 2 atoms, sometimes more. - - Partially + Partially - -
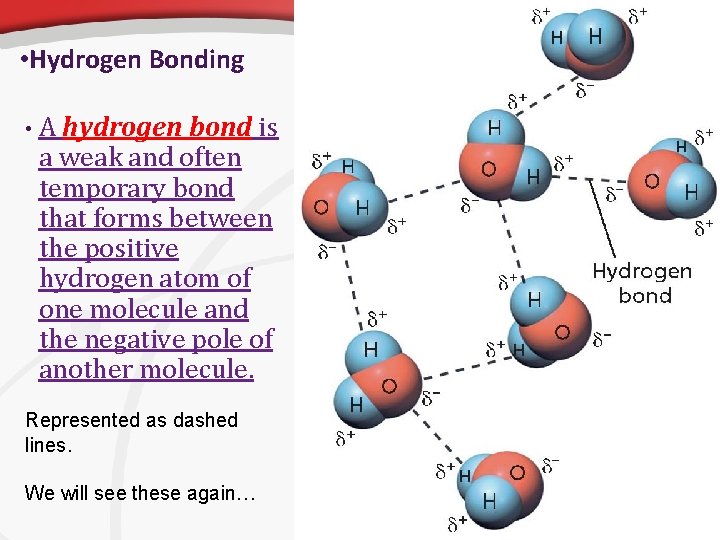
• Hydrogen Bonding • A hydrogen bond is a weak and often temporary bond that forms between the positive hydrogen atom of one molecule and the negative pole of another molecule. Represented as dashed lines. We will see these again…

• Polarity, continued Hydrogen Bonds • When bonded to an oxygen, nitrogen, or fluorine atom, a hydrogen atom has a partial positive charge nearly as great as a proton’s charge. • It attracts the negative pole of other nearby molecules. • This attraction is stronger than attractions between other molecules, but not as strong as covalent bonds. • However, hydrogen bonding plays an important role in many of the molecules that make up living things.
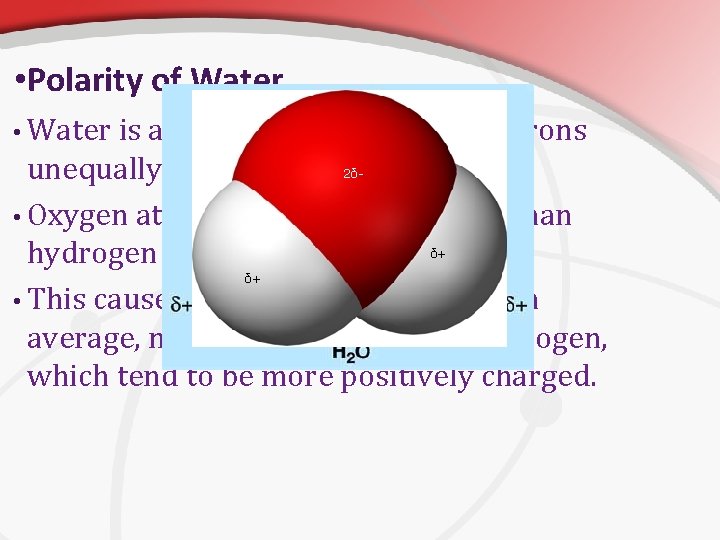
• Polarity of Water • Water is a molecule that shares electrons unequally. • Oxygen attracts the electrons more than hydrogen does. • This causes the oxygen atom to be, on average, more negative than the hydrogen, which tend to be more positively charged. 2δ- δ+ δ+
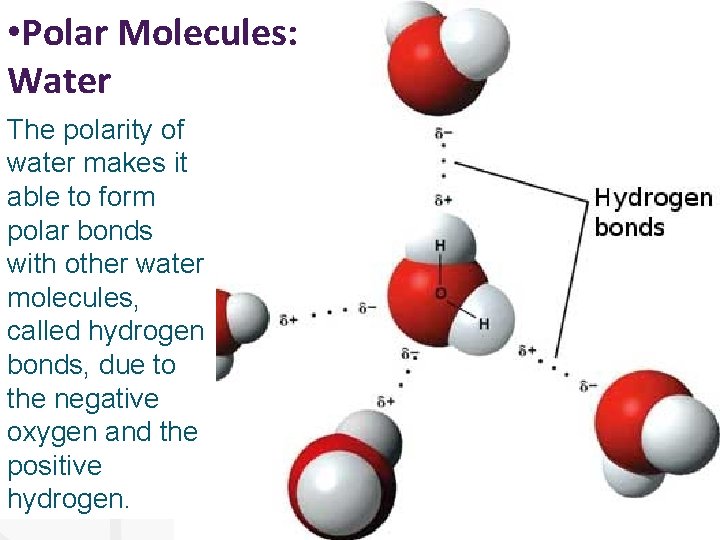
• Polar Molecules: Water The polarity of water makes it able to form polar bonds with other water molecules, called 2δhydrogen bonds, due to δ+ δ+ the negative oxygen and the positive hydrogen.

• Atomic Models • There a variety of ways to represent an atom… • It really depends on why you are showing the atom that will determine how you show it. Electron-Dot Diagrams = show electrons & protons for ions & bonding • Space filling models = show the molecule looks in 3 D • Bohr’s model = shows all electrons to represent energy levels. WHAT YOU BUILT • Lewis structures = shows valence electrons (only) for bonding. Also Built. •

• Concept Review • • • • • What are three subatomic particles? Protons, neutrons, electrons Where are they located? P=/n 0 in nucleus, e- in electron cloud Do electrons go anywhere they want? No. they exist in distinct regions called energy levels What is an isotope? An atom of an element that has too many or too few neutrons. What is an ionic bond? Bond that forms between atoms that swap electrons. What is a covalent bond? Bond that forms between atoms that shares electrons. What makes a molecule polar? When there is a partial negative charge on one side with a partial positive charge on the other of the covalent molecule. Which is a Lewis structure & which is a Bohr’s model? Lewis = Symbol + valence electrons Bohr = protons and neutrons in nucleus surrounded by all energy levels with electrons distributed properly in them.
• Why Bonding is Important? • The reason why we review bonding is because the ability of atoms to interact with each other allows for them to build larger molecules. • Depending on the types of numbers of atoms an almost limitless number of molecules and compounds can be formed, each with its own unique properties. • These build on one another to make life possible.
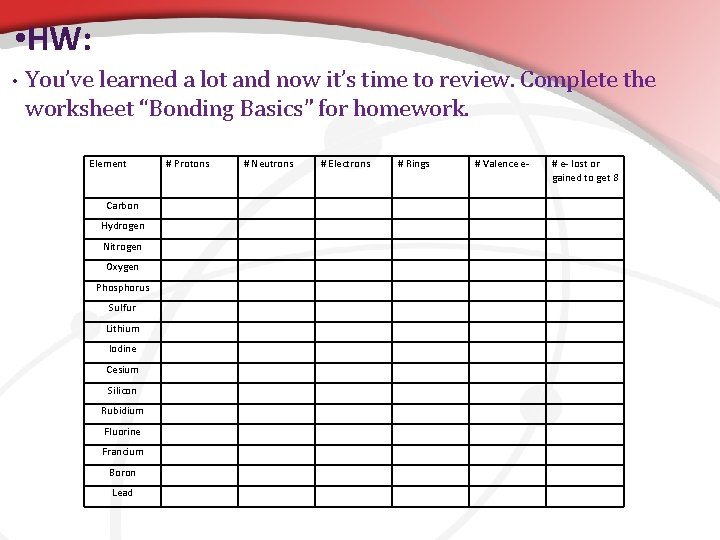
• HW: • You’ve learned a lot and now it’s time to review. Complete the worksheet “Bonding Basics” for homework. Element Carbon Hydrogen Nitrogen Oxygen Phosphorus Sulfur Lithium Iodine Cesium Silicon Rubidium Fluorine Francium Boron Lead # Protons # Neutrons # Electrons # Rings # Valence e- # e- lost or gained to get 8

• Closure… • Questions? • What did you learn today? • Reflect in your warm ups…
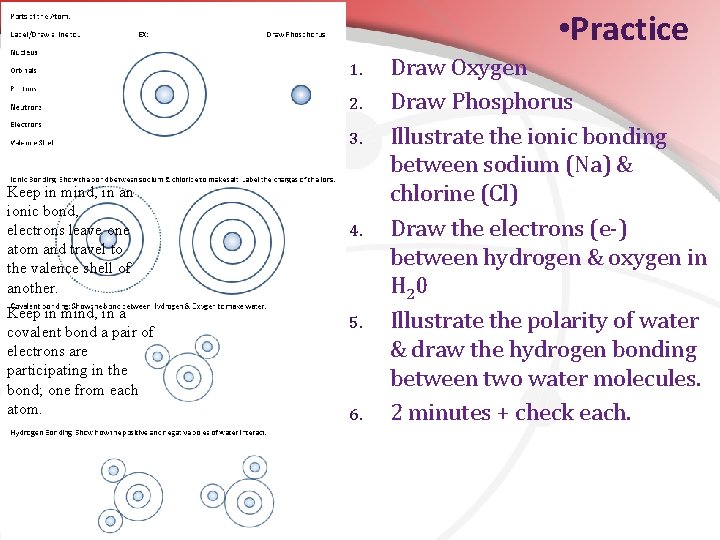
• Practice 1. 2. 3. Keep in mind, in an ionic bond, electrons leave one atom and travel to the valence shell of another. Keep in mind, in a covalent bond a pair of electrons are participating in the bond; one from each atom. 4. 5. 6. Draw Oxygen Draw Phosphorus Illustrate the ionic bonding between sodium (Na) & chlorine (Cl) Draw the electrons (e-) between hydrogen & oxygen in H 20 Illustrate the polarity of water & draw the hydrogen bonding between two water molecules. 2 minutes + check each.

• The following slides are not shown and not necessary to look at…
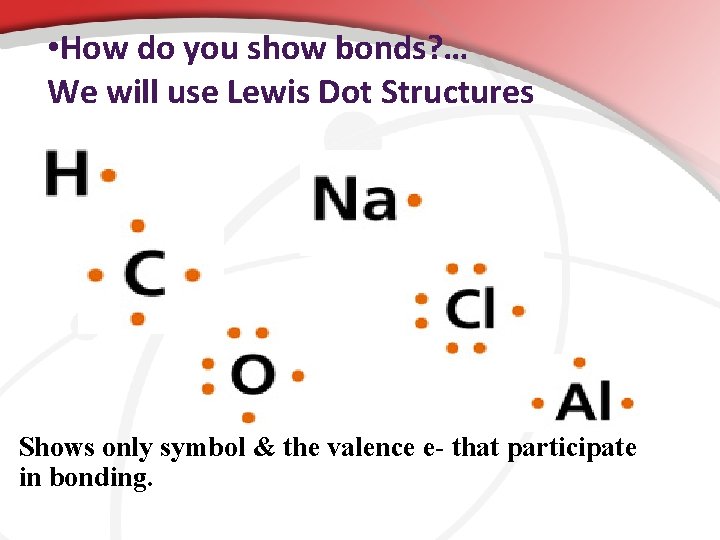
• How do you show bonds? … We will use Lewis Dot Structures Shows only symbol & the valence e- that participate in bonding.

• Sample Questions: Self Quiz. Answers on the next slide. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. A(n) _____ is the smallest unit of matter that cannot be broken down by chemical means. Identify the 3 sub atomic particles of atoms and their locations in words or in an illustration. A(n) _____ is a substance made of only one kind of atom that has the same number of protons. Locate oxygen and tell me how many protons, neutrons, and electrons it has. (use your periodic table…the atomic number is extremely useful). Atoms are most stable when they have 8 electrons in their ______. How many protons, electrons, neutrons, energy levels, and valence electrons does carbon have? Name two ways that atoms can form bonds (combine to become more stable). Describe polarity and a popular polar molecule. Explain how polarity can cause salt & sugar to dissolve in water. Water is an example of a compound that is held together by ____ bonds. Atoms gain or lose ______ to form ions. The opposite ends of a polar molecule have ______ charges. A(n) _______ is a weak chemical attraction between polar molecules.

• Answers 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. atom is the smallest unit of matter that cannot be broken down by A(n) _____ chemical means. Identify the 3 sub atomic particles of atoms and their locations in words or in an illustration. p+, e-, n 0… p+, n 0 in nucleus, e- in electron cloud A(n)element _____ is a substance made of only one kind of atom that has the same number of protons. 8 8 8 Locate oxygen and tell me how many protons, neutrons, and electrons it has. (use your periodic table…the atomic number is extremely useful). valence _____. shell Atoms are most stable when they have 8 electrons in their _____ 6 6 6 2 4 How many protons, electrons, neutrons, energy levels, and valence electrons does carbon have? Name two ways that atoms can form bonds (combine to become more stable). Ionic (swapping electrons), covalent (sharing electrons) When a molecule has a partial + & partial – Describe polarity and a popular polar molecule. charge. Water is polar and can dissolve the ions & polar molecules in sugar & salt Explain how polarity can cause salt & sugar to dissolve in water. because of polarity. covalent bonds. Water is an example of a compound that is held together by ______ electrons to form ions. Atoms gain or lose ________ The opposite ends of a polar molecule have opposite ______ charges. bond is a weak chemical attraction between polar molecules. A(n)hydrogen _____

• Quick Facts. Put an atom into perspective. • If you used a stadium to model and atom… The Houston Astrodome (Home of the Houston Astros…a baseball team) seats 60, 000 fans, covers 9 acres and the dome rises to a height of 200 ft. ++ • If you could blow up an +atom to the size of this stadium +++ a basketball could represent the volume of an atom's nucleus. • The rest of the open area is the volume of where the electrons, maybe the size of tiny BB’s, would be constantly flying around at speeds approaching the speed of light through the electron cloud in their orbitals. •
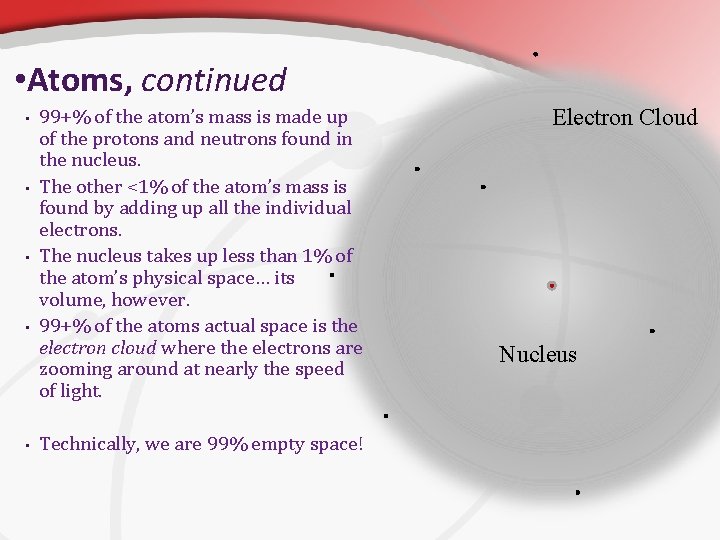
• Atoms, continued • • • 99+% of the atom’s mass is made up of the protons and neutrons found in the nucleus. The other <1% of the atom’s mass is found by adding up all the individual electrons. The nucleus takes up less than 1% of the atom’s physical space… its volume, however. 99+% of the atoms actual space is the electron cloud where the electrons are zooming around at nearly the speed of light. Technically, we are 99% empty space! Electron Cloud Nucleus

• Technically, We Are 99. 9% Empty Space? • Why do we appear solid? • • Think about a plane propeller. Then imagine hundreds stacked on top of each other. Our skin is hundreds of cells thick, constructed of millions of atoms each.

• Why Do Things Feel Solid? We actually feel very little that is solid. What we feel is the interpretation of what solid is because of the electron clouds pushing away from one another.

• CW/HW Answers Oxygen Sodium Chlorine Tellurium

• CW/HW Answers Oxygen Sodium Chlorine Uranium Sorry, bad example
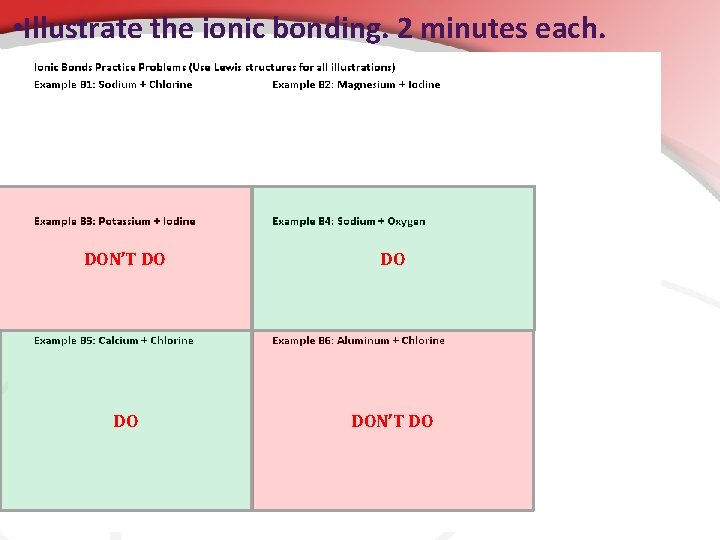
• Illustrate the ionic bonding. 2 minutes each. DON’T DO

• Illustrate the covalent binding. 2 minutes each DON’T DO DO DO
- Slides: 87