What would happen if the number of protons

• What would happen if the number of protons were to change in an atom? • Here are three isotopes of an element: 12 C 6, 13 C 6, 14 C 6 • a. The element is: _________ • b. The number 6 refers to the ____ • c. The numbers 12, 13, and 14 refer to the _____ • d. How many protons and neutrons are in the first isotope? _________ • e. How many protons and neutrons are in the second isotope? _________ • f. How many protons and neutrons are in the third isotope? _________

Isotopes and Ions Variations on the Atom Dr. M. Hazlett Mandeville High School

Isotopes • All atoms of an element have the SAME number of protons (p+) • The p+ number is the atomic number (Z) – This is a constant – For example: All Sodium (Na) atoms have 11 p+ – If an atom loses a proton, it becomes a different element • If Na loses 1 p+, then it has become Neon (Ne)

Z = atomic number = + p • The number of protons identifies the atom and which element it is • In a stable atom: – # p+ = # n 0 = # e – Thus, Na in its stable form has 11 p+; 11 n 0; and 11 e– If it has an unequal number of p+ and n 0, then it is called an ISOTOPE

• Theoretically – an element can have as many isotopes of itself as it has neutrons, or it can add an unlimited number of n 0 • For example: H has 3; C has 16; Al has 25 – These can be looked up in the CRC (the Chemistry/Physics Data Bible) or on the internet – Remember – a change in the number of n 0 does not change the element’s atom – only a change in the number of protons can do that!
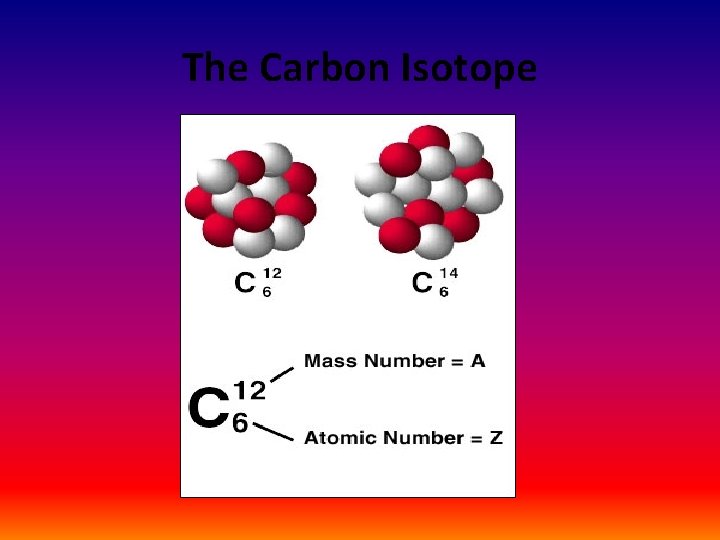
The Carbon Isotope

Ions • Ions are when an atom has an unequal number of p+ and e • Remember – a stable atom has a neutral overall charge due its equal number of p+ and e • When an atom loses or gains an e-, its charge changes accordingly – Loss of e- means a + charge; gaining an e- means a – charge for the atom

Losing or Gaining e-. . . • If an atom loses an e-, then it has more p+ than e- and it will have an overall positive charge • Different elements’ atoms can lose 1, 2, 3, or even 4 electrons depending on various factors • If an atom has LOST e-, then it is called a CATION or a positive ion – A Cation would be written as Al+ (the one being understood) or Al+3

• Atoms can also gain electrons • If an atom gains electrons (from 1 up to 4), then it will have more e- than p+ and will end up having an overall negative charge • A negatively charged ion is called an ANION – The element is shown this like: Na- (the 1 is understood) or Na-2 • The losing or gaining of electrons determines what type of bonds the atoms will form, and which atoms will bond to others
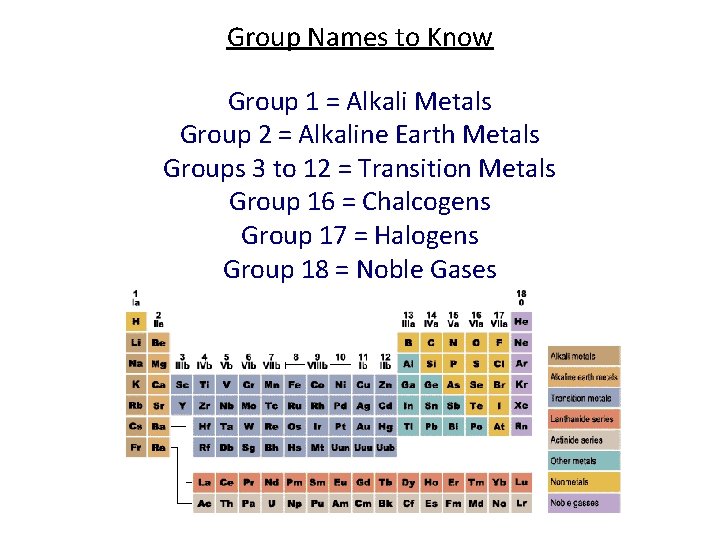
Group Names to Know Group 1 = Alkali Metals Group 2 = Alkaline Earth Metals Groups 3 to 12 = Transition Metals Group 16 = Chalcogens Group 17 = Halogens Group 18 = Noble Gases
- Slides: 10