What is the periodic table compact way of


What is the periodic table? • compact way of organizing elements • contains a lot of information • allows us to make predictions about behavior and properties of elements • Elements


History of the Periodic Table • End of 1700’s – less than 30 elements known • Many elements discovered during 1800’s • Many experiments done to determine atomic masses
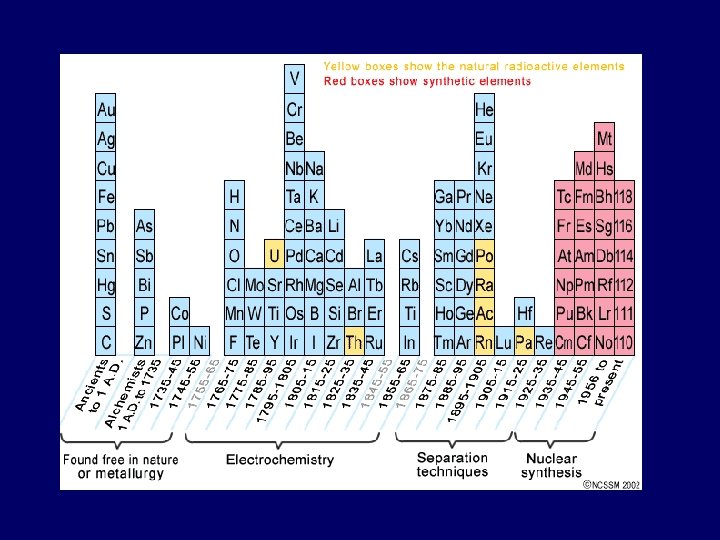

John Newlands • 1864: if elements arranged by atomic mass - properties repeat every 8 th element • Law of Octaves – did not work for all known elements • Key idea was correct: Properties of elements do repeat in periodic way

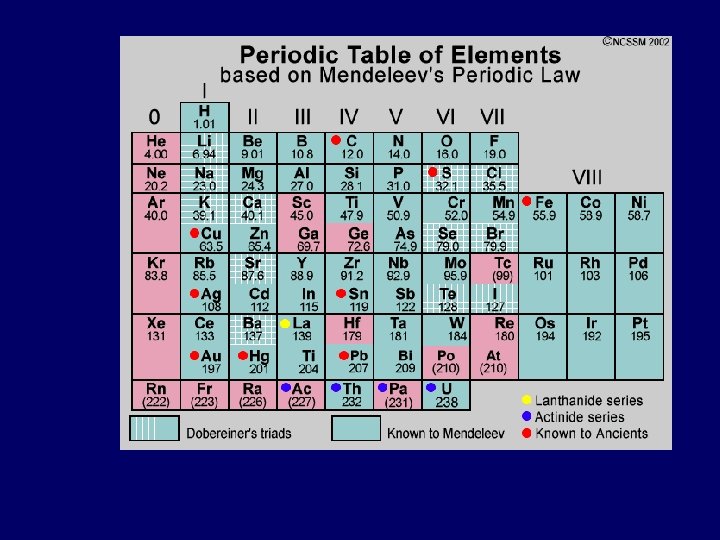
Mendeleev & Meyer 1869: Mendeleev produced 1 st accepted PT • Elements ordered by ↑ atomic mass into columns with similar properties • Predicted existence & properties of undiscovered elements • Not totally correct – more accurate atomic mass calculations showed some elements weren’t in right place

Remember 1860’s: • Dalton’s billiard ball model of the atom • No subatomic particles yet discovered

1913 – Henry Moseley • by 1913, protons & electrons discovered – neutrons were predicted • Moseley determined atoms of each element contain unique # protons (= atomic number) • re-arranged Mendeleev’s PT according to atomic number instead of atomic mass • problems with elements in wrong place disappeared

Periodic Law There is a periodic repetition of chemical and physical properties of elements when arranged by increasing atomic number

Glenn Seaborg: 1950’s • Lanthanide and Actinide Series
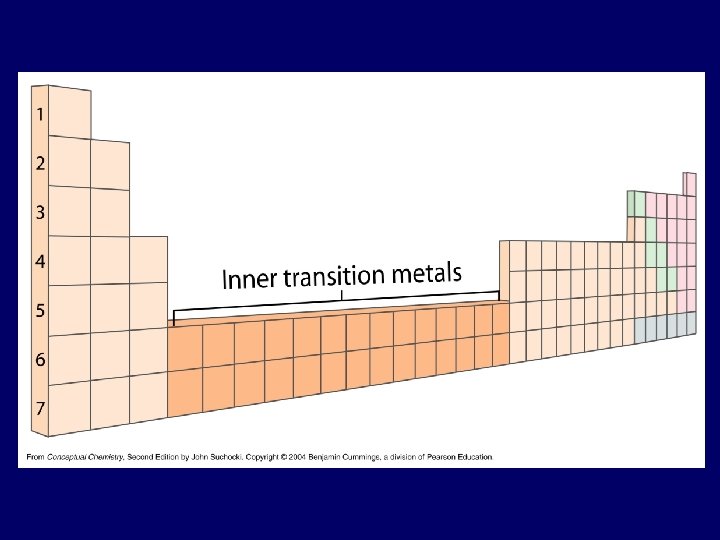

Newlands Seaborg Mendeleev Mosley

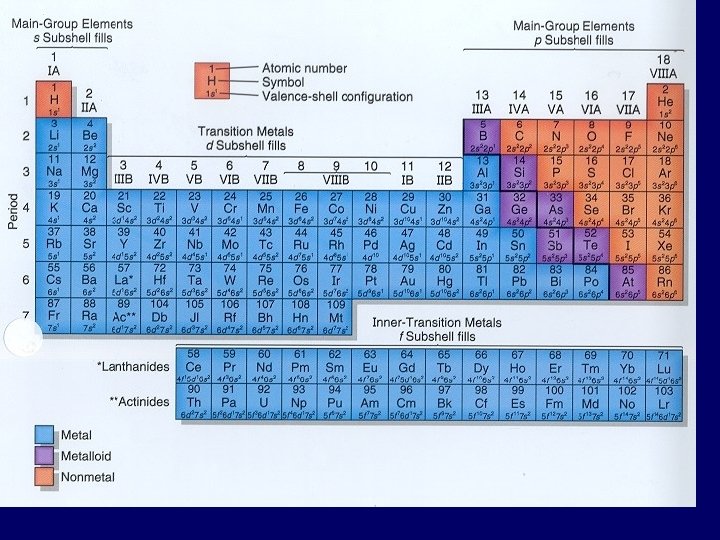
Vocabulary of PT • Columns called groups or families – Today: #1 thru 18, Arabic numerals – Past: A & B groups, Roman numerals • A-Group – Columns 1, 2, 13 -18 (= representative elements) – IA – 8 A • B-Group – Transition metals (columns 3 -12) – IB - 8 B • Rows are called series or periods – #1 thru 7
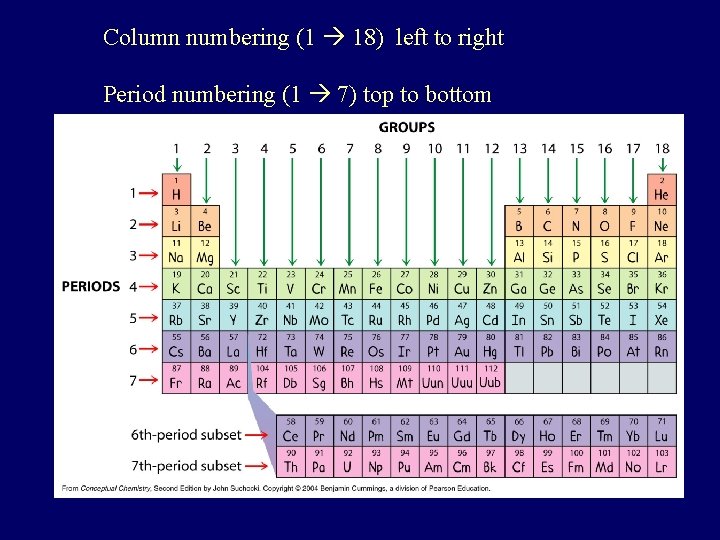
Column numbering (1 18) left to right Period numbering (1 7) top to bottom

Structure of Periodic Table • Closely related to electron configuration of each element

Energy Levels = Row Number • Elements in same row have same # of principal energy levels – so # of principal energy levels = to row #
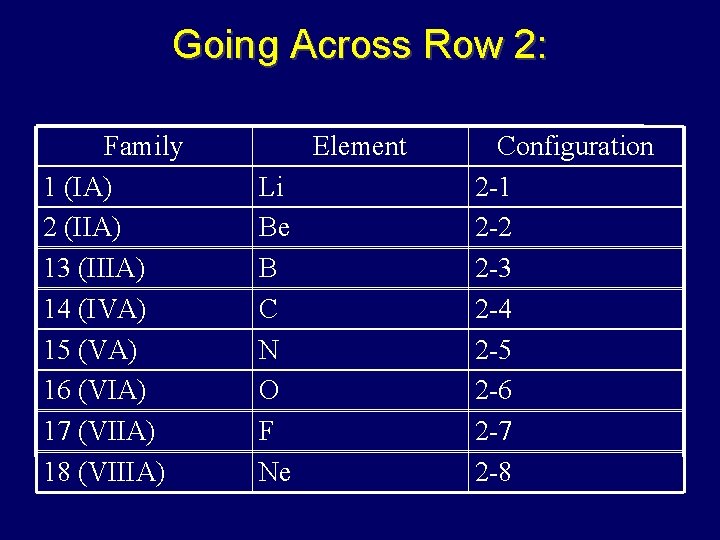
Going Across Row 2: Family 1 (IA) 2 (IIA) 13 (IIIA) 14 (IVA) 15 (VA) 16 (VIA) 17 (VIIA) 18 (VIIIA) Element Li Be B C N O F Ne Configuration 2 -1 2 -2 2 -3 2 -4 2 -5 2 -6 2 -7 2 -8

Valence Electrons • Chemical behavior determined by # valence electrons • Elements with same # valence electrons will have similar chemical properties – Elements in same column have similar chemical properties
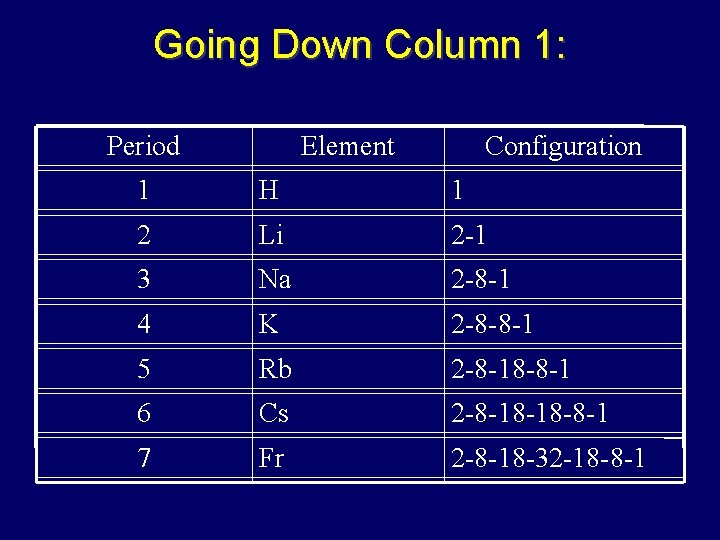
Going Down Column 1: Period Element Configuration 1 H 1 2 Li 2 -1 3 Na 2 -8 -1 4 K 2 -8 -8 -1 5 Rb 2 -8 -18 -8 -1 6 Cs 2 -8 -18 -18 -8 -1 7 Fr 2 -8 -18 -32 -18 -8 -1
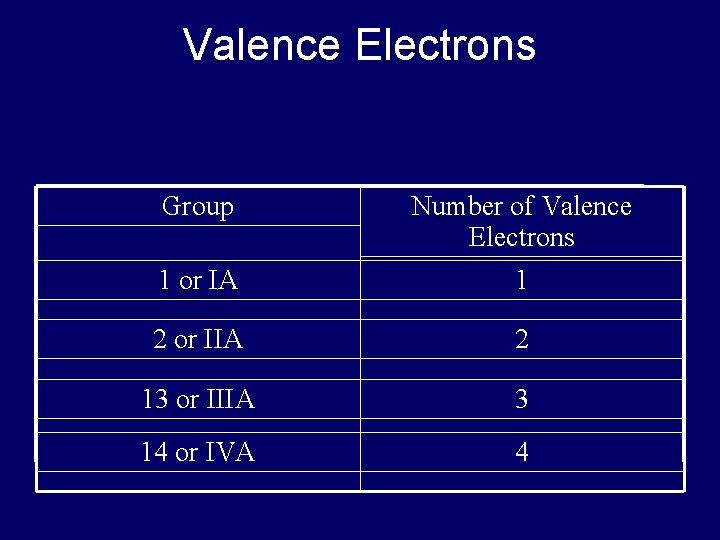
Valence Electrons Group Number of Valence Electrons 1 or IA 1 2 or IIA 2 13 or IIIA 3 14 or IVA 4

Classifying the Elements • 2/3 (75%) of elements are metals • Remaining elements: non-metals & metalloids (semi-metals) • Metalloids: – have properties of both metals & non-metals • Staircase: – dividing line between metals & non-metals – elements to left are metals (except H) – elements to right are non-metals

Properties of Metals • • • malleable – flattened into sheets ductile – drawn into wires & tubes have Luster good conductors of heat & electricity solid at room temperature (except Hg) metals lose electrons & form positive ions “Metals are losers” • low ionization energy • low electronegativity

Properties of Non-metals • • • generally gases or solids (except Br 2) solids are brittle solids are dull poor conductors of heat & electricity non-metals gain electrons & form negative ions “Nonmetals are winners” • high ionization energy • high electronegativity

Properties of Metalloids 7 metalloids: – 5 on right of staircase: B, Si, As, Te, At – 2 on left of staircase: Ge, Sb Each metalloid has some metallic and some nonmetallic properties example: Si shiny like metal but brittle like non-metal

Names of Families (AKA group A elements) • • • Group 1 = Alkali Metals Group 2 = Alkaline Earth Metals Groups 3 -12: Transition metals Group 13 = Boron family Group 14 = Carbon family Group 15 = Nitrogen family Group 16 = Oxygen family Group 17 = Halogens Group 18 = Noble Gases

Transition Metals • Groups 3 through 12 • AKA group B elements • Actinide & Lanthanide series – inner transition elements • put the COLOR in your life – form brightly colored salts/solutions

Elements that are gases at STP Diatomics: H 2, N 2, O 2, F 2, Cl 2 Monatomics: noble gases He, Ne, Ar, Kr, Xe, Rn

Two elements are liquid at room temperature Br 2 (non-metal) and Hg (metal)

All other elements are solids at room temperature
- Slides: 33