What is the periodic table A compact way


What is the periodic table? • A compact way of organizing the elements that contains a lot of information and allows us to make predictions about the behavior and properties of the elements. • Elements


History of the Periodic Table • End of the 1700’s – less than 30 elements known. • Many elements were discovered during the 1800’s. Many experiments were done to determine atomic masses.


John Newlands - Octaves • 1864: Newlands noticed that when the known elements were arranged by atomic masses, their properties repeated every 8 th element. • Law of Octaves did not work for all known elements. • Key idea was correct: Properties of elements do repeat in a periodic way.

Mendeleev & Meyer • 1869: Mendeleev produced 1 st accepted periodic table. • Elements ordered by increasing atomic mass into columns with similar properties. • Predicted the existence & properties of undiscovered elements. (Sc, Ge, Ga) • Not totally correct. As more accurate determinations of atomic mass were made, several elements weren’t in the right place.



Remember • 1860’s: No subatomic particles had been discovered yet. • People were going by Dalton’s billiard ball model of the atom.

1913 – Henry Moseley • By 1913, protons & electrons discovered. Neutrons predicted. • When cathode rays hit stuff, they produce X-rays. • Mosely used 30 different elements as “stoppers” • Found that the greater the atomic weight of the “stopper, ” the shorter the wavelength of the xrays.


What does any good scientist do? PLOTS THE DATA! • Tried correlating the wavelength of the x-ray with the atomic mass of the stopper. Not so nice. Change in much more regular than change in atomic weight. • Then tried correlating the wavelength of the x-ray with an integer, n. Got a very pretty graph.


Atomic Number - 1913 • Mosley interpreted the integer, n, to be the positive charge on the nucleus. • Mosley suggested that the size of the nuclear charge increased by 1 with each step up the periodic table. (Before, it was organized by atomic weight. )

1913 – Moseley discovers Atomic # • Moseley determined that atoms of each element contain a unique number of protons – atomic number. • Moseley rearranged Mendeleev’s periodic table by atomic number instead of mass. • Problems disappeared.

Periodic Law • There is a periodic repetition of chemical and physical properties of the elements when they are arranged by increasing atomic number

Glenn Seaborg: 1950’s • Lanthanide and Actinide Series




Vocabulary of the P. T. • Columns are called groups or families 2 different notations. – 1 thru 18, Arabic numerals. – Split into A & B groups. Use Roman numerals I to VIII. • A-Group = Columns 1, 2, 13 -18 = representative elements • B-Group = Transition metals • Rows are called series or periods Numbered 1 thru 7.

Structure of the Periodic Table • Closely related to electron configuration.

Energy Levels = Row Number • Elements in the same row have the same number of principal energy levels. (The valence electrons of same-row elements have the same principal quantum number. ) • The number of principal energy levels is equal to the row number.
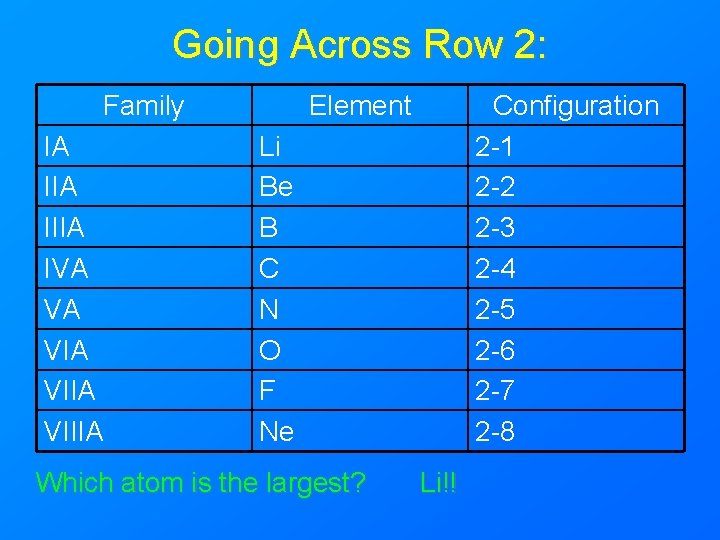
Going Across Row 2: Family IA IIIA IVA VA VIIA VIIIA Element Configuration 2 -1 2 -2 2 -3 2 -4 2 -5 2 -6 2 -7 2 -8 Li Be B C N O F Ne Which atom is the largest? Li!!
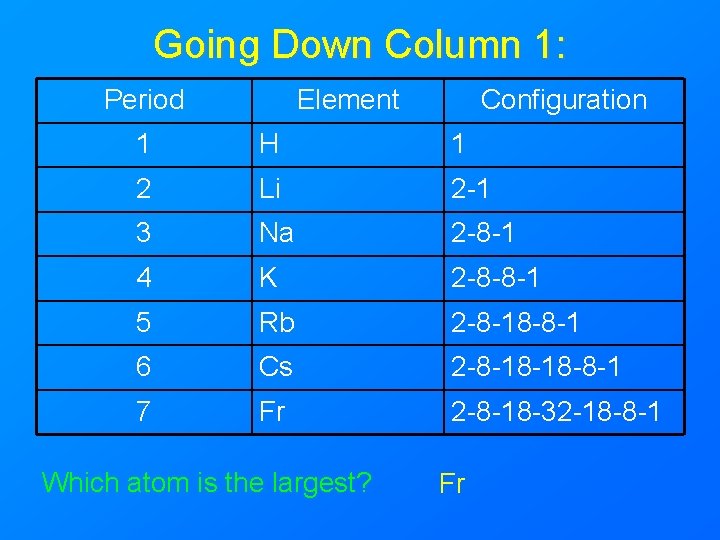
Going Down Column 1: Period Element Configuration 1 H 1 2 Li 2 -1 3 Na 2 -8 -1 4 K 2 -8 -8 -1 5 Rb 2 -8 -18 -8 -1 6 Cs 2 -8 -18 -18 -8 -1 7 Fr 2 -8 -18 -32 -18 -8 -1 Which atom is the largest? Fr
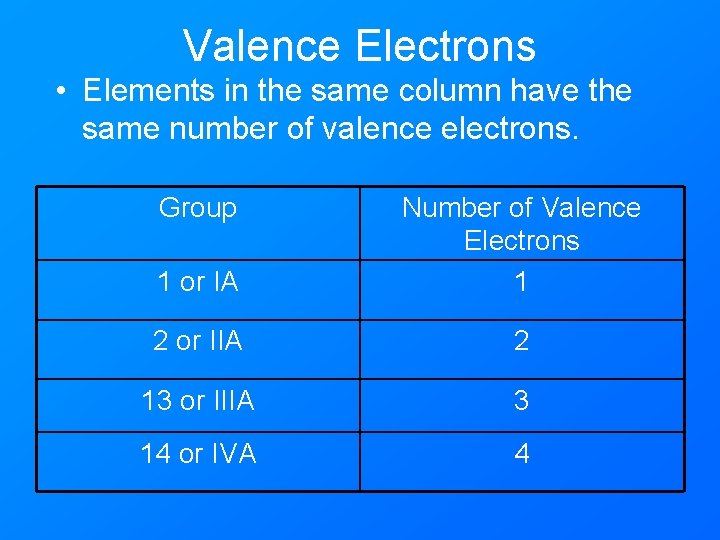
Valence Electrons • Elements in the same column have the same number of valence electrons. Group 1 or IA Number of Valence Electrons 1 2 or IIA 2 13 or IIIA 3 14 or IVA 4

Valence Electrons • Chemical behavior is determined by the number of valence electrons. • Elements with the same number of valence electrons will have similar chemical properties. • Elements in the same column have similar chemical properties.

Classifying the Elements • 2/3 of the elements are metals • Remaining elements are non-metals and metalloids (semi-metals). • Metalloids have some properties of metals and some properties of nonmetals. • Know the “staircase” staircase dividing line on the P. T. between metals & nonmetals. Everything to left, except H, is a metal.

Properties of Metals • • • Malleable – pounded into sheets Ductile – drawn into wires Have Luster Good conductors of heat & electricity Solids at room temperature, except Hg. Metals lose electrons to form positive ions. “Metals are losers. ” • Low ionization energy • Low electronegativity

Properties of Nonmetals • • • Generally gases or solids, except Br 2. Solids are brittle. Solids are dull. Poor conductors of heat & electricity. Nonmetals gain electrons to form negative ions. “Nonmetals are winners. ” • High ionization energy. • High electronegativity. • Properties are the OPPOSITE of metals.

Properties of Metalloids • Each metalloid has some metallic and some nonmetallic properties. • For example, Si is shiny like a metal but brittle like a nonmetal.

Names of Families • • Group 1 = Alkali Metals (IA) Group 2 = Alkaline Earth Metals (IIA) Group 17 = Halogens (XVIIA) Group 18 = Noble Gases (XVIIIA or O) – The noble gases are extremely unreactive. Weren’t discovered until 1890’s.

Transition Metals • Elements in Columns 3 through 12 • Also called the group B elements • Transition elements form brightly colored salts and brightly colored solutions. • Transition elements may form more than 1 positive ion. • Actinide and Lanthanide series = inner transition elements

Elements that are gases at STP H 2, N 2, O 2, F 2, Cl 2 + the noble gases – He, Ne, Ar, Kr, Xe, Rn

Elements that are liquids at STP Br 2 (non-metal) and Hg (metal)

Allotrope • Different forms of an element in the same phase. Have different structures and properties. • O 2 and O 3 - both gas phase • Graphite, diamond, buckey balls – all solid phase carbon.
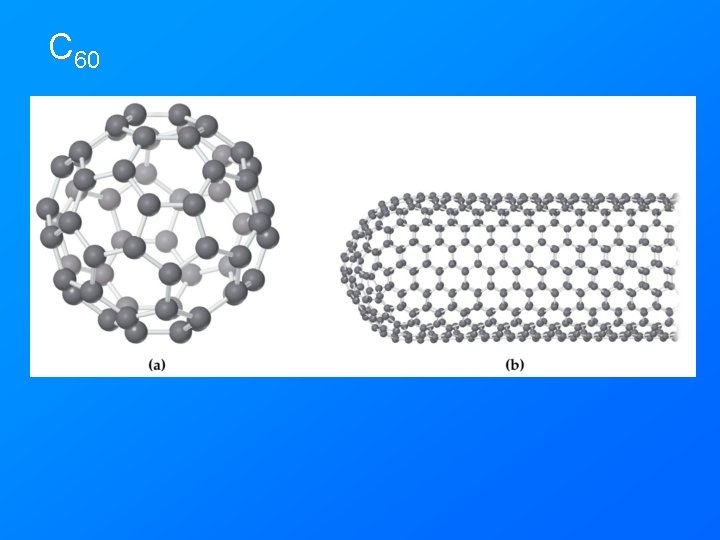
C 60

Graphite and Diamond
- Slides: 39