What Is the Optimal Systolic Blood Pressure Goal

What Is the Optimal Systolic Blood Pressure Goal in Hypertension Therapy? William C. Cushman, MD Chief, Preventive Medicine Section, Memphis VA Medical Center Professor, Preventive Medicine, and Physiology University of Tennessee Health Science Center
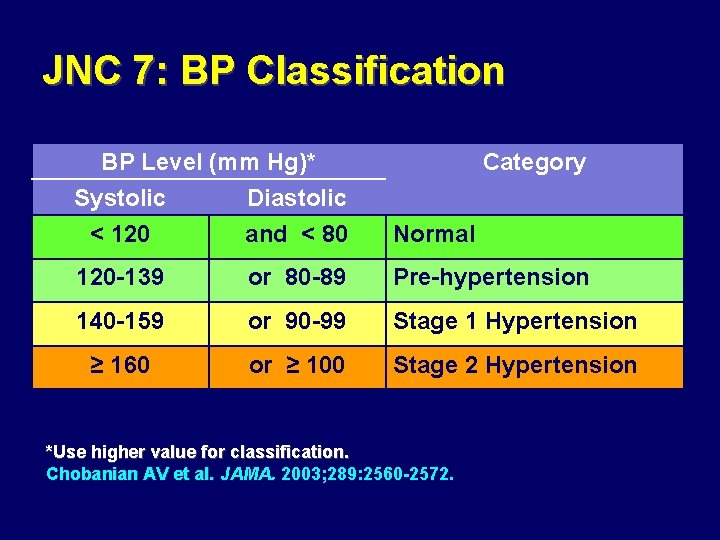
JNC 7: BP Classification BP Level (mm Hg)* Systolic Diastolic < 120 and < 80 Category Normal 120 -139 or 80 -89 Pre-hypertension 140 -159 or 90 -99 Stage 1 Hypertension ≥ 160 or ≥ 100 Stage 2 Hypertension *Use higher value for classification. Chobanian AV et al. JAMA. 2003; 289: 2560 -2572.
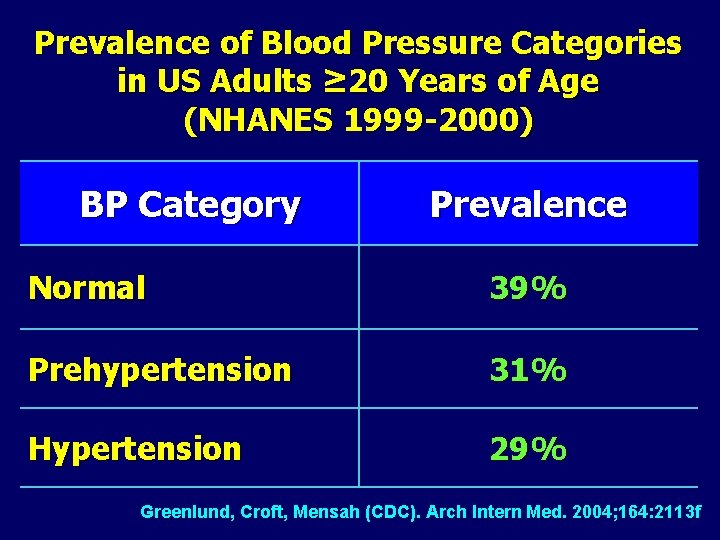
Prevalence of Blood Pressure Categories in US Adults ≥ 20 Years of Age (NHANES 1999 -2000) BP Category Prevalence Normal 39% Prehypertension 31% Hypertension 29% Greenlund, Croft, Mensah (CDC). Arch Intern Med. 2004; 164: 2113 f
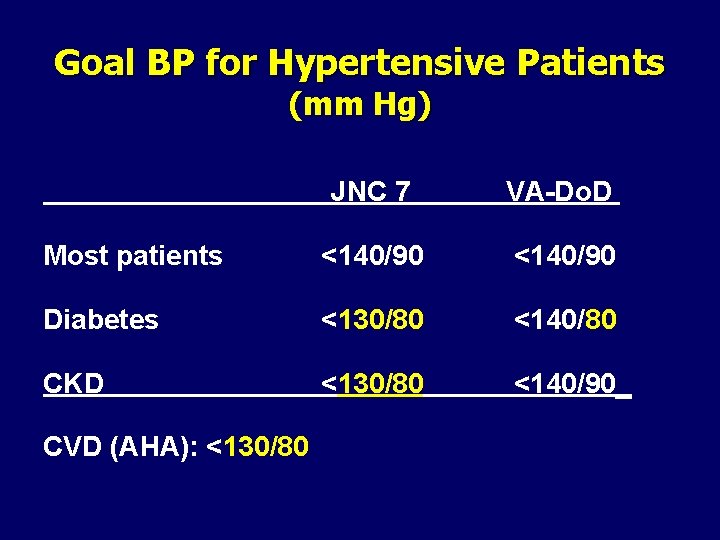
Goal BP for Hypertensive Patients (mm Hg) JNC 7 VA-Do. D Most patients <140/90 Diabetes <130/80 <140/80 CKD <130/80 <140/90_ CVD (AHA): <130/80
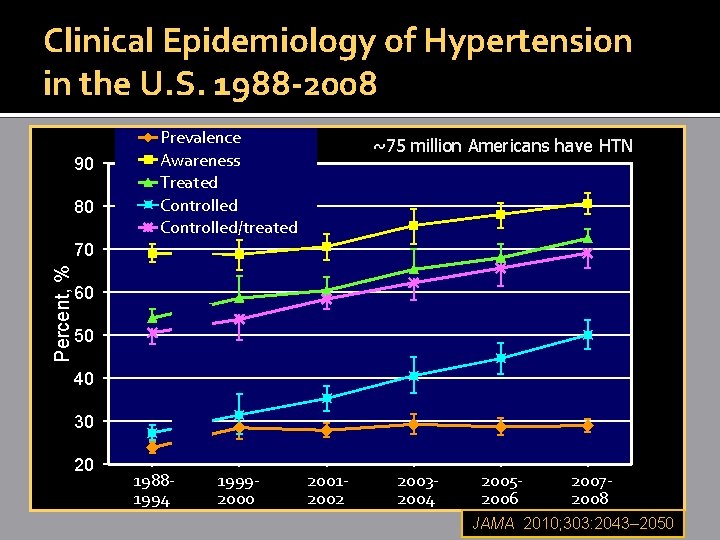
Clinical Epidemiology of Hypertension in the U. S. 1988 -2008 90 80 Prevalence Awareness Treated Controlled/treated ~75 million Americans have HTN Percent, % 70 60 50 40 30 20 19881994 19992000 20012002 20032004 20052006 20072008 JAMA 2010; 303: 2043– 2050
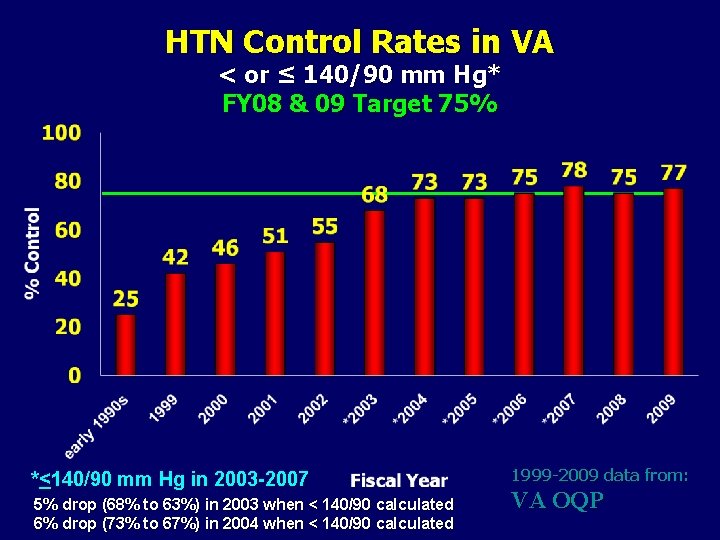
HTN Control Rates in VA < or ≤ 140/90 mm Hg* FY 08 & 09 Target 75% *<140/90 mm Hg in 2003 -2007 5% drop (68% to 63%) in 2003 when < 140/90 calculated 6% drop (73% to 67%) in 2004 when < 140/90 calculated 1999 -2009 data from: VA OQP

High Blood Pressure and CHD Risk Lancet 2002; 360: 1903– 13

High Blood Pressure and Stroke Risk Lancet 2002; 360: 1903– 13

Why is it important not to intensify medications to reduce BP below the level proven in trials? • Treating to lower BP levels may be harmful (J-curve? )
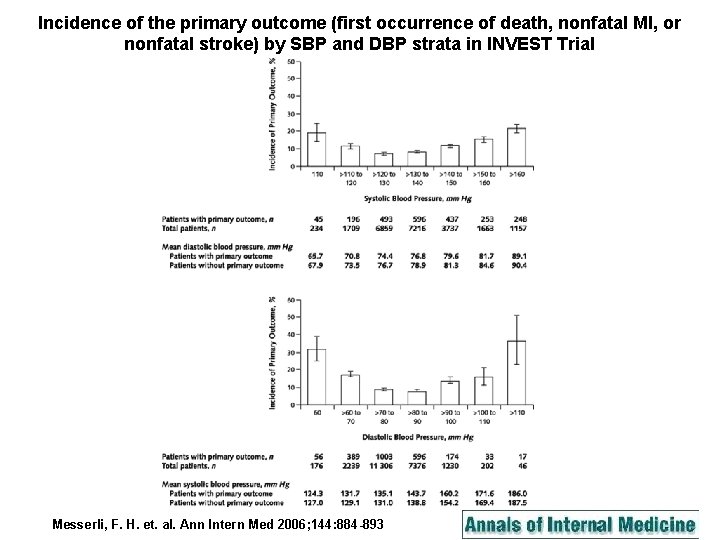
Incidence of the primary outcome (first occurrence of death, nonfatal MI, or nonfatal stroke) by SBP and DBP strata in INVEST Trial Messerli, F. H. et. al. Ann Intern Med 2006; 144: 884 -893

Why is it important not to intensify medications to reduce BP below the level proven in trials? • Treating to lower BP levels may be harmful (J-curve? ) • If neither beneficial nor harmful – Adding unneeded drugs wastes patients’ and payers’ resources and time: • More visits, drugs, monitoring, treating AEs – More drugs may contribute to reduced adherence to other evidence-based drug treatments (e. g. , statins and aspirin)

Systolic BP in Trials Testing SBP Goals: SHEP, Syst-Eur, Syst-China, HYVET SHEP Syst-Eur Syst-China HYVET Entry SBP 160 -219 160 -199 Goal SBP <148* <150 Baseline 170 174 171 173 Achieved : drug 142 151 144 Achieved: placebo 155 161 160 159 * Integrated SBP goal, personal communication, Barry Davis SBP = systolic blood pressure

What should the SBP treatment goal be in patients with hypertension? • Clinical trial evidence is very consistent for SBP goal <150 mm Hg. • Indirect evidence, mostly from trials with DBP goals, suggest SBP goal <140 mm Hg may be reasonable. • Public health: SBP goal <140 mm Hg has been recommended by many guidelines for several decades.

What should the Systolic BP treatment goal be in patients with diabetes mellitus?
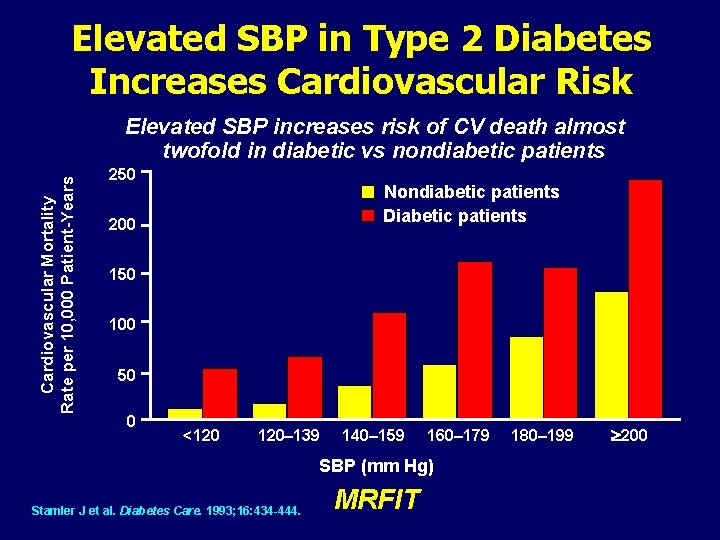
Elevated SBP in Type 2 Diabetes Increases Cardiovascular Risk Cardiovascular Mortality Rate per 10, 000 Patient-Years Elevated SBP increases risk of CV death almost twofold in diabetic vs nondiabetic patients 250 Nondiabetic patients Diabetic patients 200 150 100 50 0 <120 120– 139 140– 159 160– 179 SBP (mm Hg) Stamler J et al. Diabetes Care. 1993; 16: 434 -444. MRFIT 180– 199 ³ 200


ACCORD Double 2 x 2 Factorial Design Lipid Placebo BP (Systolic) Fibrate Intensive Standard <120 mm Hg <140 mm Hg Intensive Glycemic Control 1383 1374 1178 1193 5128 Standard Glycemic Control 1370 1391 1184 1178 5123 2765 2362 2371 10, 251 2753 5518 4733*

ACCORD BP Trial Design • Primary BP question: does a therapeutic strategy targeting systolic blood pressure (SBP) <120 mm Hg reduce CVD events compared to a strategy targeting SBP <140 mm Hg in patients with type 2 diabetes at high risk for CVD events? • 4733 participants with type 2 diabetes were randomly assigned to intensive (SBP <120 mm Hg) or standard (SBP <140 mm Hg) therapy. • Mean follow-up: 4. 7 years. • 94% power for 20% reduction in primary event rate, assuming standard group rate of 4%/yr and 5. 6 yrs follow-up.

Primary and Secondary Outcomes for ACCORD BP Trial Primary outcome -- composite of: ◦ Nonfatal MI, nonfatal stroke, CVD death Secondary outcomes: 1. Expanded macrovascular outcome: 1º + any revascularization and hospitalization for HF 2. major CAD events 3. nonfatal MI 4. combined fatal and nonfatal stroke 5. nonfatal stroke 6. total mortality 7. CV mortality 8. HF

ACCORD BP Trial Eligibility • Stable Type 2 Diabetes >3 months • Hb. A 1 c 7. 5% to 11% (or <9% if on more meds) • High CVD risk = clinical or subclinical disease or ≥ 2 risk factors • Age (limited to <80 years after Vanguard) ≥ 40 yrs with history of clinical CVD (secondary prevention) ≥ 55 yrs otherwise • Systolic blood pressure 130 to 160 mm Hg (if on 0 -3 meds) 161 to 170 mm Hg (if on 0 -2 meds) 171 to 180 mm Hg (if on 0 -1 meds) • Urine protein <1. 0 gm/24 hours or equivalent • Serum Creatinine ≤ 1. 5 mg/dl

ACCORD BP Protocol Many drugs/combinations provided to achieve goal BP according to randomized assignment Intensive Intervention: ◦ 2 -drug therapy initiated: thiazide-type diuretic + ACEI, ARB, or bblocker ◦ Drugs added and/or titrated at each visit to achieve SBP <120 mm Hg ◦ At periodic “milepost” visits: addition of another drug “required” if not at goal Standard Intervention: ◦ Intensify therapy if SBP ≥ 160 mm Hg @ 1 visit or ≥ 140 mm Hg @ 2 consecutive visits ◦ Down-titration if SBP <130 mm Hg @ 1 visit or <135 mm Hg @ 2 consecutive visits
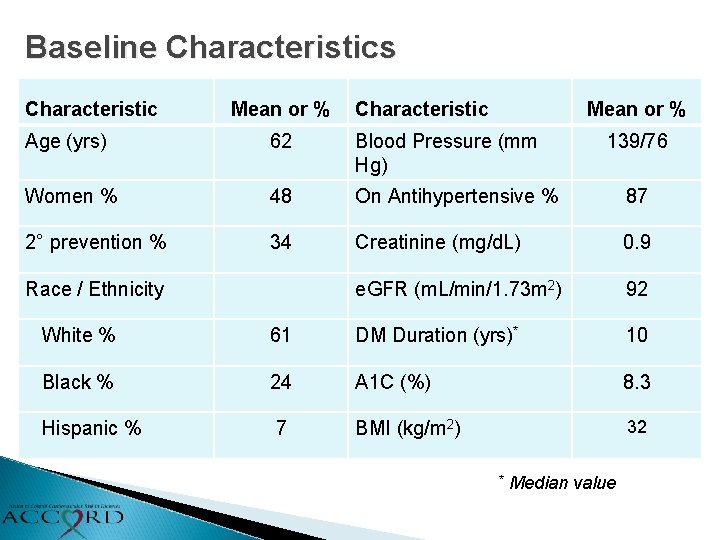
Baseline Characteristics Characteristic Mean or % Age (yrs) 62 Blood Pressure (mm Hg) Women % 48 On Antihypertensive % 87 2° prevention % 34 Creatinine (mg/d. L) 0. 9 e. GFR (m. L/min/1. 73 m 2) 92 Race / Ethnicity 139/76 White % 61 DM Duration (yrs)* 10 Black % 24 A 1 C (%) 8. 3 Hispanic % 7 BMI (kg/m 2) 32 * Median value
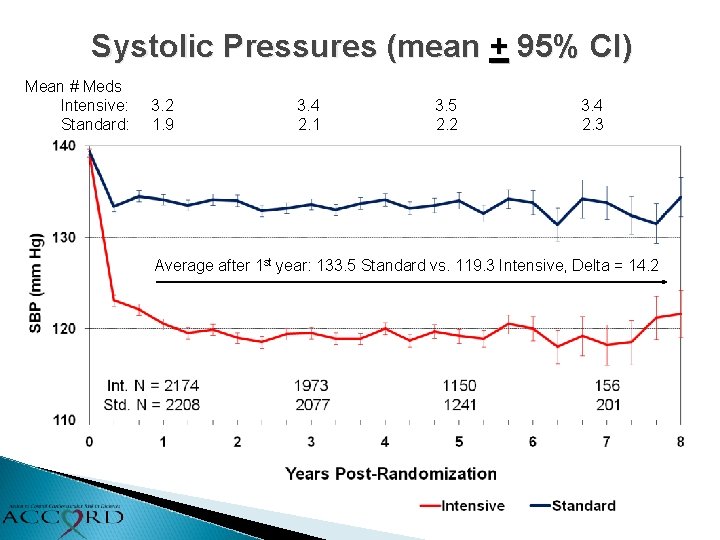
Systolic Pressures (mean + 95% CI) Mean # Meds Intensive: Standard: 3. 2 1. 9 3. 4 2. 1 3. 5 2. 2 3. 4 2. 3 Average after 1 st year: 133. 5 Standard vs. 119. 3 Intensive, Delta = 14. 2
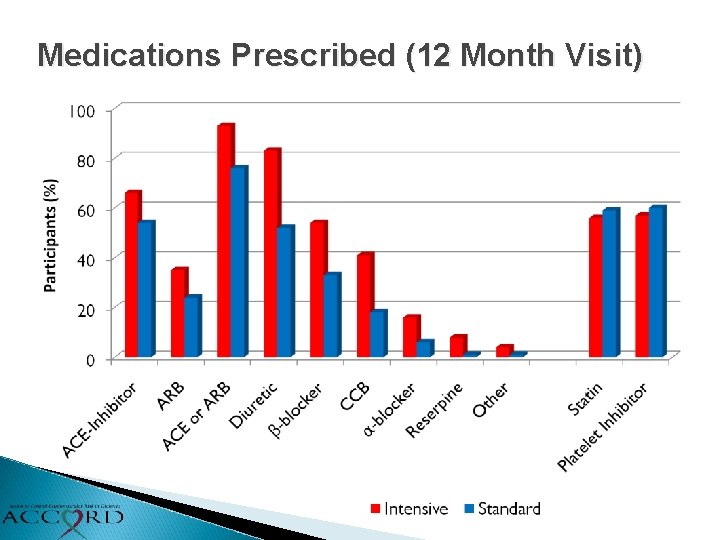
Medications Prescribed (12 Month Visit)
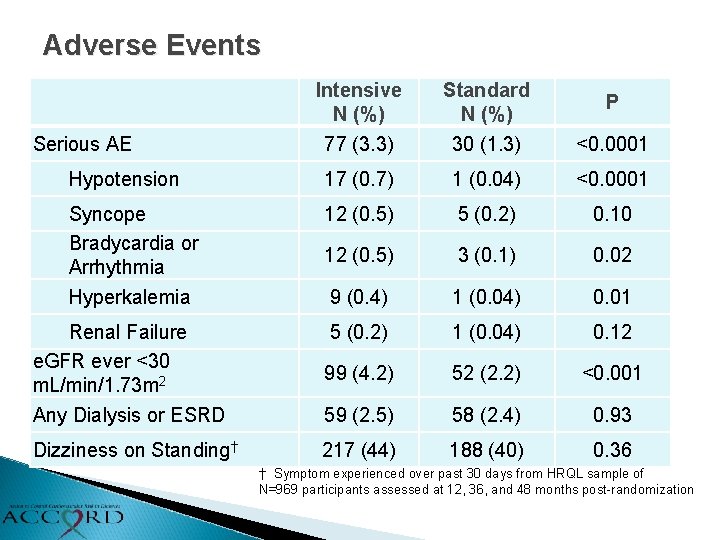
Adverse Events Intensive N (%) 77 (3. 3) Standard N (%) 30 (1. 3) <0. 0001 Hypotension 17 (0. 7) 1 (0. 04) <0. 0001 Syncope Bradycardia or Arrhythmia Hyperkalemia 12 (0. 5) 5 (0. 2) 0. 10 12 (0. 5) 3 (0. 1) 0. 02 9 (0. 4) 1 (0. 04) 0. 01 Renal Failure e. GFR ever <30 m. L/min/1. 73 m 2 Any Dialysis or ESRD 5 (0. 2) 1 (0. 04) 0. 12 99 (4. 2) 52 (2. 2) <0. 001 59 (2. 5) 58 (2. 4) 0. 93 Dizziness on Standing† 217 (44) 188 (40) 0. 36 Serious AE P † Symptom experienced over past 30 days from HRQL sample of N=969 participants assessed at 12, 36, and 48 months post-randomization
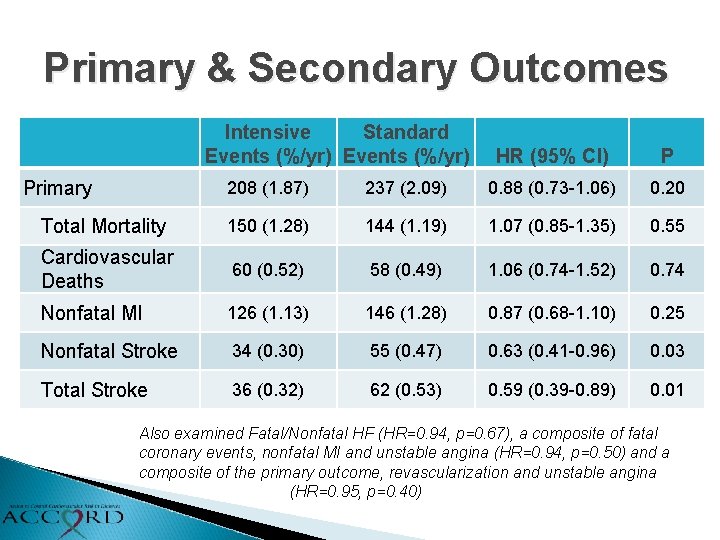
Primary & Secondary Outcomes Intensive Standard Events (%/yr) Primary HR (95% CI) P 208 (1. 87) 237 (2. 09) 0. 88 (0. 73 -1. 06) 0. 20 Total Mortality 150 (1. 28) 144 (1. 19) 1. 07 (0. 85 -1. 35) 0. 55 Cardiovascular Deaths 60 (0. 52) 58 (0. 49) 1. 06 (0. 74 -1. 52) 0. 74 Nonfatal MI 126 (1. 13) 146 (1. 28) 0. 87 (0. 68 -1. 10) 0. 25 Nonfatal Stroke 34 (0. 30) 55 (0. 47) 0. 63 (0. 41 -0. 96) 0. 03 Total Stroke 36 (0. 32) 62 (0. 53) 0. 59 (0. 39 -0. 89) 0. 01 Also examined Fatal/Nonfatal HF (HR=0. 94, p=0. 67), a composite of fatal coronary events, nonfatal MI and unstable angina (HR=0. 94, p=0. 50) and a composite of the primary outcome, revascularization and unstable angina (HR=0. 95, p=0. 40)
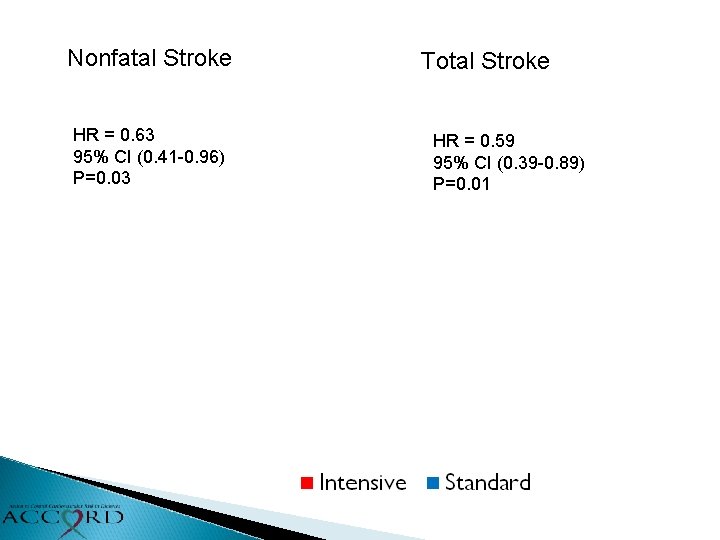
Nonfatal Stroke HR = 0. 63 95% CI (0. 41 -0. 96) P=0. 03 Total Stroke HR = 0. 59 95% CI (0. 39 -0. 89) P=0. 01
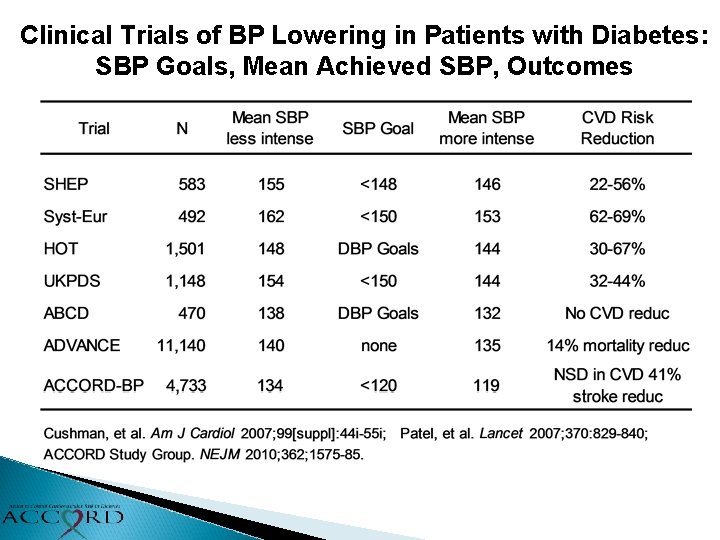
Clinical Trials of BP Lowering in Patients with Diabetes: SBP Goals, Mean Achieved SBP, Outcomes

Conclusions The ACCORD BP trial evaluated the effect of targeting a SBP goal of 120 mm Hg, compared to a goal of 140 mm Hg, in patients with type 2 diabetes at increased cardiovascular risk. The results provide no conclusive evidence that the intensive BP control strategy reduces the rate of a composite of major CVD events in such patients.
- Slides: 29