What is the evidence that a solution is






























- Slides: 33

What is the evidence that a solution is saturated? Postlab Session

What is a solution? What are the properties of solutions? How can particles of a solution pass through the filter paper? Why does a solution have a uniform appearance? Recap

1. 2. 3. 4. 5. What is the title of Activity No. 3? Give one objective for the activity. What was the solid solute used in the activity? How much water was placed on the container before the start of the activity? How many times should an experiment be done to be sure of the data gathered? Pop Quiz!

What is the evidence that a solution is saturated? 1. What is the title of Activity No. 3?

Determine how much solid solute dissolves completely in a given volume of water; Describe the appearance of a saturated solution. 2. Give one objective of the activity.

sugar 3. What was the solid solute used in the activity?

50 m. L 4. How much water was placed on the container before the start of the activity?

At least 3 times 5. How many times should an experiment be repeated to be sure of the result of the experiment?

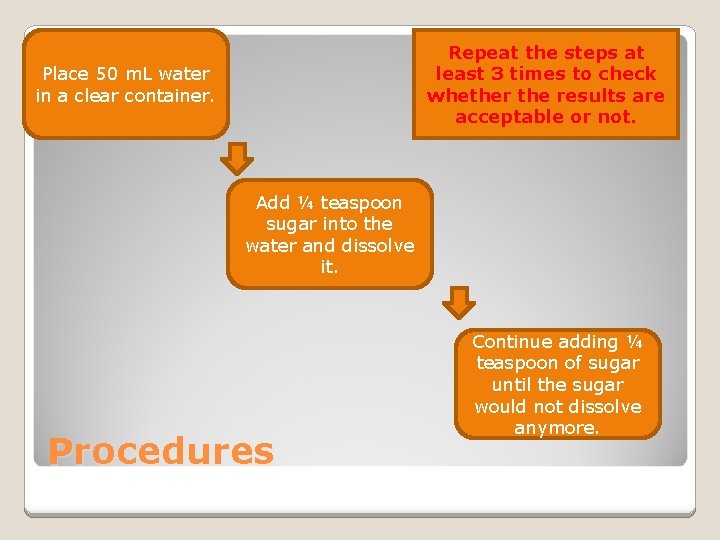
Repeat the steps at least 3 times to check whether the results are acceptable or not. Place 50 m. L water in a clear container. Add ¼ teaspoon sugar into the water and dissolve it. Procedures Continue adding ¼ teaspoon of sugar until the sugar would not dissolve anymore.

Olga is tasked by her groupmates to determine the mass of a 100 -gram metal. Upon using the triple beam balance, she got the following measurements after three trials: 95. 12 grams, 95. 20 grams, and 94. 96 grams. Is the triple beam balance precise or accurate in its measurements? Accurate or Precise?

Accurate – measurements are compared against a reference or accepted value. Precise – refers to how close the measurements to each other. Critical points

Olga is tasked by her groupmates to determine the mass of a 100 -gram metal. Upon using the triple beam balance, she got the following measurements after three trials: 95. 12 grams, 95. 20 grams, and 94. 96 grams. Is the triple beam balance precise or accurate in its measurements? Going back… Accurate? Precise?

Which is more important in experiments? Accuracy of precision?

After calibrating the triple beam balance, Olga measured again the 100 -gram metal and got the following measurements: 100. 1 grams, 100. 0 grams, 99. 99 grams. Are the data accurate or precise? Going back…

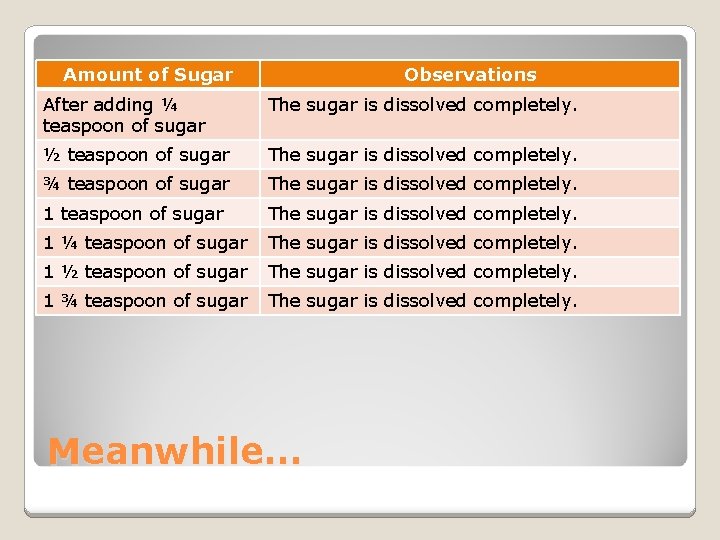
Amount of Sugar Observations After adding ¼ teaspoon of sugar The sugar is dissolved completely. ½ teaspoon of sugar The sugar is dissolved completely. ¾ teaspoon of sugar The sugar is dissolved completely. 1 ¼ teaspoon of sugar The sugar is dissolved completely. 1 ½ teaspoon of sugar The sugar is dissolved completely. 1 ¾ teaspoon of sugar The sugar is dissolved completely. Meanwhile…

Amount of Sugar Observations 4 teaspoons of sugar The sugar is dissolved completely. 4 ¼ teaspoons of sugar The sugar is dissolved completely. 4 ½ teaspoons of sugar The sugar is dissolved completely. 4 ¾ teaspoons of sugar The sugar is dissolved completely. 5 ¼ teaspoons of sugar The sugar is dissolved completely. 5 ½ teaspoons of sugar Sugar doesn’t dissolve anymore. After adding more…

There is a MAXIMUM AMOUNT of solute that can dissolve in a given amount of solvent at a given temperature. Why wouldn’t the sugar dissolve in water?

Amount of Sugar Observations 4 teaspoons of sugar The sugar is dissolved completely. 4 ¼ teaspoons of sugar The sugar is dissolved completely. 4 ½ teaspoons of sugar The sugar is dissolved completely. 4 ¾ teaspoons of sugar The sugar is dissolved completely. 5 ¼ teaspoons of sugar The sugar is dissolved completely. 5 ½ teaspoons of sugar Sugar doesn’t dissolve anymore. What is the maximum amount of sugar that can dissolve in 50 m. L water at room temperature?

When the solvent contains the MAXIMUM AMOUNT of solute that it can dissolve, it is a called SATURATED SOLUTION.

A SATURATED SOLUTION is a solution that contains the MAXIMUM AMOUNT of solute that the solvent can dissolve. What is a SATURATED SOLUTION?

Amount of Sugar 4 teaspoons of sugar Observations The sugar is dissolved completely. What happens when you add more solute to a. Thesaturated solution? 4 ½ teaspoons of sugar is dissolved completely. 4 ¼ teaspoons of sugar 4 ¾ teaspoons of sugar The sugar is dissolved completely. 5 ¼ teaspoons of sugar The sugar is dissolved completely. 5 ½ teaspoons of sugar Sugar doesn’t dissolve anymore. How would you know that a solution is saturated?

The presence of excess solute which can no longer dissolve is an evidence that a solution is saturated. What is the evidence that a solution is saturated?

SATURATED SOLUTION What is an UNSATURATED SOLUTION?

A solution is UNSATURATED when it contains LESS SOLUTE than the MAXIMUM AMOUNT it can dissolve in a given temperature.

A SATURATED SOLUTION is a solution that contains the MAXIMUM AMOUNT of solute that the solvent can dissolve. The presence of excess solute which can no longer dissolve is an evidence that a solution is saturated. solution is UNSATURATED when it contains LESS SOLUTE than the MAXIMUM AMOUNT it can dissolve in a given temperature. A Summing up…

50 grams of salt in 20 m. L water 50 grams of sugar in 20 m. L water How would you know which of these solutions is SATURATED?

The MAXIMUM AMOUNT of solute that can dissolve in a given volume of solvent at a given temperature is called the SOLUBILITY OF THE SOLUTE. Moving forward…

The solubility of salt at 250 C is 36 grams in 100 m. L of water. The MAXIMUM AMOUNT of solute that can dissolve in a given volume of solvent at a given temperature is called the SOLUBILITY OF THE SOLUTE.

The solubility of salt at 250 C is 36 grams in 100 m. L of water. 1. 2. 3. 4. 5. 15 30 36 60 72 grams grams of of of salt salt in in in 100 100 200 m. L m. L of of of water water Describe the following solutions.

Ricky was trying to determine the solubility of salt in 100 m. L of water. He did three trials and got the following results: First Trial Second Trial Third Trial 35. 83 grams 36. 24 grams 36. 07 grams Accurate or Precise?

A group of students were able to gather the following data and observations after dissolving sugar in 100 m. L of water. Step Amount of Solute added Observations 1 1 teaspoon After stirring, sugar dissolves completely. 2 1 teaspoon After stirring, sugar dissolves completely. 3 1 teaspoon After stirring, sugar dissolves completely. 4 1 teaspoon After stirring, sugar dissolves completely. 5 1 teaspoon After stirring, some sugar is seen at the bottom of the container

An unsaturated solution is a solution that contains the maximum amount of solute that can be dissolved. 2. The presence of excess solute that can no longer be dissolved is an evidence that the solution is saturated. 3. The maximum amount of solute that can dissolve in a given volume of solvent at a given temperature is called the solubility of the solvent. 1. True or False?

Please do advance reading of our lessons through your text. Chapter 5 (Solutions) Checking of textbooks on Monday. Ensure that you have your books on that date. Bring a calculator on the next session. It DOESN’T have to be a scientific calculator. Reminders!