What is Solubility The maximum amount of a

What is Solubility? • The maximum amount of a substance that will dissolve in a certain amount of solvent at a given temperature is called that substances solubility in that solvent • Solute = is dissolved • Solvent = does the dissolving • Example: __ grams of salt (Na. Cl) in 100 g of water at __ ˚C.

Temperature and Solubility • The temperature of the solution affects how much of the solute is dissolved by the solvent. • Increasing the temperature does not always increase the solubility. • Solubility curves are used to show the solubility of a substance changes with temperature.
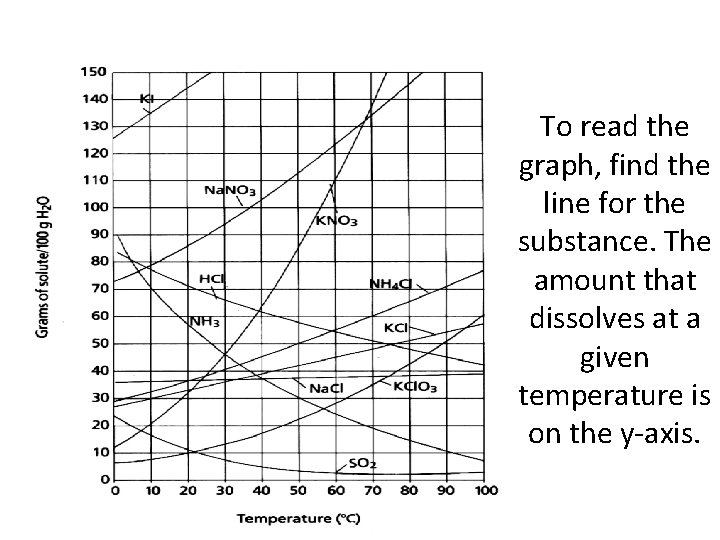
To read the graph, find the line for the substance. The amount that dissolves at a given temperature is on the y-axis.
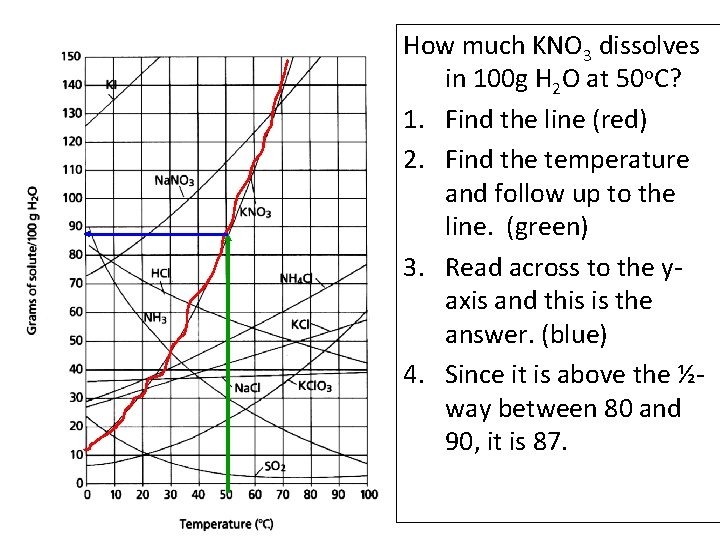
How much KNO 3 dissolves in 100 g H 2 O at 50 o. C? 1. Find the line (red) 2. Find the temperature and follow up to the line. (green) 3. Read across to the yaxis and this is the answer. (blue) 4. Since it is above the ½way between 80 and 90, it is 87.

What kind of solution do you have? • A point on the line is a saturated solution. • Above the line is supersaturated. • Below the line is unsaturated.

Your turn! Use your graph 1. How much Na. Cl will dissolve in 100 g H 2 O at 35 o. C? 2. How much NH 4 Cl will dissolve at 50 o. C? 3. What is the solubility of SO 2 at 25 o. C? 4. What is the solubility of KCl. O 3 in 100 grams of water at 35 o. C? 5. What is the solubility of KI in 100 grams of water at 10 o. C?
- Slides: 6