What is Physical Science Natural Sciences along with
























- Slides: 32

What is Physical Science? • Natural Sciences along with Biological Sciences • Physical Sciences = Physics, Chemistry, Geology, Meteorology, and Astronomy Section 1. 1

This Course • Physics & Chemistry – Matter & Energy • Attempt to describe the physical world in which we live • Measurements – movement, temperature, weather conditions, time, etc. Section 1. 1

Experiment • Experiment – observation of natural phenomena that can be – – Carried out in a controlled manner – Results can be duplicated – Can be done by other researchers Section 1. 2

Scientific Method • Scientific Method - the process of experimentation and explanation • Question • Hypothesis • Procedure • Test • Analyze • Start over Section 1. 2

Hypothesis • Hypothesis – tentative explanation (best guess) • Example: Matter consists of small particles (atoms) that simply rearrange themselves • A good hypothesis can be supported by previous experiences • It must suggest new experiments that serve to test its validity. Section 1. 2

Theory • Theory – tested explanation (Several Tests prove it is valid) • Example: Atomic Theory – This theory has withstood testing for 200+ years and continues to correctly predict atomic behavior. • Evolutionary Theory – is it really a theory?

Scientific Law • Scientific Law – Mathematical statement after a series of experiments (D=m/v, e=mc 2) • Example – Law of Conservation of Mass (no gain or loss during chemical reaction) • The law simply states the finding, but does not explain the behavior. Section 1. 2

Hypothesis, Theory or Law? 1. The sun will die in 7. 5 billion years. – Hypothesis 2. Light is an electromagnetic phenomenon described by Maxwell’s Laws. – Theory 3. The sun will rise tomorrow. – Hypothesis 4. For every action, there is an equal and opposite reaction – law 5. Earth’s magnetic field is generated by a conduction fluid in its core. – Theory 6. Momentum is the product of a bodies mass and its velocity. – Law 7. Space exits in 10 -dimensions not just 3. – Hypothesis

The Senses • Sight, Hearing, Smell, Taste, Touch • Sight and Hearing provide the most information to our brains about our environment. • Sensory Limitations – can be reduced by using measuring devices (Significant Figures) • Instruments extend our ability to measure and learn about our environment. • Our senses can also be deceived Section 1. 3

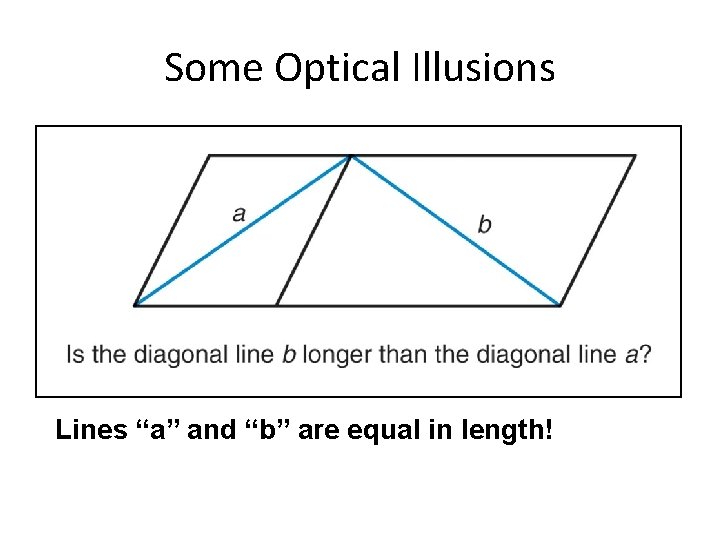
Some Optical Illusions Lines “a” and “b” are equal in length! Section 1. 3

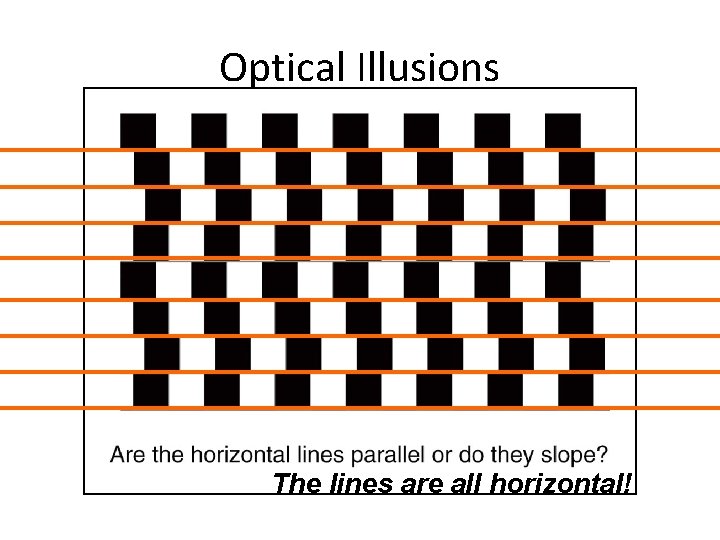
Optical Illusions The lines are all horizontal! Section 1. 3

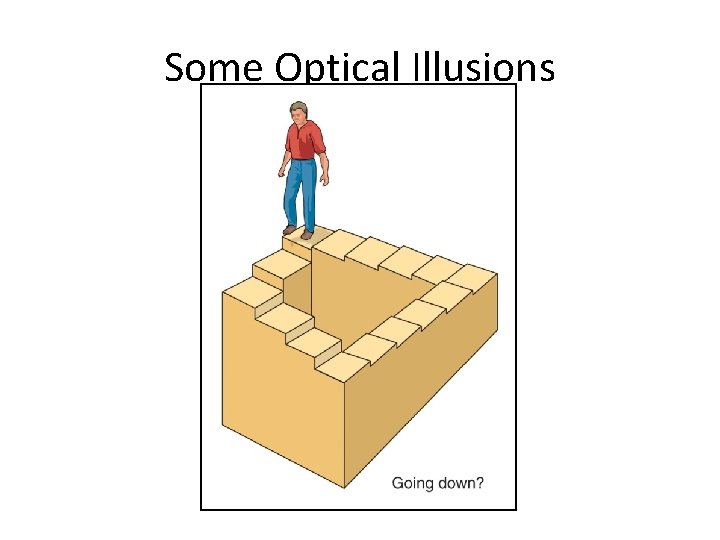
Some Optical Illusions Section 1. 3

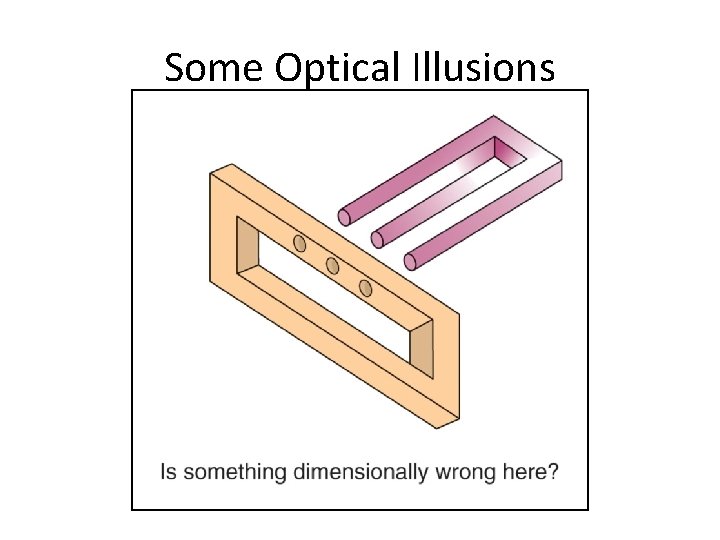
Some Optical Illusions Section 1. 3

Standard Units and Systems of Units • Two major systems of units • British (English) system – only used widely in the United States (miles, inches, pounds, seconds, etc. ) • Metric system – used throughout most of the world (kilometers, grams, etc. ) • The U. S. “officially” adopted the metric system in 1893, but continues to use the British system. Section 1. 4

A Brief, Chronological History of the Metric System

Length • The measurement of space in any direction • Space has three dimensions – length, width, and height. Section 1. 4

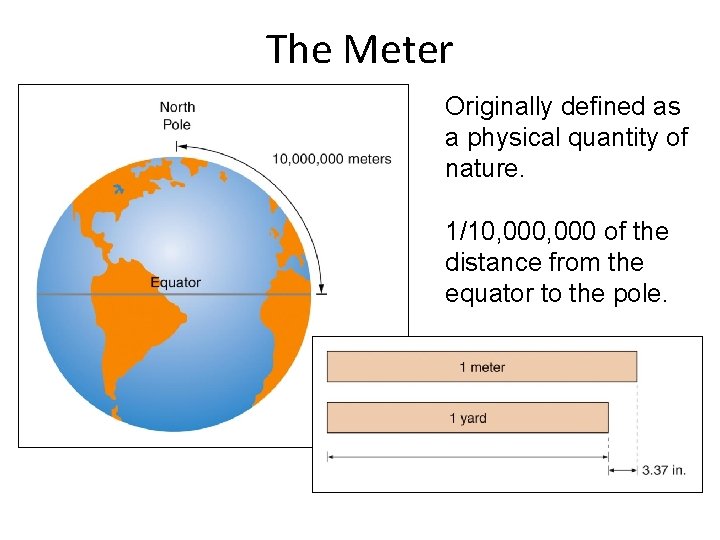
The Meter Originally defined as a physical quantity of nature. 1/10, 000 of the distance from the equator to the pole. Section 1. 4

About how high is this road in feet? Section 1. 6

Mass (metric) • The amount of matter an object contains • An object’s mass is always constant • Mass is a fundamental unit that will remain constant throughout the universe. • Metric Standard Unit = Kilogram (kg) – originally defined as the amount of water in a 0. 1 m cube. Now referenced to a cylinder in Paris Section 1. 4

Kilogram Standard U. S. Prototype #20 Kilogram, at NIST in Washington, D. C. Actually – 0. 999 961 kg of “the” standard in Paris Section 1. 4

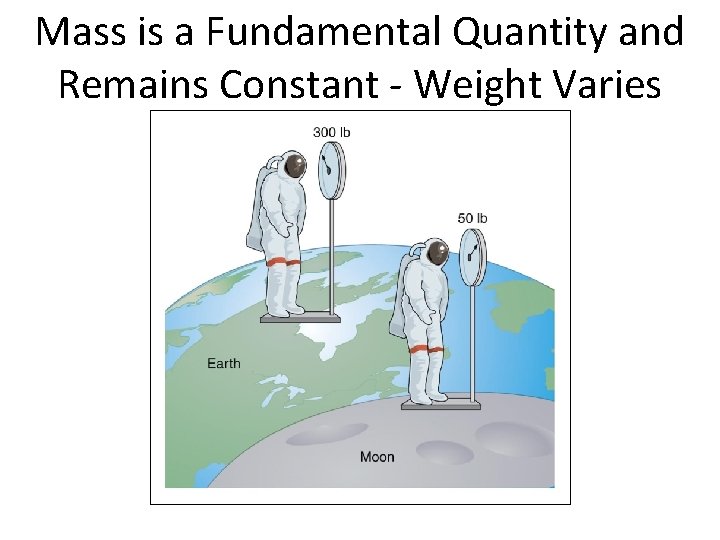
Mass (British) • The pound is actually not a unit of mass, but rather of weight, related to gravitational attraction (depends on where the object is!) • Object: Earth = 1 lb. Moon = 1/6 lb. • Weight depends on gravity • In fact, the weight of an object will vary slightly depending on where it is on earth (higher altitude less weight) Section 1. 4

Mass is a Fundamental Quantity and Remains Constant - Weight Varies Section 1. 4

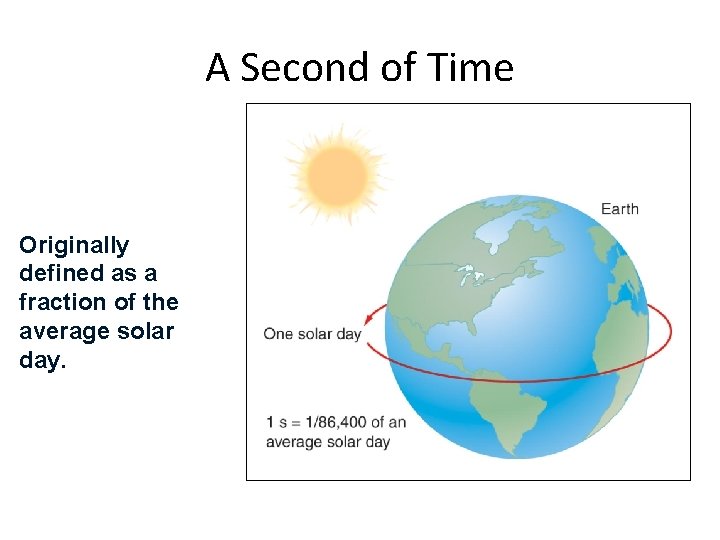
Time • Time - the continuous, forward flowing of events • Time has only one direction forward • Second (s) – the standard unit in both the metric and British systems • Originally 1/86, 400 of a solar day • Now based on the vibration of the Cs 133 atom (Atomic Clock) Section 1. 4

A Second of Time Originally defined as a fraction of the average solar day. Section 1. 4

Ch 1. 2 Metric Prefixes, Distance, Volume, Mass, Density, & Temperature

Metric System • Uses acronym “mks system” from standard units of length, mass, and time – meter, kilogram, second • It is a decimal (base-10) system – this is much better than the British system • Contains seven base units Section 1. 5

Base-10 Convenient • Easy expression and conversion • Metric examples vs. British examples – 1 kilometer = 1000 meters – 1 mile = 5280 feet – 1 meter = 100 centimeters – 1 yard = 3 feet or 36 inches – 1 liter = 1000 milliliters – 1 quart = 32 ounces or 2 pints – 1 gallon = 128 ounces Section 1. 5

Commonly Used Prefixes • • • Mega, M – 106 – 1, 000 times the base Kilo, k – 103 – 1, 000 times the base Base Unit Centi, c – 10 -2 – 1/100 th of the base Milli, m – 10 -3 – 1/1000 th of the base

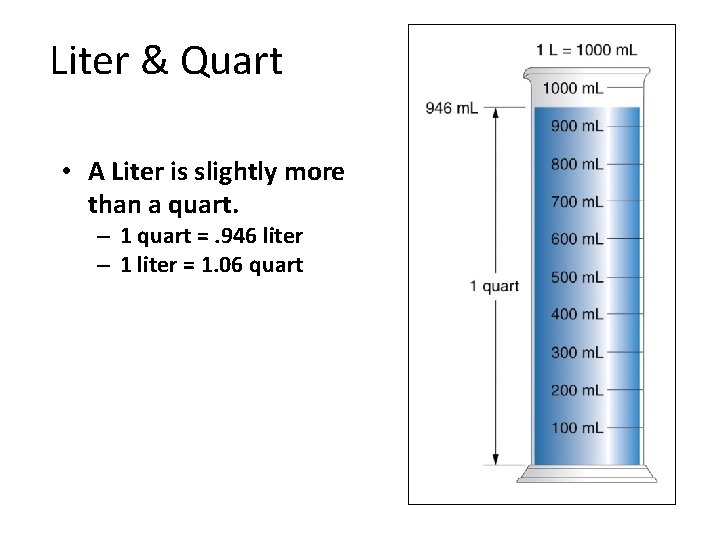
Liter & Quart • A Liter is slightly more than a quart. – 1 quart =. 946 liter – 1 liter = 1. 06 quart Section 1. 5

Temperature Three different scales are in common use • Fahrenheit – Boiling Point of water 212°F – Freezing Point of water 32 °F • Celsius – Boiling Point of water 100 °C – Freezing Point of water 0 °C

Temperature Kelvin – Absolute temperature scale. • Kelvin and Celsius both have the same divisions. • Freezing point of water is 273. 15 K • Boiling point of water is 373. 15 K • All matter stops at 0 K – We use this scale to calculate problems comparing temperature changes & matter motion. – EX: a change from 1 °C to 2 °C should say that the particle motion doubles. It does not 274 K to 275 K correctly identifies the small change in particle motion

Ch 1. 3 Graphs – very important in science you need to know this yourself! Graphing will be covered at various times throughout the semester. When in doubt about graphing look back at this section!