What Is HPLC Basic Principles LAAQBLC 001 B

What Is HPLC? Basic Principles LAAQ-B-LC 001 B 1
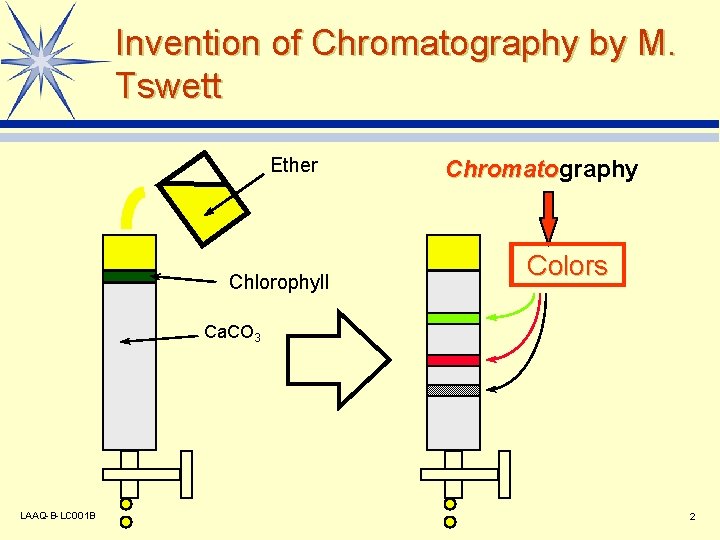
Invention of Chromatography by M. Tswett Ether Chlorophyll Chromatography Chromato Colors Ca. CO 3 LAAQ-B-LC 001 B 2
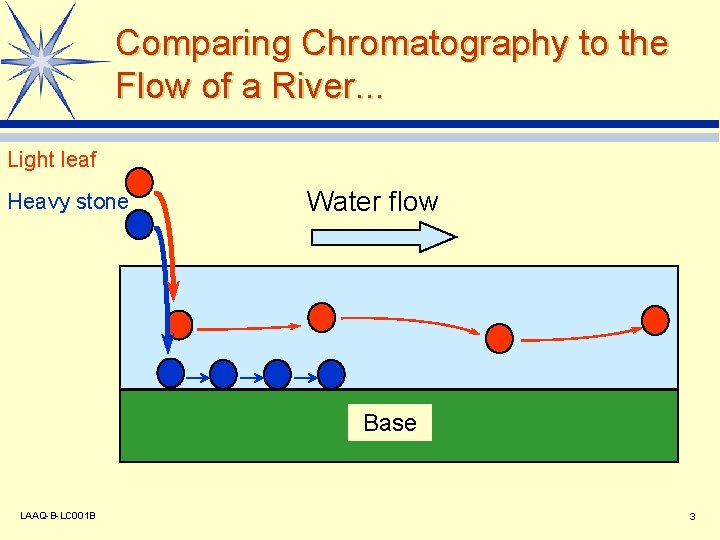
Comparing Chromatography to the Flow of a River. . . Light leaf Heavy stone Water flow Base LAAQ-B-LC 001 B 3
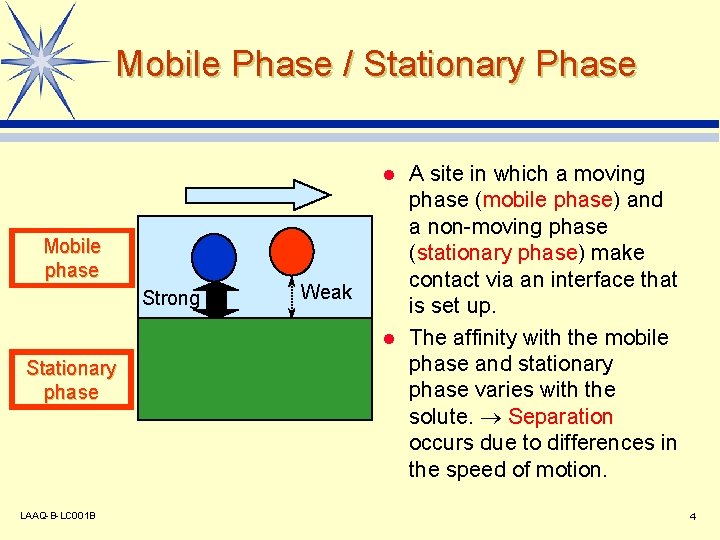
Mobile Phase / Stationary Phase A site in which a moving phase (mobile phase) and a non-moving phase (stationary phase) make contact via an interface that is set up. l The affinity with the mobile phase and stationary phase varies with the solute. Separation occurs due to differences in the speed of motion. l Mobile phase Strong Stationary phase LAAQ-B-LC 001 B Weak 4

Chromato-graphy / -graph / -gram / -grapher Chromatography: l Chromatograph: l Chromatogram: l Chromatographer: l LAAQ-B-LC 001 B Analytical technique Instrument Obtained “picture” Person 5
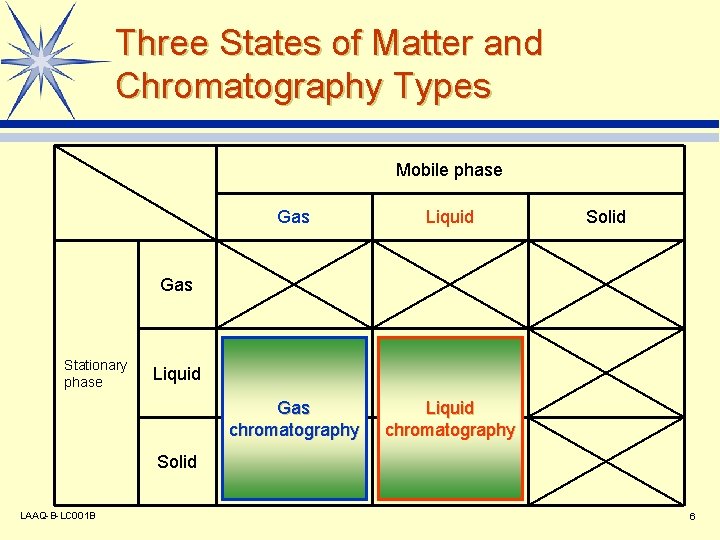
Three States of Matter and Chromatography Types Mobile phase Gas Liquid Gas chromatography Liquid chromatography Solid Gas Stationary phase Liquid Solid LAAQ-B-LC 001 B 6

Liquid Chromatography l Chromatography in which the mobile phase is a liquid. v The liquid used as the mobile phase is called the “eluent”. The stationary phase is usually a solid or a liquid. l In general, it is possible to analyze any substance that can be stably dissolved in the mobile phase. l LAAQ-B-LC 001 B 7
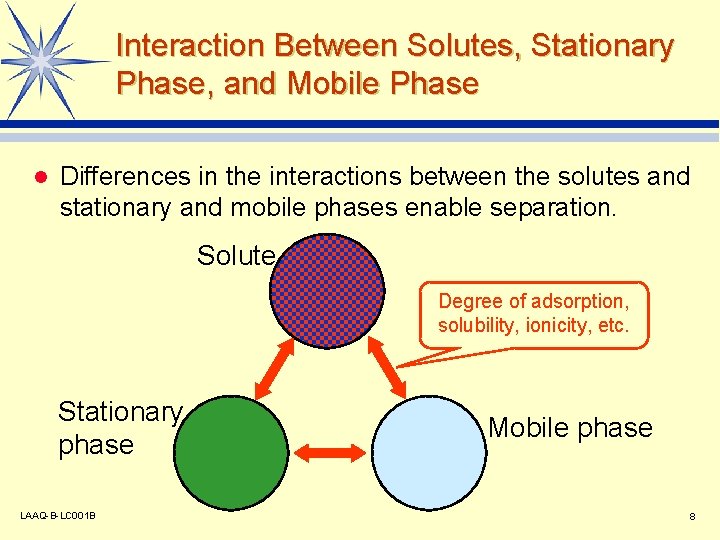
Interaction Between Solutes, Stationary Phase, and Mobile Phase l Differences in the interactions between the solutes and stationary and mobile phases enable separation. Solute Degree of adsorption, solubility, ionicity, etc. Stationary phase LAAQ-B-LC 001 B Mobile phase 8
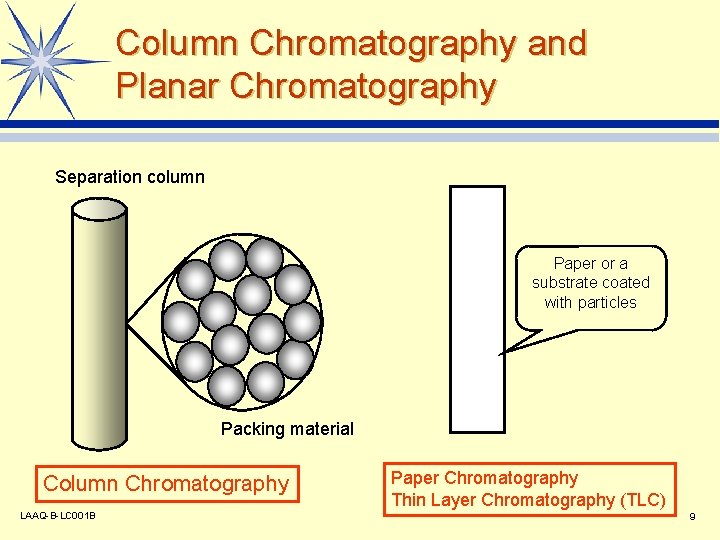
Column Chromatography and Planar Chromatography Separation column Paper or a substrate coated with particles Packing material Column Chromatography LAAQ-B-LC 001 B Paper Chromatography Thin Layer Chromatography (TLC) 9
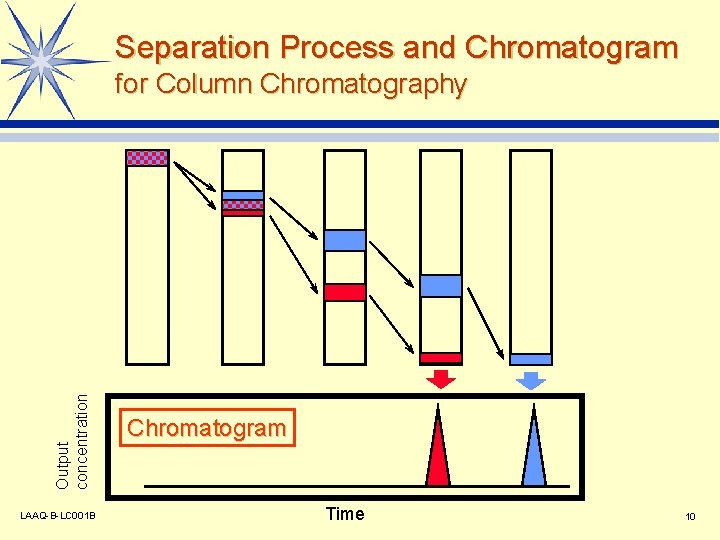
Separation Process and Chromatogram Output concentration for Column Chromatography LAAQ-B-LC 001 B Chromatogram Time 10
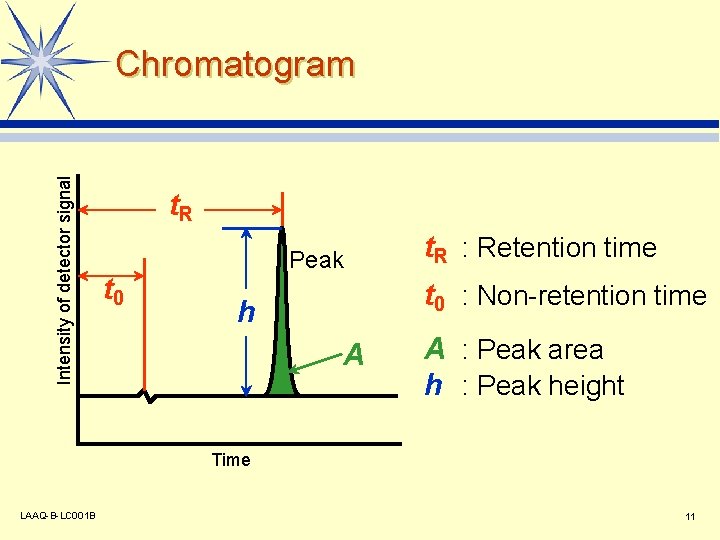
Intensity of detector signal Chromatogram t. R t 0 Peak t. R : Retention time t 0 : Non-retention time h A A : Peak area h : Peak height Time LAAQ-B-LC 001 B 11

From Liquid Chromatography to High Performance Liquid Chromatography Higher degree of separation! Refinement of packing material (3 to 10 µm) l Reduction of analysis time! Delivery of eluent by pump Demand for special equipment that can withstand high pressures l The arrival of high performance liquid chromatography! LAAQ-B-LC 001 B 12
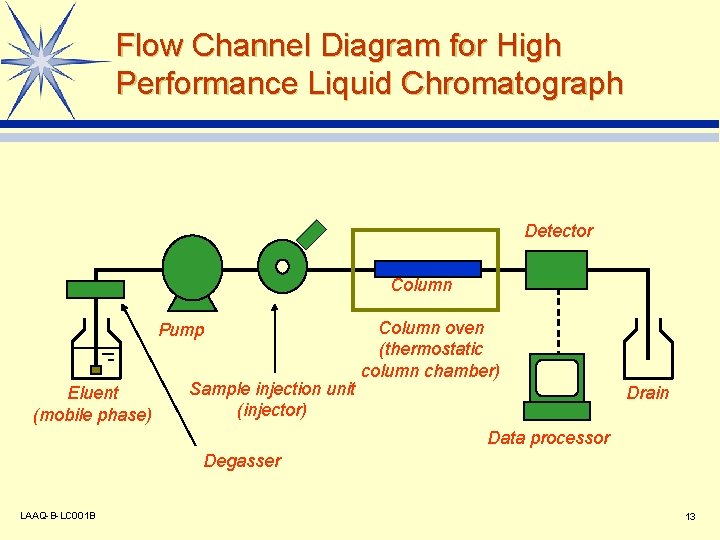
Flow Channel Diagram for High Performance Liquid Chromatograph Detector Column Pump Eluent (mobile phase) Sample injection unit (injector) Column oven (thermostatic column chamber) Drain Data processor Degasser LAAQ-B-LC 001 B 13

Advantages of High Performance Liquid Chromatography High separation capacity, enabling the batch analysis of multiple components l Superior quantitative capability and reproducibility l Moderate analytical conditions l v Unlike GC, the sample does not need to be vaporized. Generally high sensitivity l Low sample consumption l Easy preparative separation and purification of samples l LAAQ-B-LC 001 B 14

Fields in Which High Performance Liquid Chromatography Is Used l Biogenic substances v l Sugars, lipids, nucleic acids, amino acids, proteins, peptides, steroids, amines, etc. l v l Medical products v Food products Environmental samples v v Drugs, antibiotics, etc. l Inorganic ions Hazardous organic substances, etc. Organic industrial products v LAAQ-B-LC 001 B Vitamins, food additives, sugars, organic acids, amino acids, etc. Synthetic polymers, additives, surfactants, etc. 15

HPLC Hardware: Part 1 Solvent Delivery System, Degasser, Sample Injection Unit, Column Oven LAAQ-B-LC 001 B 16
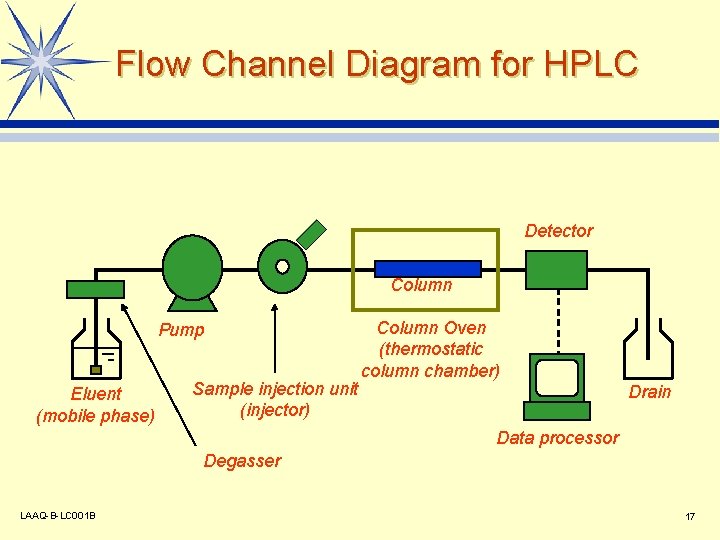
Flow Channel Diagram for HPLC Detector Column Pump Eluent (mobile phase) Sample injection unit (injector) Column Oven (thermostatic column chamber) Drain Data processor Degasser LAAQ-B-LC 001 B 17

Solvent Delivery Pump l Performance Requirements v Capacity to withstand high load pressures. v Pulsations that accompany pressure fluctuations are small. v Flow rate does not fluctuate. v Solvent replacement is easy. v The flow rate setting range is wide and the flow rate is accurate. LAAQ-B-LC 001 B 18

Solvent Delivery Pump: Representative Pumping Methods l Syringe pump l Plunger pump l Diaphragm pump LAAQ-B-LC 001 B 19
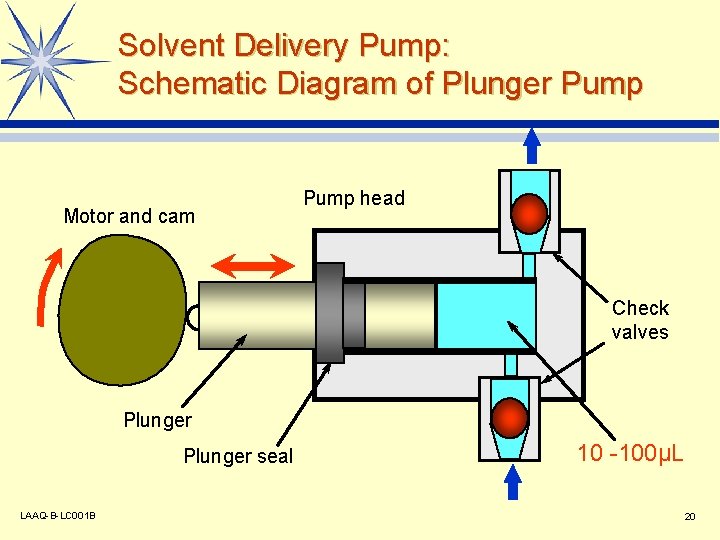
Solvent Delivery Pump: Schematic Diagram of Plunger Pump Motor and cam Pump head Check valves Plunger seal LAAQ-B-LC 001 B 10 -100µL 20
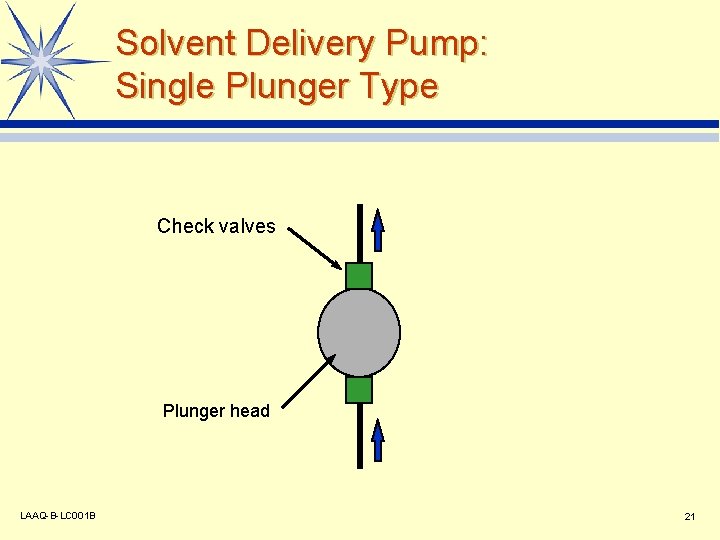
Solvent Delivery Pump: Single Plunger Type Check valves Plunger head LAAQ-B-LC 001 B 21
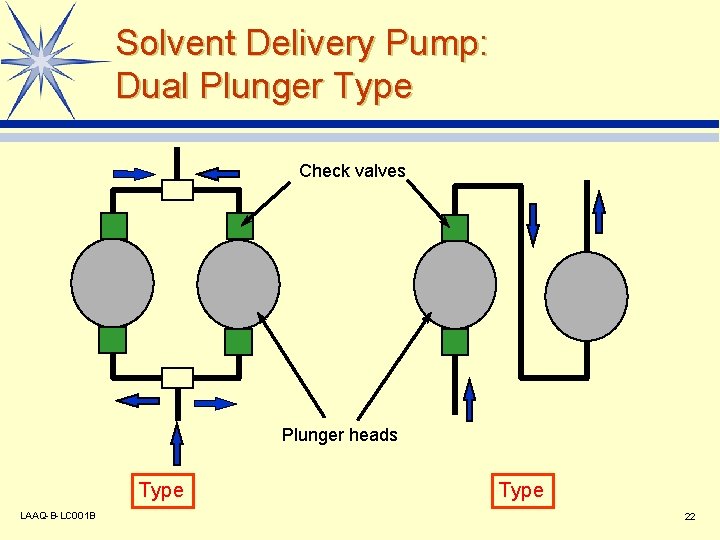
Solvent Delivery Pump: Dual Plunger Type Check valves Plunger heads Type LAAQ-B-LC 001 B Type 22

Gradient System l Isocratic system v Constant l Gradient v Varying eluent composition system eluent composition HPGE (High Pressure Gradient) LPGE (Low Pressure Gradient) LAAQ-B-LC 001 B 23
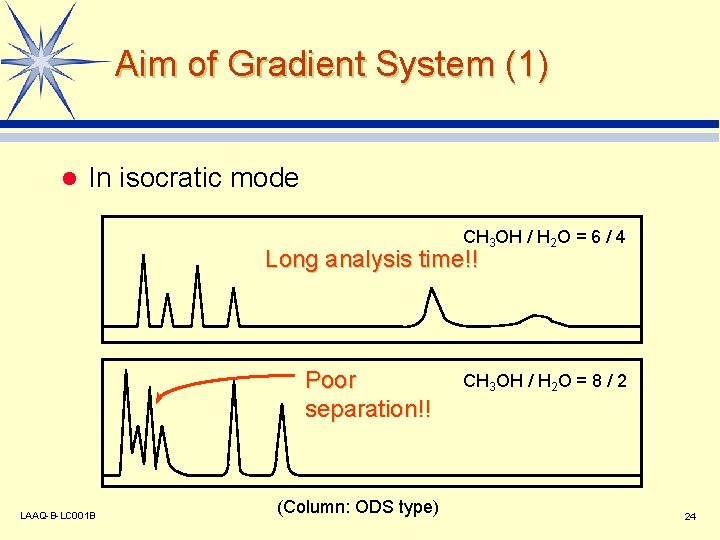
Aim of Gradient System (1) l In isocratic mode CH 3 OH / H 2 O = 6 / 4 Long analysis time!! Poor separation!! LAAQ-B-LC 001 B (Column: ODS type) CH 3 OH / H 2 O = 8 / 2 24
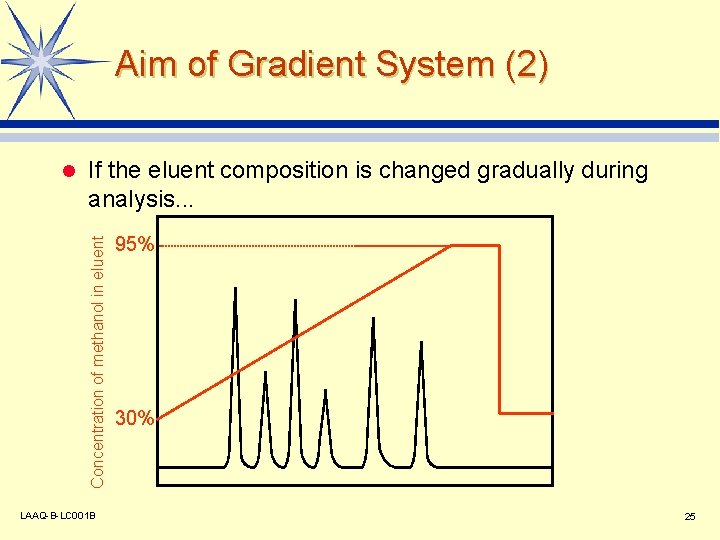
Aim of Gradient System (2) If the eluent composition is changed gradually during analysis. . . Concentration of methanol in eluent l LAAQ-B-LC 001 B 95% 30% 25
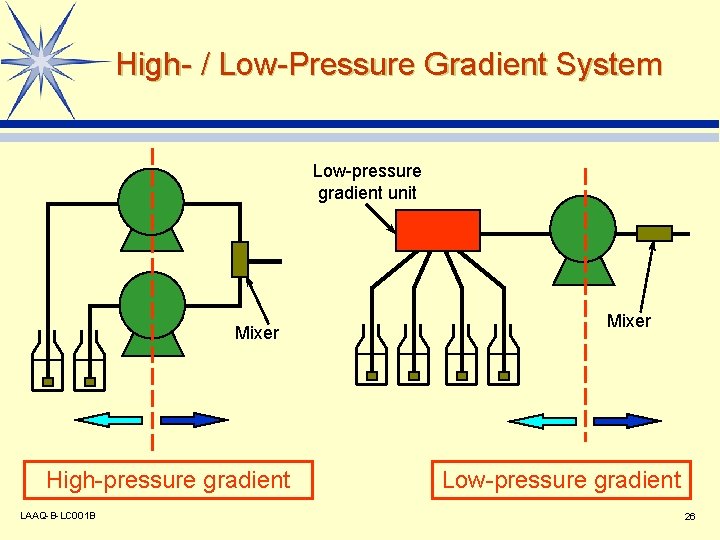
High- / Low-Pressure Gradient System Low-pressure gradient unit Mixer High-pressure gradient LAAQ-B-LC 001 B Mixer Low-pressure gradient 26

Advantages and Disadvantages of High- / Low-Pressure Gradient Systems l High-pressure gradient system v High gradient accuracy v Complex system configuration (multiple pumps required) l Low-pressure gradient system v Simple system configuration v Degasser required LAAQ-B-LC 001 B 27

Degasser l Problems caused by dissolved air in the eluent Unstable delivery by pump v More noise and large baseline drift in detector cell v In order to avoid these problems, the eluent must be degassed. LAAQ-B-LC 001 B 28
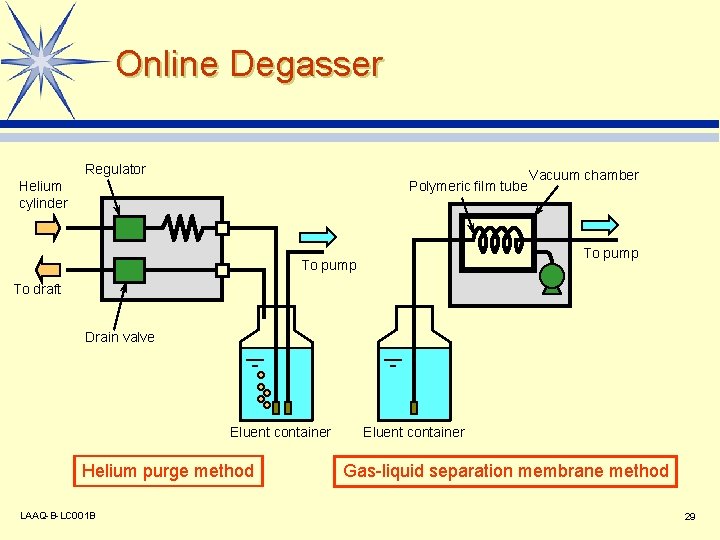
Online Degasser Regulator Helium cylinder Polymeric film tube Vacuum chamber To pump To draft Drain valve Eluent container Helium purge method LAAQ-B-LC 001 B Eluent container Gas-liquid separation membrane method 29

Sample Injection Unit (Injector) l Performance Requirements v No sample remaining in unit v Minimal broadening of sample band v Free adjustment of injection volume v Minimal loss v Superior durability and pressure resistance LAAQ-B-LC 001 B 30
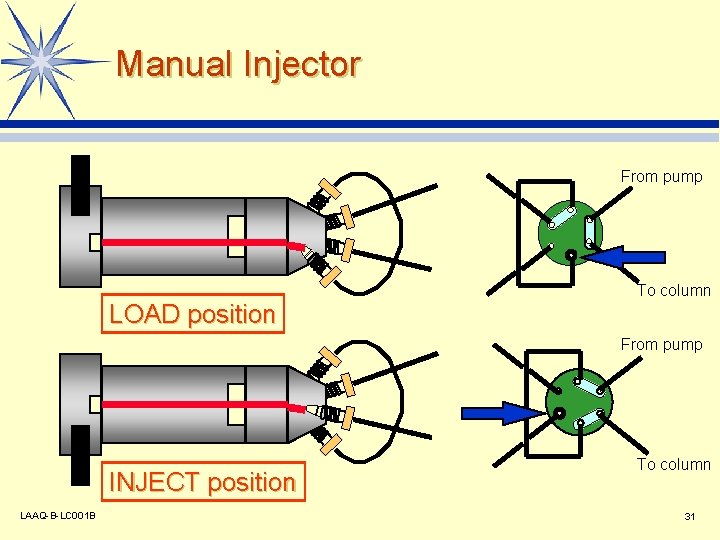
Manual Injector From pump LOAD position To column From pump INJECT position LAAQ-B-LC 001 B To column 31
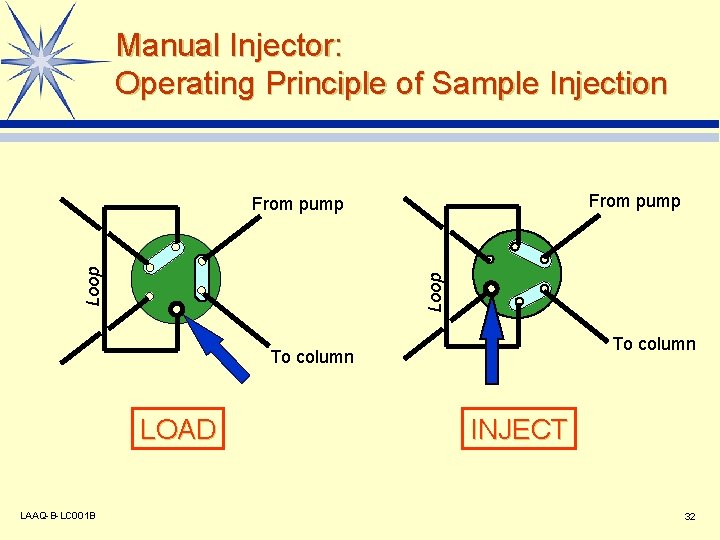
Manual Injector: Operating Principle of Sample Injection From pump Loop From pump To column LOAD LAAQ-B-LC 001 B INJECT 32

Manual Injector: Injection Method l Syringe measurement method v It is desirable that no more than half the loop volume is injected. l Loop measurement method v It is desirable that at least 3 times the loop volume is injected. LAAQ-B-LC 001 B 33
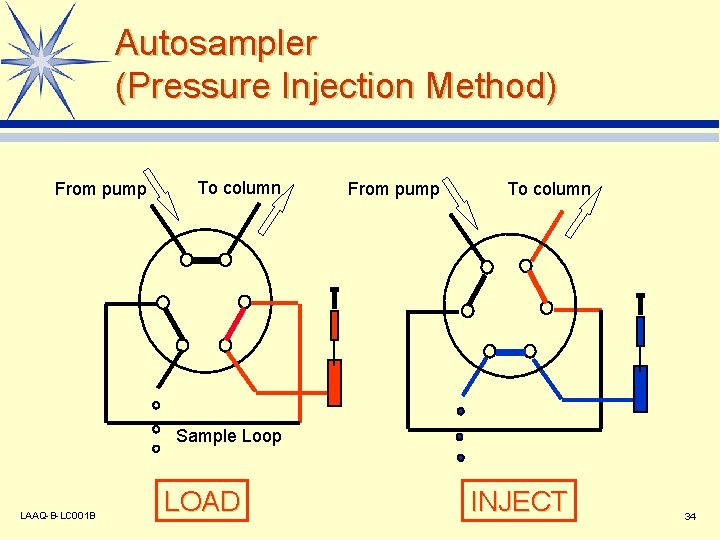
Autosampler (Pressure Injection Method) From pump To column Sample Loop LAAQ-B-LC 001 B LOAD INJECT 34
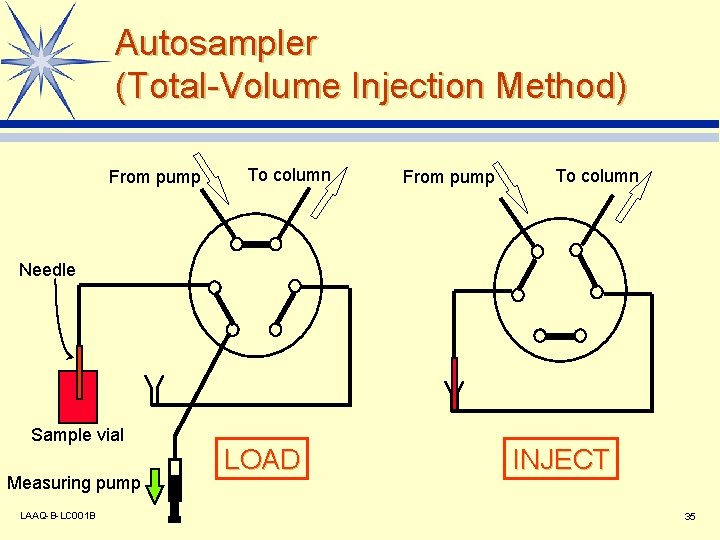
Autosampler (Total-Volume Injection Method) From pump To column Needle Sample vial Measuring pump LAAQ-B-LC 001 B LOAD INJECT 35

Column Oven l Air circulation heating type l Block heating type v Aluminum l Insulated v Water LAAQ-B-LC 001 B block heater column jacket type bath 36

Tubing and Preparation for Solvent Delivery Prior to Analysis LAAQ-B-LC 001 B 37

Tubing l Material l O. D. (outer diameter) Stainless steel (SUS) v 1. 6 mm v PEEK (polyether l I. D. (inner diameter) ketone) v 0. 1 mm v Fluororesin v 0. 3 mm v 0. 5 mm v 0. 8 mm etc. v LAAQ-B-LC 001 B 38
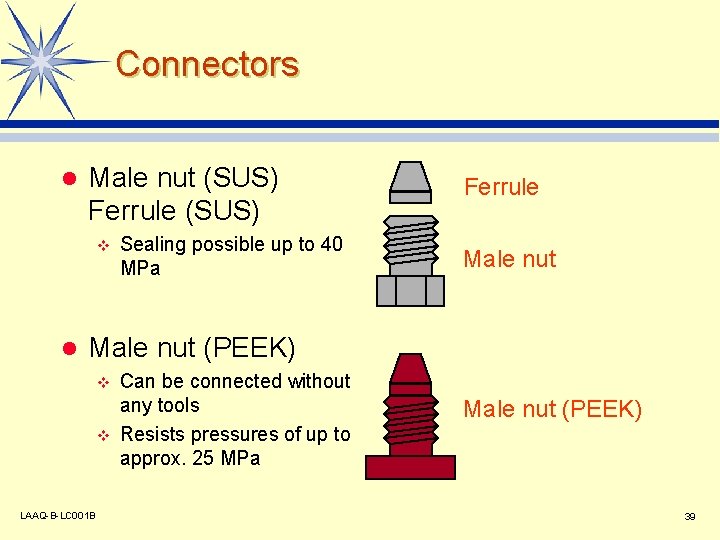
Connectors l Male nut (SUS) Ferrule (SUS) v l Sealing possible up to 40 MPa Ferrule Male nut (PEEK) v v LAAQ-B-LC 001 B Can be connected without any tools Resists pressures of up to approx. 25 MPa Male nut (PEEK) 39
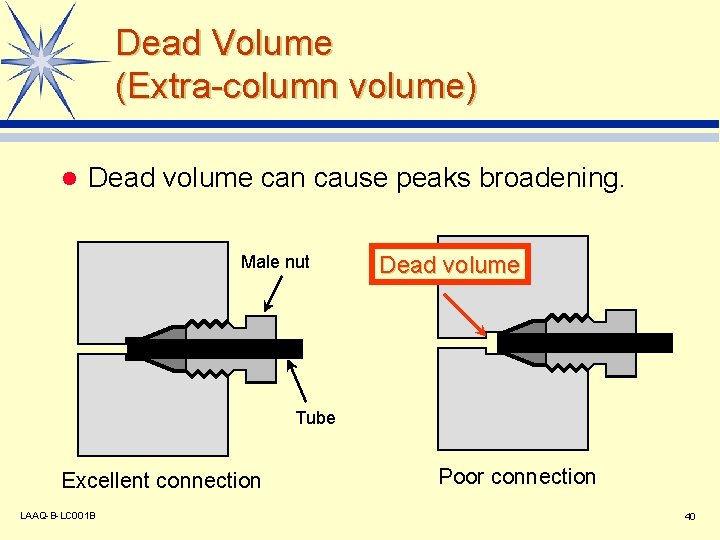
Dead Volume (Extra-column volume) l Dead volume can cause peaks broadening. Male nut Dead volume Tube Excellent connection LAAQ-B-LC 001 B Poor connection 40

Mobile Phase l Water v v “Ultrapure water” can be used with confidence. Commercial “distilled water for HPLC” is also acceptable. l Organic Solvent v v v LAAQ-B-LC 001 B HPLC-grade solvent can be used with confidence. Special-grade solvent is acceptable depending on the detection conditions. Care is required regarding solvents containing stabilizers (e. g. , tetrahydrofuran and chloroform) 41
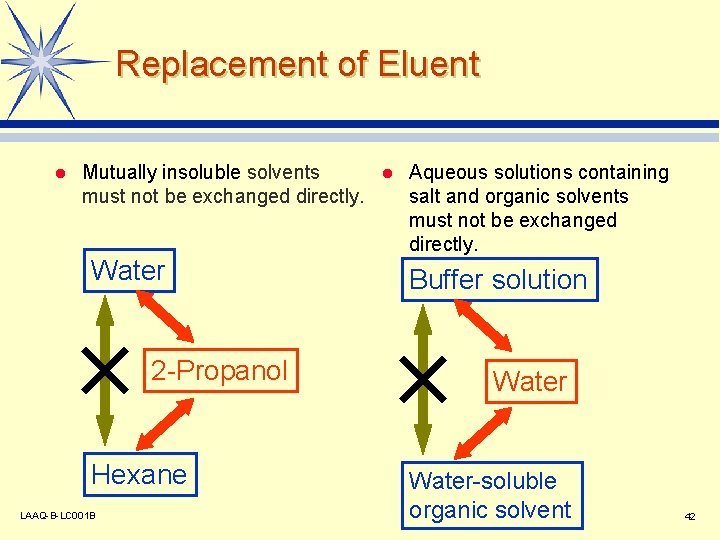
Replacement of Eluent l Mutually insoluble solvents l Aqueous solutions containing must not be exchanged directly. salt and organic solvents must not be exchanged directly. Water 2 -Propanol Hexane LAAQ-B-LC 001 B Buffer solution Water-soluble organic solvent 42
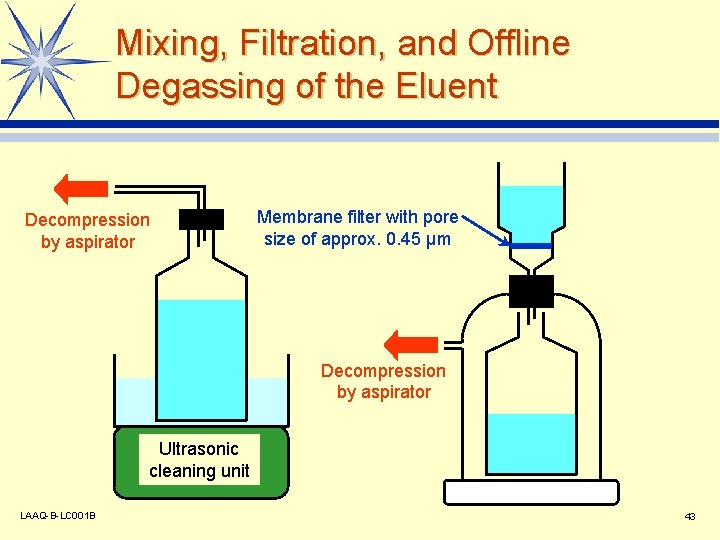
Mixing, Filtration, and Offline Degassing of the Eluent Decompression by aspirator Membrane filter with pore size of approx. 0. 45 µm Decompression by aspirator Ultrasonic cleaning unit LAAQ-B-LC 001 B 43

Reversed Phase Chromatography Part 1 Basic Principles LAAQ-B-LC 001 B 44
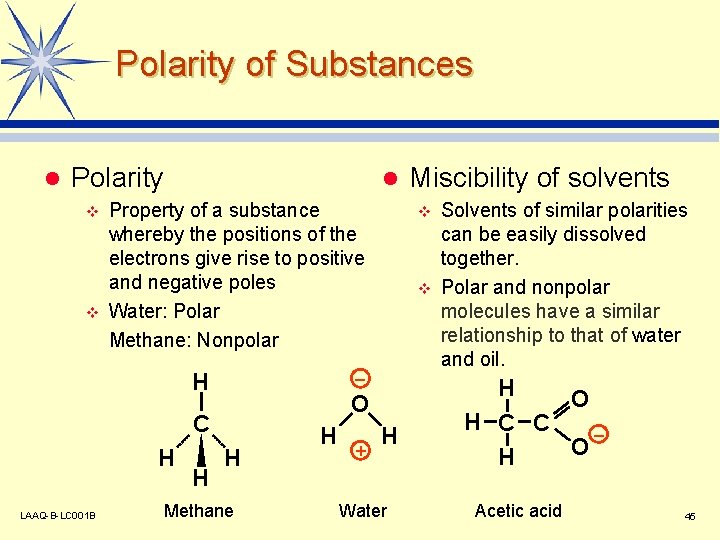
Polarity of Substances l Polarity v v l Property of a substance whereby the positions of the electrons give rise to positive and negative poles Water: Polar Methane: Nonpolar H H LAAQ-B-LC 001 B H v v – O C H Methane H + Miscibility of solvents H Water Solvents of similar polarities can be easily dissolved together. Polar and nonpolar molecules have a similar relationship to that of water and oil. H O H C C – O H Acetic acid 45

Nonpolar (Hydrophobic) Functional Groups and Polar (Hydrophilic) Functional Groups l Nonpolar Functional Groups v -(CH 2)n. CH 3 Alkyl v l Polar Functional Groups v groups -C 6 H 5 Phenyl -COOH Carboxyl v groups -NH 2 Amino v groups -OH Hydroxyl LAAQ-B-LC 001 B groups 46

Partition Chromatography l. A liquid (or a substance regarded as a liquid) is used as the stationary phase, and the solute is separated according to whether it dissolves more readily in the stationary or mobile phase. l Liquid-liquid chromatography LAAQ-B-LC 001 B 47
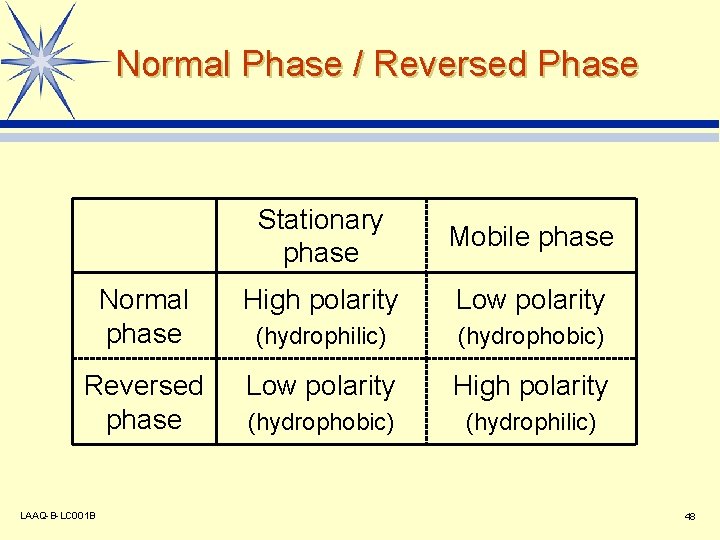
Normal Phase / Reversed Phase Stationary phase Mobile phase Normal phase High polarity Low polarity (hydrophilic) (hydrophobic) Reversed phase Low polarity High polarity (hydrophobic) (hydrophilic) LAAQ-B-LC 001 B 48

Reversed Phase Chromatography l Stationary phase: Low polarity v Octadecyl l Mobile group-bonded silical gel (ODS) phase: High polarity v Water, methanol, acetonitrile v Salt is sometimes added. LAAQ-B-LC 001 B 49
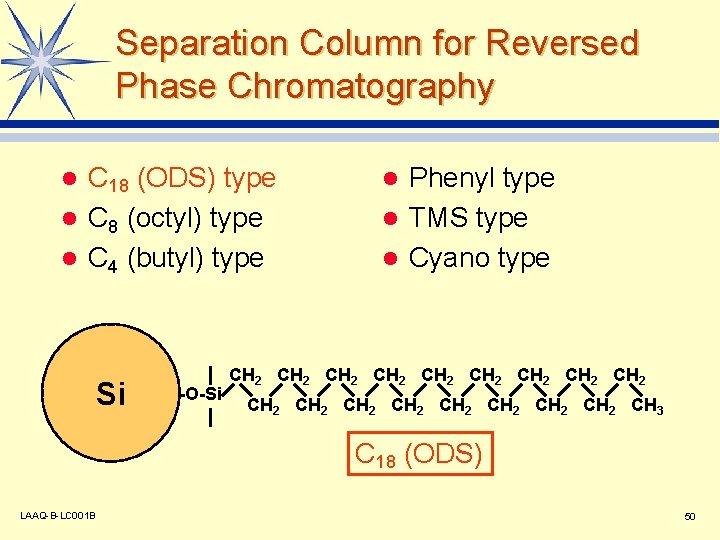
Separation Column for Reversed Phase Chromatography C 18 (ODS) type l C 8 (octyl) type l C 4 (butyl) type l Si -O-Si Phenyl type l TMS type l Cyano type l CH 2 CH 2 CH 2 CH 2 CH 2 CH 3 C 18 (ODS) LAAQ-B-LC 001 B 50
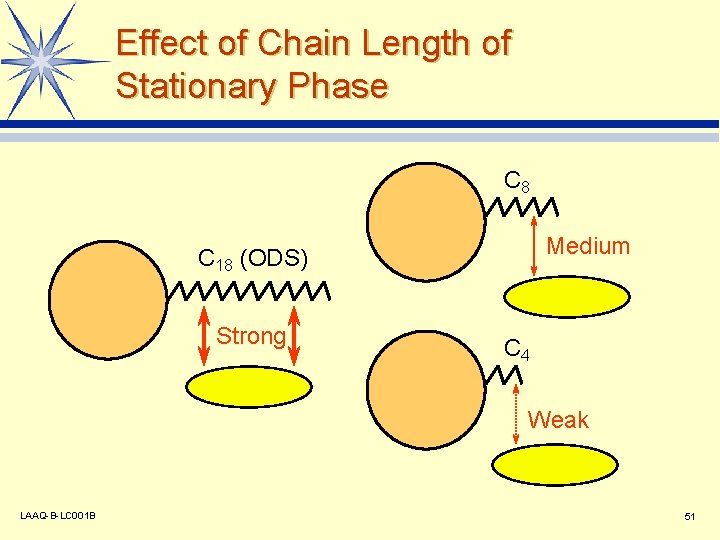
Effect of Chain Length of Stationary Phase C 8 Medium C 18 (ODS) Strong C 4 Weak LAAQ-B-LC 001 B 51
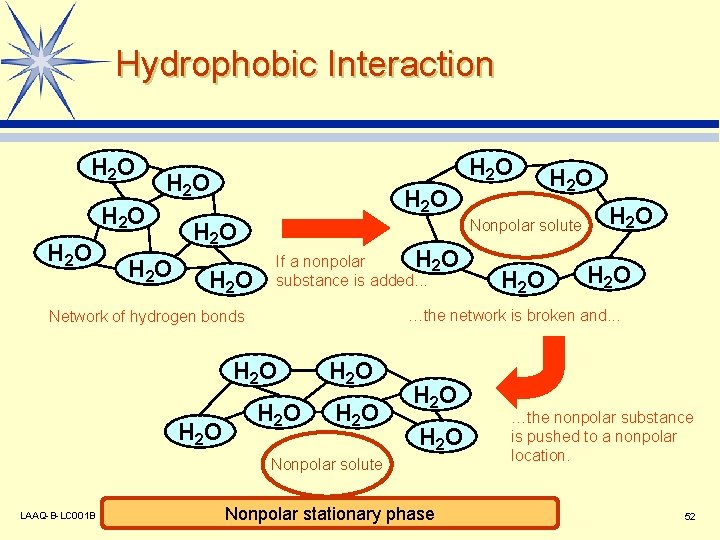
Hydrophobic Interaction H 2 O H 2 O H 2 O Nonpolar solute If a nonpolar H 2 O substance is added. . . H 2 O H 2 O Nonpolar solute LAAQ-B-LC 001 B H 2 O …the network is broken and. . . Network of hydrogen bonds H 2 O Nonpolar stationary phase …the nonpolar substance is pushed to a nonpolar location. 52
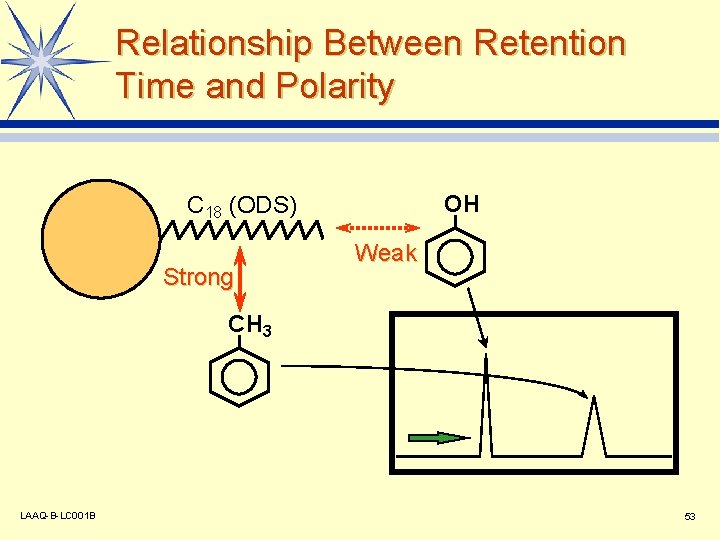
Relationship Between Retention Time and Polarity OH C 18 (ODS) Strong Weak CH 3 LAAQ-B-LC 001 B 53

Basic Settings for Eluent Used in Reversed Phase Mode l Water (buffer solution) + water-soluble organic solvent Water-soluble organic solvent: Methanol Acetonitrile Tetrahydrofuran etc. v The mixing ratio of the water (buffer solution) and organic solvent has the greatest influence on separation. v If a buffer solution is used, its p. H value is an important separation parameter. v LAAQ-B-LC 001 B 54
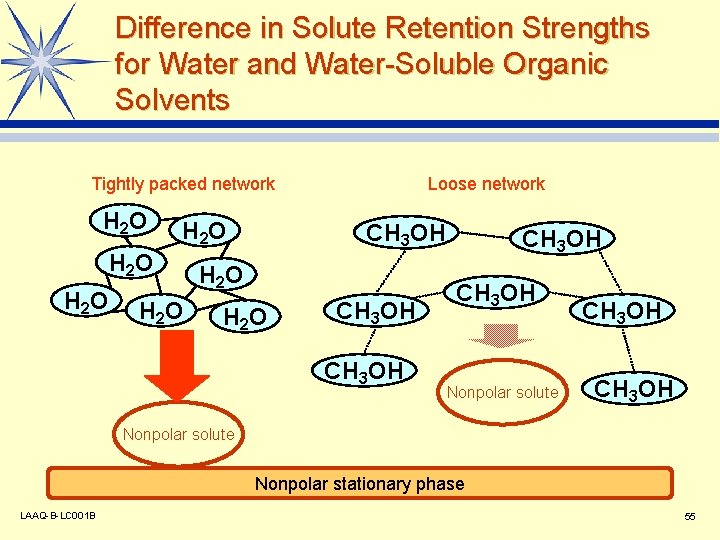
Difference in Solute Retention Strengths for Water and Water-Soluble Organic Solvents Tightly packed network H 2 O H 2 O Loose network CH 3 OH H 2 O CH 3 OH Nonpolar solute CH 3 OH Nonpolar solute Nonpolar stationary phase LAAQ-B-LC 001 B 55
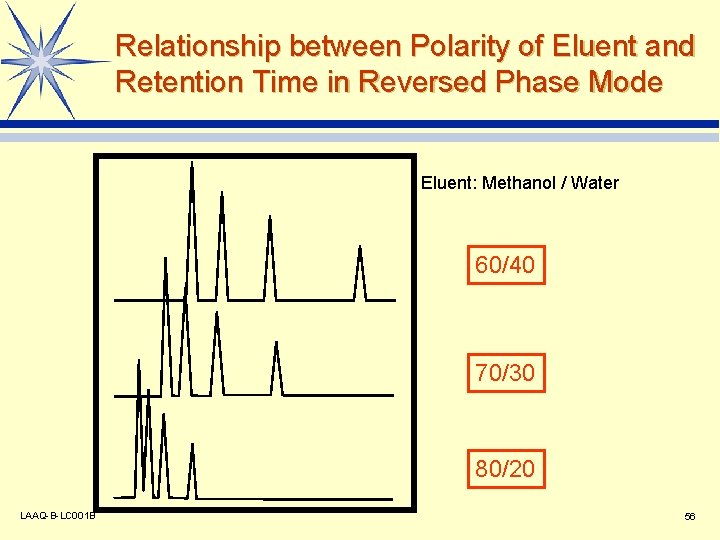
Relationship between Polarity of Eluent and Retention Time in Reversed Phase Mode Eluent: Methanol / Water 60/40 70/30 80/20 LAAQ-B-LC 001 B 56

Chromatogram Parameters Methods for Expressing Separation and Column Performance LAAQ-B-LC 001 B 57
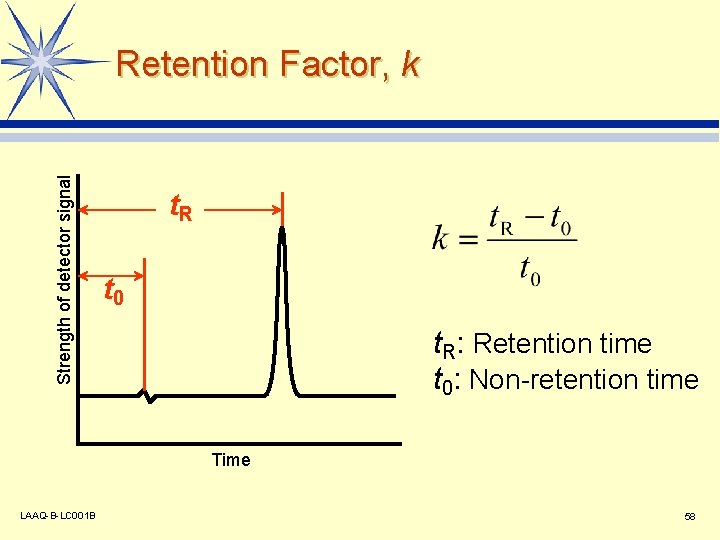
Strength of detector signal Retention Factor, k t. R t 0 t. R: Retention time t 0: Non-retention time Time LAAQ-B-LC 001 B 58
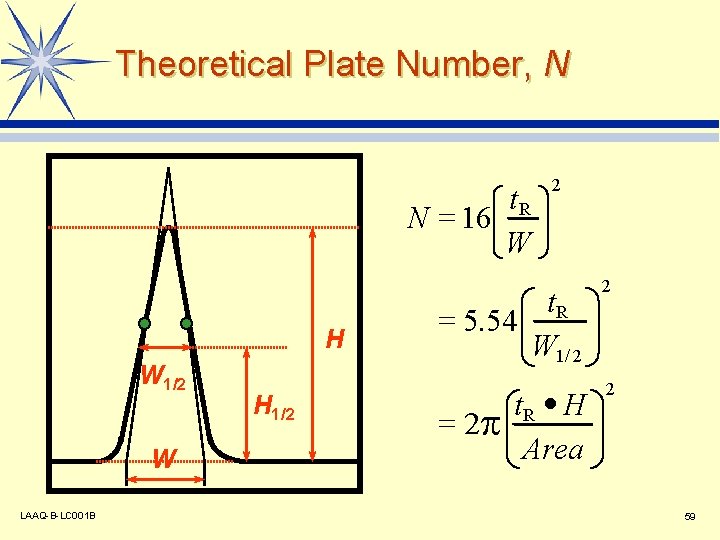
Theoretical Plate Number, N t. R N = 16 W H W 1/2 W LAAQ-B-LC 001 B H 1/2 2 t. R = 5. 54 W 1/ 2 2 t. R · H Area 2 = 2 59
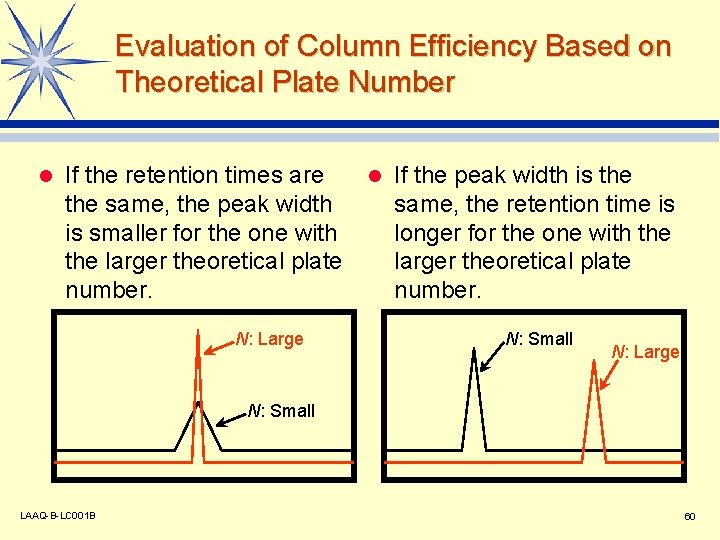
Evaluation of Column Efficiency Based on Theoretical Plate Number l If the retention times are the same, the peak width is smaller for the one with the larger theoretical plate number. N: Large l If the peak width is the same, the retention time is longer for the one with the larger theoretical plate number. N: Small N: Large N: Small LAAQ-B-LC 001 B 60
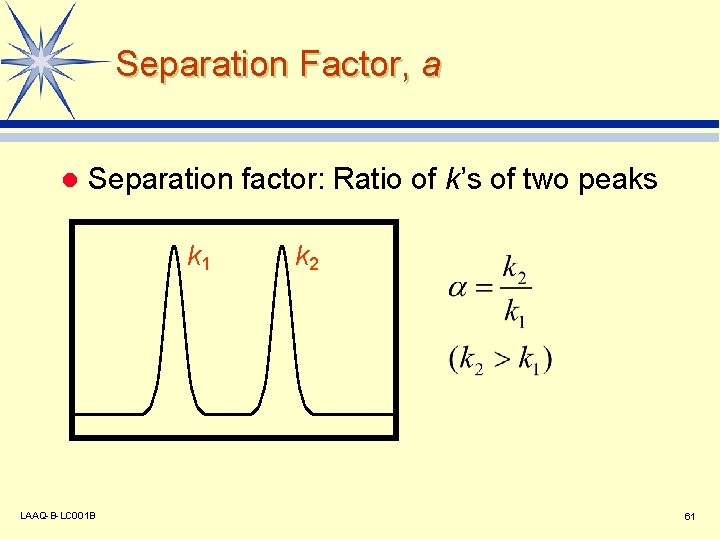
Separation Factor, a l Separation factor: Ratio of k’s of two peaks k 1 LAAQ-B-LC 001 B k 2 61
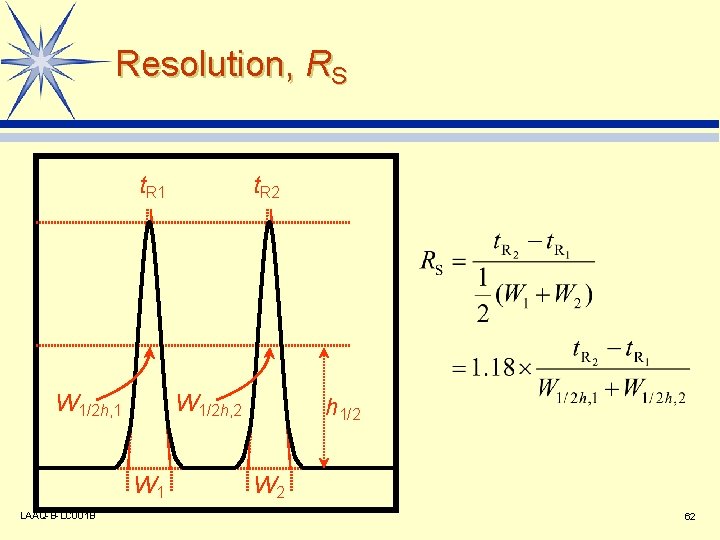
Resolution, RS t. R 1 W 1/2 h, 2 W 1 LAAQ-B-LC 001 B t. R 2 h 1/2 W 2 62
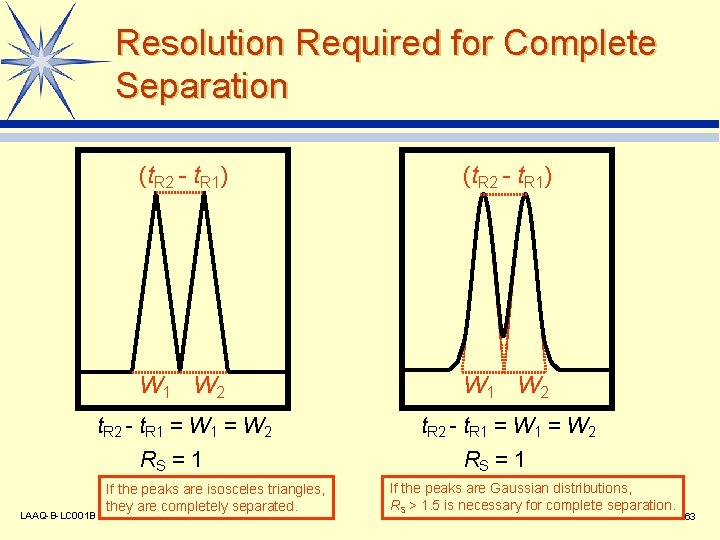
Resolution Required for Complete Separation (t. R 2 - t. R 1) W 1 W 2 t. R 2 - t. R 1 = W 2 RS = 1 LAAQ-B-LC 001 B If the peaks are isosceles triangles, they are completely separated. RS = 1 If the peaks are Gaussian distributions, RS > 1. 5 is necessary for complete separation. 63

Relationship Between Resolution and Other Parameters The resolution is a function of the separation factor, theoretical plate number, and the retention factor. l The separation can be improved by improving these 3 parameters! l LAAQ-B-LC 001 B RS = t. R 2 - t. R 1 1 (W 1 + W 2 ) 2 -1 1 = N 4 k’ 2 + 1 64
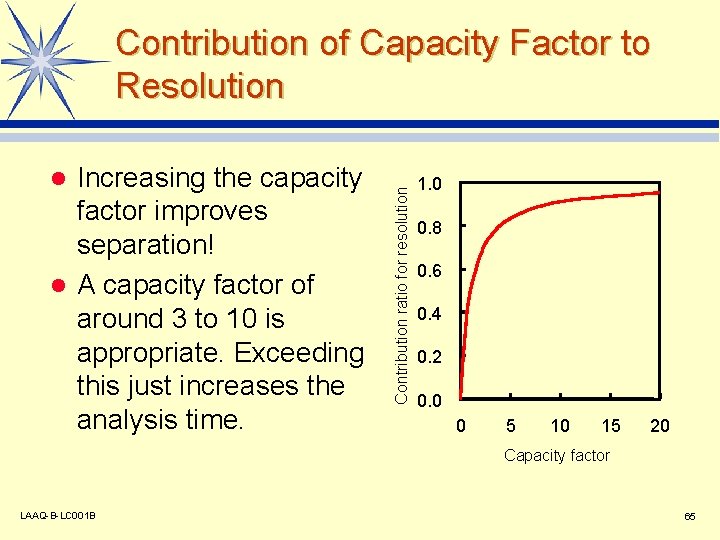
Increasing the capacity factor improves separation! l A capacity factor of around 3 to 10 is appropriate. Exceeding this just increases the analysis time. l Contribution ratio for resolution Contribution of Capacity Factor to Resolution 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 0 5 10 15 20 Capacity factor LAAQ-B-LC 001 B 65
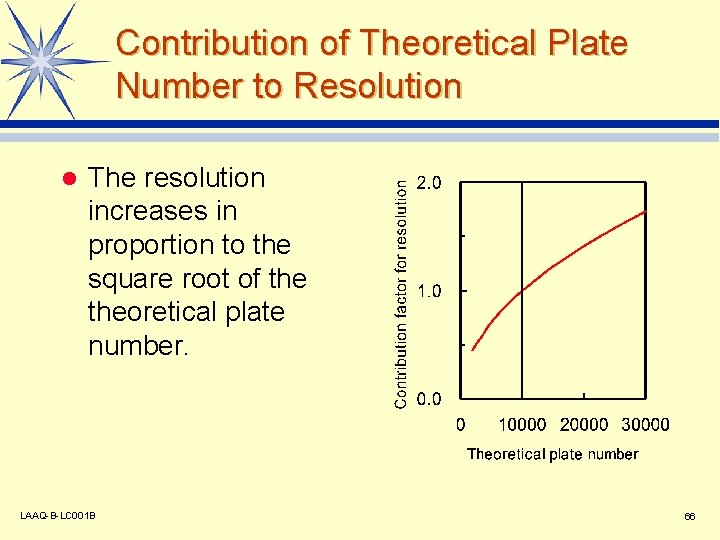
Contribution of Theoretical Plate Number to Resolution l The resolution increases in proportion to the square root of theoretical plate number. LAAQ-B-LC 001 B 66
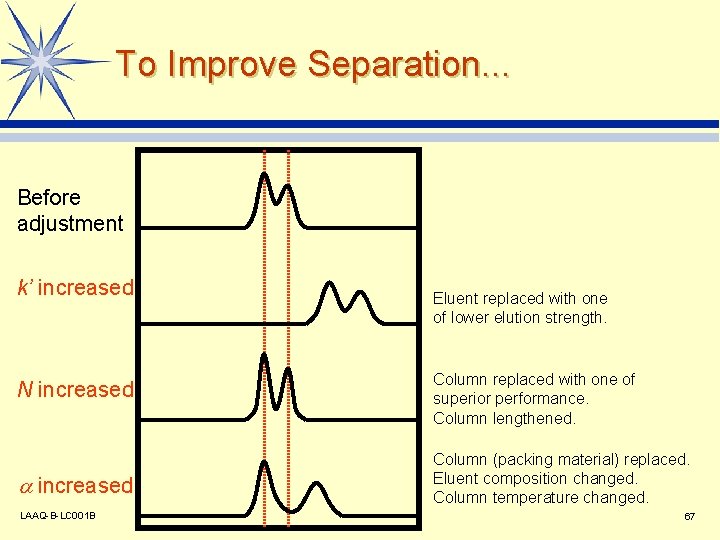
To Improve Separation. . . Before adjustment k’ increased N increased LAAQ-B-LC 001 B Eluent replaced with one of lower elution strength. Column replaced with one of superior performance. Column lengthened. Column (packing material) replaced. Eluent composition changed. Column temperature changed. 67

p. H Buffer Solution Used for Eluent Selection and Preparation of Buffer Solution LAAQ-B-LC 001 B 68
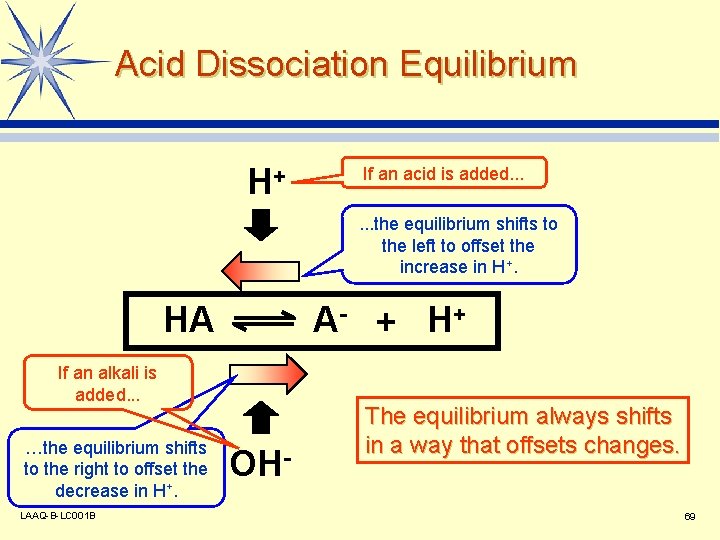
Acid Dissociation Equilibrium H+ If an acid is added. . . the equilibrium shifts to the left to offset the increase in H+. HA A- + H+ If an alkali is added. . . …the equilibrium shifts to the right to offset the decrease in H+. LAAQ-B-LC 001 B OH- The equilibrium always shifts in a way that offsets changes. 69
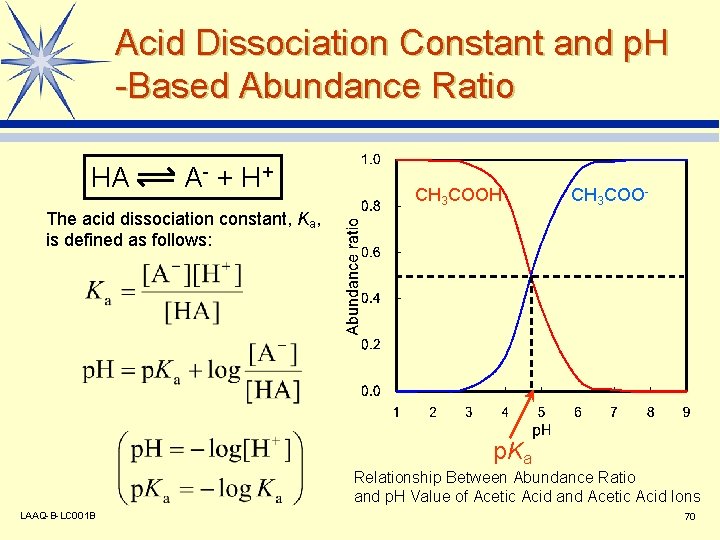
Acid Dissociation Constant and p. H -Based Abundance Ratio HA A - + H+ CH 3 COOH CH 3 COO- The acid dissociation constant, Ka, is defined as follows: p. Ka Relationship Between Abundance Ratio and p. H Value of Acetic Acid and Acetic Acid Ions LAAQ-B-LC 001 B 70

Preparing p. H Buffer Solution l Use a weak acid with a p. Ka value close to the desired p. H value. v l Example: Preparing a buffer solution for a p. H value of around 4. 8. Use acetic acid, which has a p. Ka value of 4. 8. Make the concentrations of HA and A- roughly equal. Mix an acid with its salt. v Example: Mix acetic acid and sodium acetate so that they have the same molar concentration. LAAQ-B-LC 001 B 71

Buffer Solutions Used for HPLC Eluent l Requirements l Commonly Used Acids v Phosphoric acid High buffering power at p. Ka 2. 1, 7. 2, 12. 3 prescribed p. H. v Acetic acid v Does not adversely p. Ka 4. 8 affect detection. v Citric acid v Does not damage p. Ka 3. 1, 4. 8, 6. 4 column or equipment. l Concentration v Inexpensive. v If only to adjust p. H, 10 mmol/L is sufficient. v LAAQ-B-LC 001 B 72

Characteristics of Phosphate Buffer Solution l Advantages v Three dissociation states (p. Ka 2. 1, 7. 2, 12. 3) Possible to prepare buffer solutions of various p. H values. l Disadvantages v No volatility Difficult to use for LCMS or evaporative light scattering detection. No UV absorption v Inexpensive v LAAQ-B-LC 001 B 73

Reversed Phase Chromatography Part 2 Consideration of Analytical Conditions LAAQ-B-LC 001 B 74

Guidelines for Setting Mobile Phase Conditions (1) Neutral (Nonionic) Substances l Eluent Composition v Water / acetonitrile v Water / methanol l Separation Adjustment v Changing the mixing ratio of the water and organic solvent v Changing the type of organic solvent LAAQ-B-LC 001 B 75
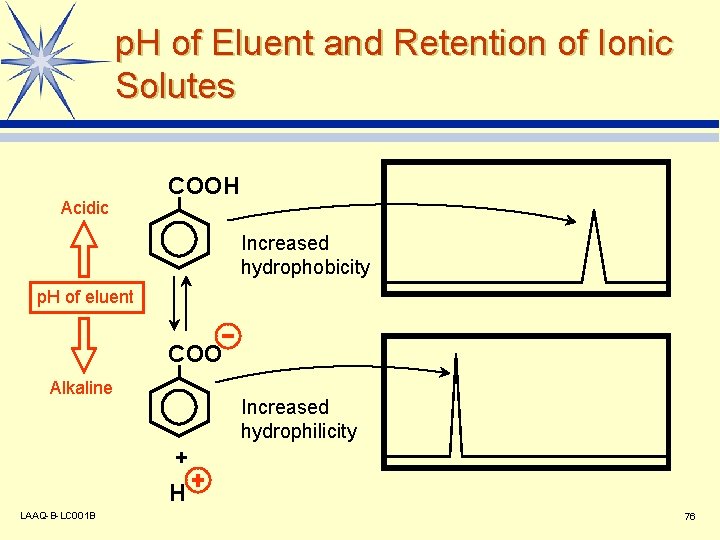
p. H of Eluent and Retention of Ionic Solutes Acidic COOH Increased hydrophobicity p. H of eluent COO Alkaline Increased hydrophilicity + H LAAQ-B-LC 001 B 76

Guidelines for Setting Mobile Phase Conditions (2) Acidic (Anionic) Substances l Eluent Composition Acidic buffer solution / acetonitrile v Acidic buffer solution / methanol v Increase retention strength by making the eluent acidic and suppressing ionization! LAAQ-B-LC 001 B 77
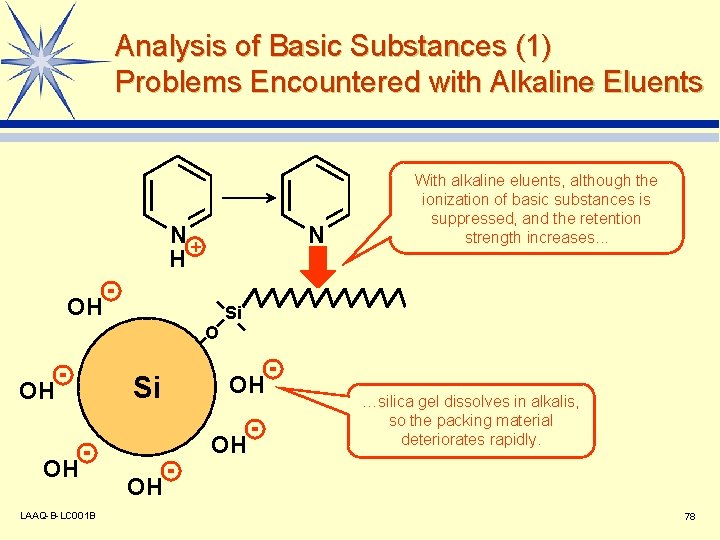
Analysis of Basic Substances (1) Problems Encountered with Alkaline Eluents N+ H N OH With alkaline eluents, although the ionization of basic substances is suppressed, and the retention strength increases. . . Si O OH OH LAAQ-B-LC 001 B Si OH OH …silica gel dissolves in alkalis, so the packing material deteriorates rapidly. OH 78
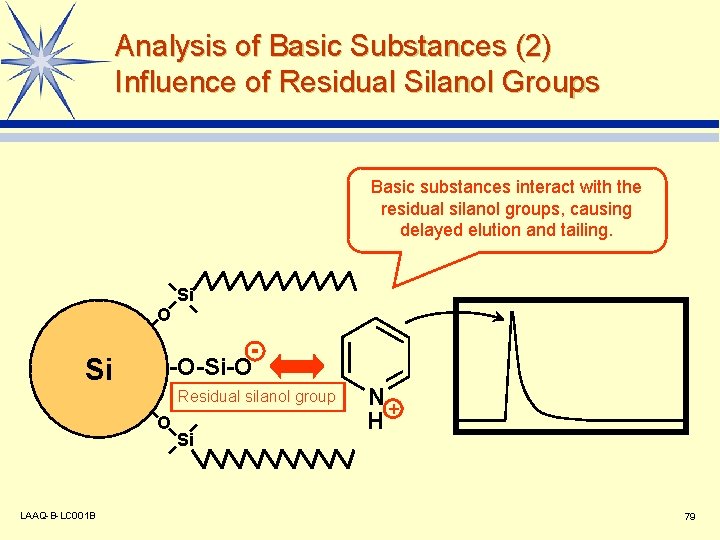
Analysis of Basic Substances (2) Influence of Residual Silanol Groups Basic substances interact with the residual silanol groups, causing delayed elution and tailing. Si O Si -O-Si-O Residual silanol group O LAAQ-B-LC 001 B Si N+ H 79
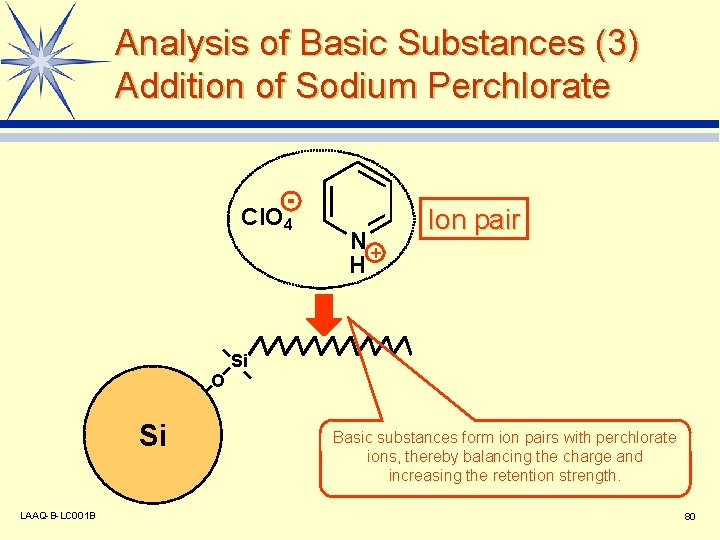
Analysis of Basic Substances (3) Addition of Sodium Perchlorate Cl. O 4 N+ H Ion pair Si O Si LAAQ-B-LC 001 B Basic substances form ion pairs with perchlorate ions, thereby balancing the charge and increasing the retention strength. 80

Guidelines for Setting Mobile Phase Conditions (3) Basic Substances (Cationic Substances) l Eluent Composition Acidic buffer solution containing anions with a low charge density (e. g. , perchlorate ions) / acetonitrile v As above / methanol v Making eluent acidic Suppresses dissociation of residual silanol groups Prevents tailing! Adding perchlorate ions Forms ion pairs Increases retention strength! Suppresses tailing! LAAQ-B-LC 001 B 81
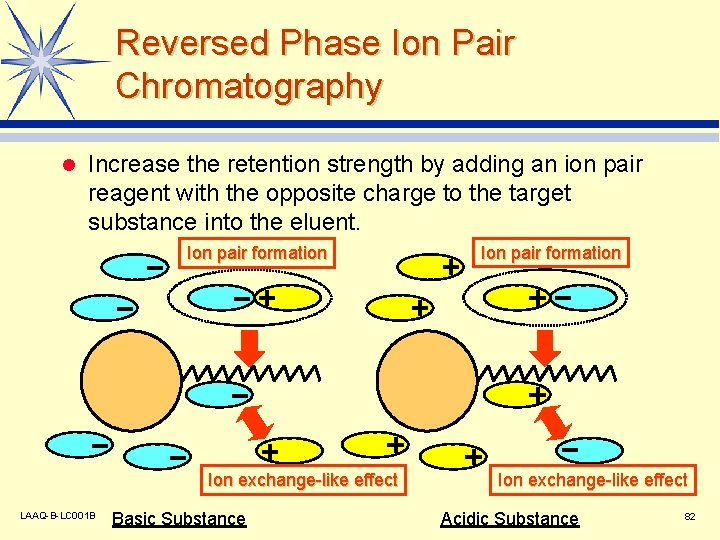
Reversed Phase Ion Pair Chromatography l Increase the retention strength by adding an ion pair reagent with the opposite charge to the target substance into the eluent. Ion pair formation Ion exchange-like effect LAAQ-B-LC 001 B Basic Substance Ion pair formation Ion exchange-like effect Acidic Substance 82

Representative Ion Pair Reagents l Anionic Compounds v Tetra-n-butylammonium l Cationic hydroxide (TBA) Compounds v Pentanesulfonic acid sodium salt (C 5) v Hexanesulfonic acid sodium salt (C 6) v Heptanesulfonic acid sodium salt (C 7) v Octanesulfonic acid sodium salt (C 8) LAAQ-B-LC 001 B 83

Points to Note Concerning the Use of Ion Pairs l Selection of Ion Pair Reagent v l p. H of Eluent v l The retention strength changes according to whether or not ionization takes place. Concentration of Ion Pair Reagent v l In general, the retention strength increases with the length of the alkyl chain. In general, the retention strength increases with the ion pair concentration, but there is an upper limit. Proportion of Organic Solvent in Eluent v LAAQ-B-LC 001 B Optimize the separation conditions by considering the type and concentration of the ion pair reagent. 84

HPLC Separation Modes Other Than Reversed Phase Chromatography LAAQ-B-LC 001 B 85

HPLC Separation Modes l Adsorption (liquid-solid) chromatography l Partition (liquid-liquid) chromatography v Normal phase partition chromatography v Reversed phase partition chromatography l Ion exchange chromatography l Size exclusion chromatography LAAQ-B-LC 001 B 86

Adsorption Chromatography l. A solid such as silica gel is used as the stationary phase, and differences, mainly in the degree of adsorption to its surface, are used to separate the solutes. l Liquid-solid chromatography l The retention strength increases with the hydrophilicity of the solute. LAAQ-B-LC 001 B 87

Partition Chromatography l. A liquid (or a substance regarded as a liquid) is used as the stationary phase, and the solute is separated according to whether it dissolves more readily in the stationary or mobile phase. l Liquid-liquid chromatography LAAQ-B-LC 001 B 88
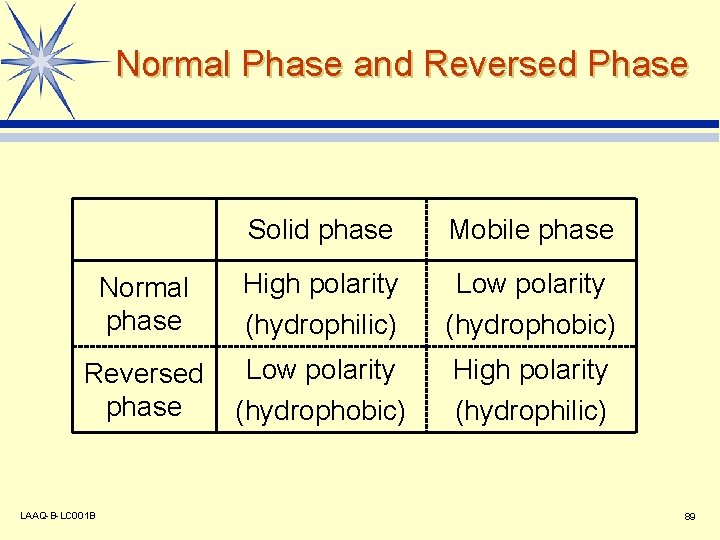
Normal Phase and Reversed Phase Solid phase Mobile phase Normal phase High polarity (hydrophilic) Low polarity (hydrophobic) Reversed phase Low polarity (hydrophobic) High polarity (hydrophilic) LAAQ-B-LC 001 B 89

Normal Phase (Partition) Chromatography Partition chromatography in which the stationary phase has a high polarity (hydrophilic) and the mobile phase has a low polarity (hydrophobic) l Essentially based on the same separation mechanism as adsorption chromatography in which the stationary phase has a hydrophilic base, such as silica gel l LAAQ-B-LC 001 B 90
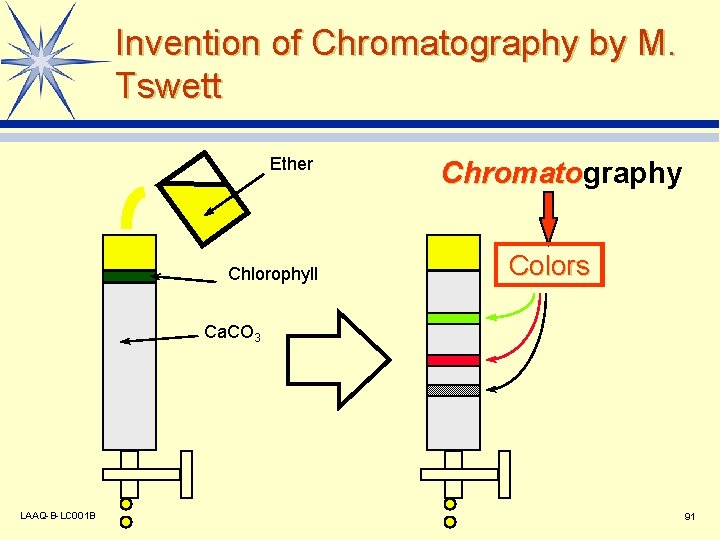
Invention of Chromatography by M. Tswett Ether Chlorophyll Chromatography Chromato Colors Ca. CO 3 LAAQ-B-LC 001 B 91

Stationary Phase and Mobile Phase Used in Normal Phase Mode l Stationary Phase Silica gel: -Si-OH v Cyano type: -Si-CH 2 CH 2 CN v Amino type: -Si-CH 2 CH 2 NH 2 v Diol type: -Si-CH 2 CH 2 OCH(OH)-CH 2 OH v l Mobile Phase Basic solvents: Aliphatic hydrocarbons, aromatic hydrocarbons, etc. v Additional solvents: Alcohols, ethers, etc. v LAAQ-B-LC 001 B 92
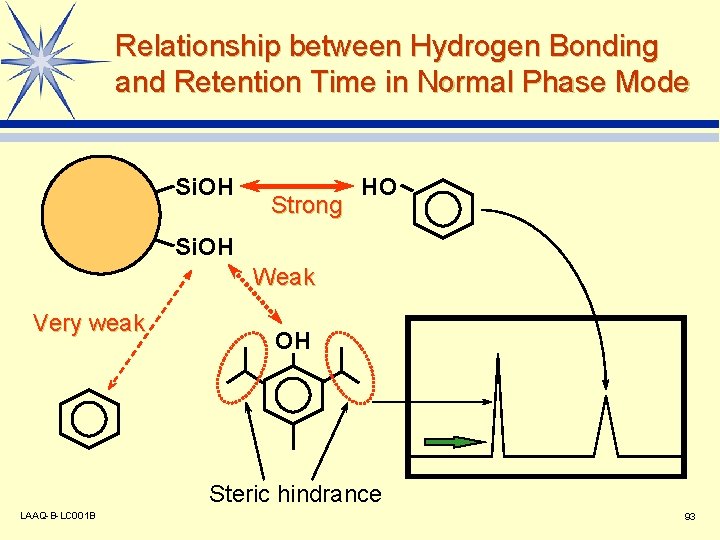
Relationship between Hydrogen Bonding and Retention Time in Normal Phase Mode Si. OH Strong HO Si. OH Weak Very weak OH Steric hindrance LAAQ-B-LC 001 B 93
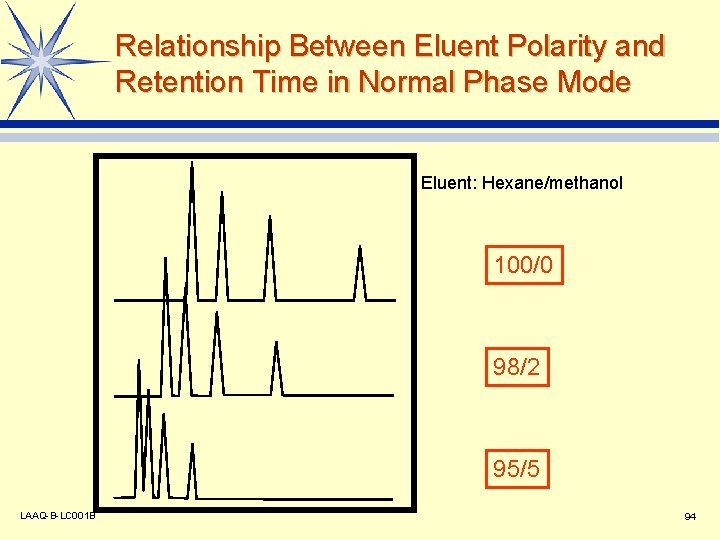
Relationship Between Eluent Polarity and Retention Time in Normal Phase Mode Eluent: Hexane/methanol 100/0 98/2 95/5 LAAQ-B-LC 001 B 94

Comparison of Normal Phase and Reversed Phase l Normal Phase v v LAAQ-B-LC 001 B Effective for separation of structural isomers Offers separation selectivity not available with reversed phase Stabilizes slowly and is prone to fluctuations in retention time Eluents are expensive l Reversed Phase v v v Wide range of applications Effective for separation of homologs Stationary phase has long service life Stabilizes quickly Eluents are inexpensive and easy to use 95
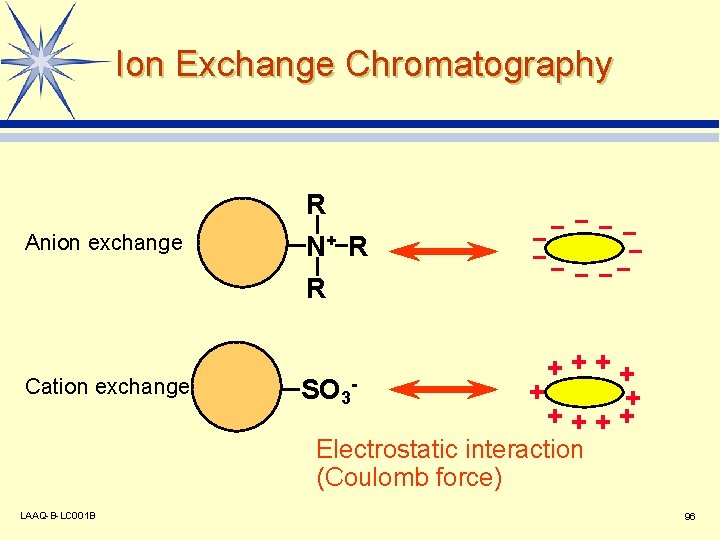
Ion Exchange Chromatography Anion exchange Cation exchange R N+ R R SO 3 - ++++ + + ++++ Electrostatic interaction (Coulomb force) LAAQ-B-LC 001 B 96

Stationary Phase Used in Ion Exchange Mode l Base Material Resin is often used. v Silica gel is also used. v l Cation Exchange Column Strong cation exchange v Week cation exchange v l (SCX) (WCX) -SO 3 -COO- (SAX) (WAX) -NR 3+ -NHR 2+ Anion Exchange Column Strong anion exchange v Week anion exchange v LAAQ-B-LC 001 B 97
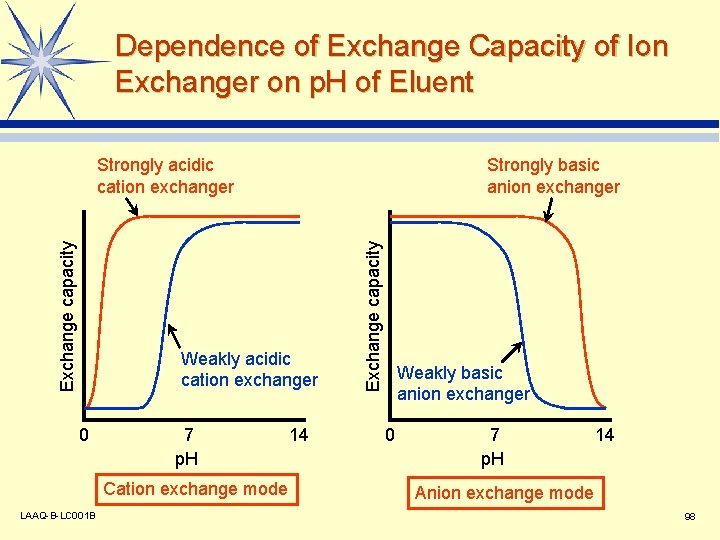
Dependence of Exchange Capacity of Ion Exchanger on p. H of Eluent Strongly basic anion exchanger Weakly acidic cation exchanger 0 7 p. H Cation exchange mode LAAQ-B-LC 001 B 14 Exchange capacity Strongly acidic cation exchanger Weakly basic anion exchanger 0 7 p. H 14 Anion exchange mode 98
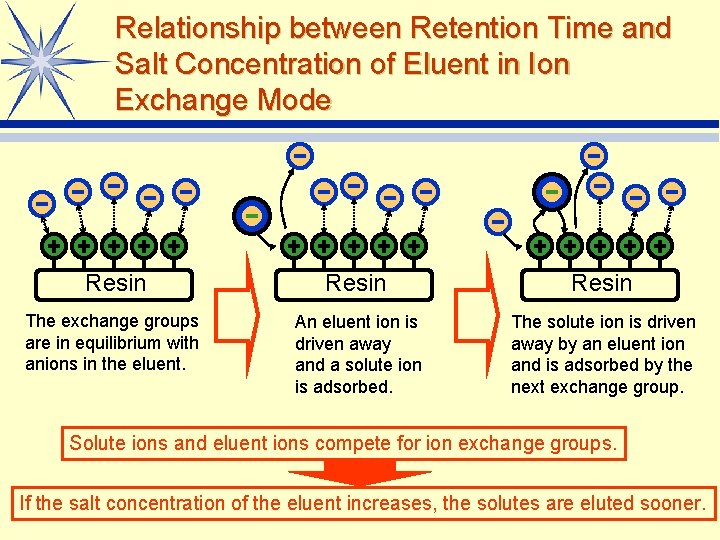
Relationship between Retention Time and Salt Concentration of Eluent in Ion Exchange Mode Resin The exchange groups are in equilibrium with anions in the eluent. An eluent ion is driven away and a solute ion is adsorbed. The solute ion is driven away by an eluent ion and is adsorbed by the next exchange group. Solute ions and eluent ions compete for ion exchange groups. If the salt concentration of the eluent increases, the solutes are eluted sooner. LAAQ-B-LC 001 B 99
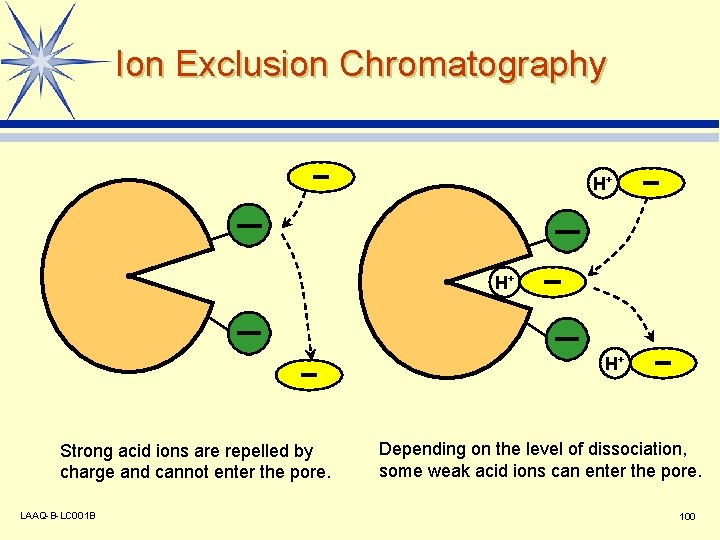
Ion Exclusion Chromatography H+ H+ H+ Strong acid ions are repelled by charge and cannot enter the pore. LAAQ-B-LC 001 B Depending on the level of dissociation, some weak acid ions can enter the pore. 100

Size Exclusion Chromatography Separation is based on the size (bulkiness) of molecules. l The name varies with the application field! l v Size Exclusion Chromatography (SEC) v Gel Permeation Chromatography (GPC) Chemical industry fields, synthetic polymers, nonaqueous systems v Gel Filtration Chromatography (GFC) Biochemical fields, biological macromolecules, aqueous systems LAAQ-B-LC 001 B 101
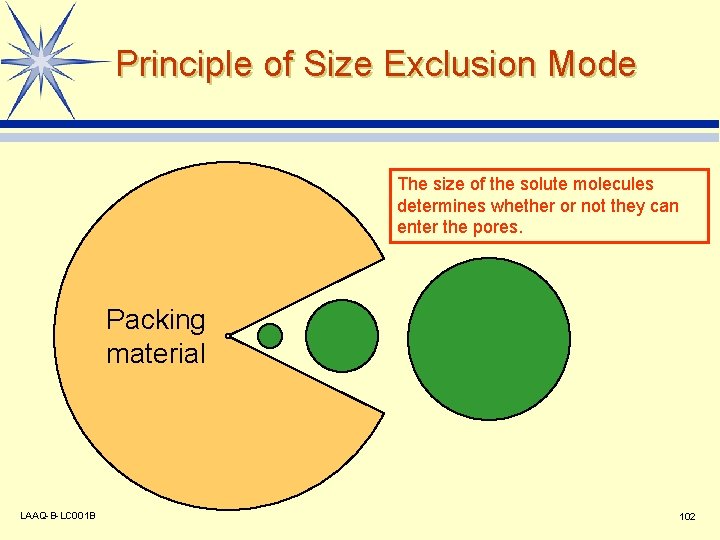
Principle of Size Exclusion Mode The size of the solute molecules determines whether or not they can enter the pores. Packing material LAAQ-B-LC 001 B 102
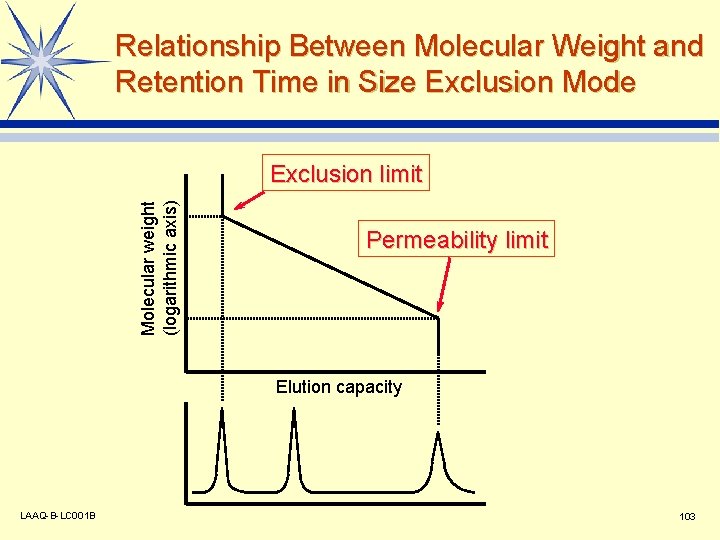
Relationship Between Molecular Weight and Retention Time in Size Exclusion Mode Molecular weight (logarithmic axis) Exclusion limit Permeability limit Elution capacity LAAQ-B-LC 001 B 103
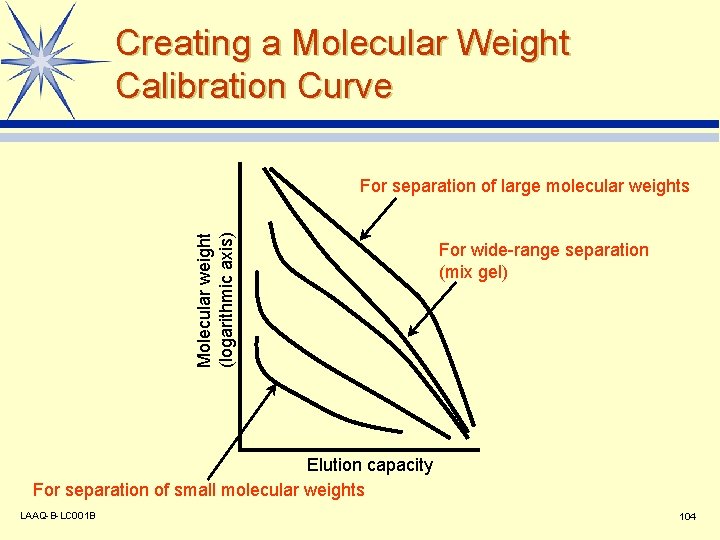
Creating a Molecular Weight Calibration Curve Molecular weight (logarithmic axis) For separation of large molecular weights For wide-range separation (mix gel) Elution capacity For separation of small molecular weights LAAQ-B-LC 001 B 104

Calculating Molecular Weights Chromatogram l Various Average Molecular Weights v Calibration curve v v l Retention time LAAQ-B-LC 001 B Mn: Number-average molecular weight Mw: Weight-average molecular weight Mz: Z-average molecular weight, etc. Molecular weights and molecular weight distributions are calculated using special calculation software. 105

Guidelines for Selecting Separation Mode (1) Required Information l Soluble solvent l Molecular weight l Structural formula and chemical properties v Do the substances ionize? v Is there UV absorption or fluorescence? v Is derivatization possible? etc. LAAQ-B-LC 001 B 106

Guidelines for Selecting Separation Mode (2) Basic Policy Reversed phase mode using an ODS column is the first choice! l Exceptions l Large molecular weight (> 2, 000) Size exclusion v Optical isomers Chiral column v Stereoisomers, positional isomers Normal phase / adsorption v Inorganic ions Ion chromatography v Sugars, amino acids, short-chain fatty acids Special column v LAAQ-B-LC 001 B 107

HPLC Hardware: Part 2 Detectors and Their Ranges of Application LAAQ-B-LC 001 B 108

Detection Condition Requirements l Sensitivity v The detector must have the appropriate level of sensitivity. l Selectivity v The detector must be able to detect the target substance without, if possible, detecting other substances. Adaptability to separation conditions l Operability, etc. l LAAQ-B-LC 001 B 109

Representative HPLC Detectors l l l l LAAQ-B-LC 001 B UV-VIS absorbance detector Photodiode array-type UV-VIS absorbance detector Fluorescence detector Refractive index detector Evaporative light scattering detector Electrical conductivity detector Electrochemical detector Mass spectrometer 110
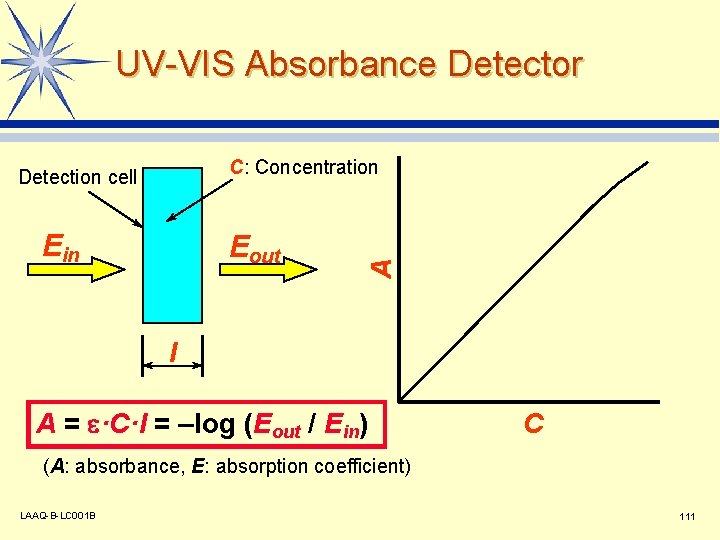
UV-VIS Absorbance Detector Ein Eout A C: Concentration Detection cell l A = e·C·l = –log (Eout / Ein) C (A: absorbance, E: absorption coefficient) LAAQ-B-LC 001 B 111
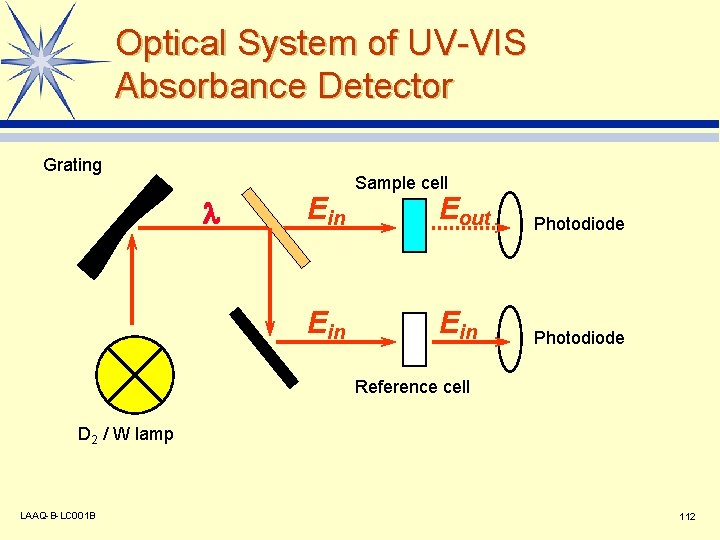
Optical System of UV-VIS Absorbance Detector Grating l Ein Sample cell Eout Photodiode Ein Photodiode Reference cell D 2 / W lamp LAAQ-B-LC 001 B 112
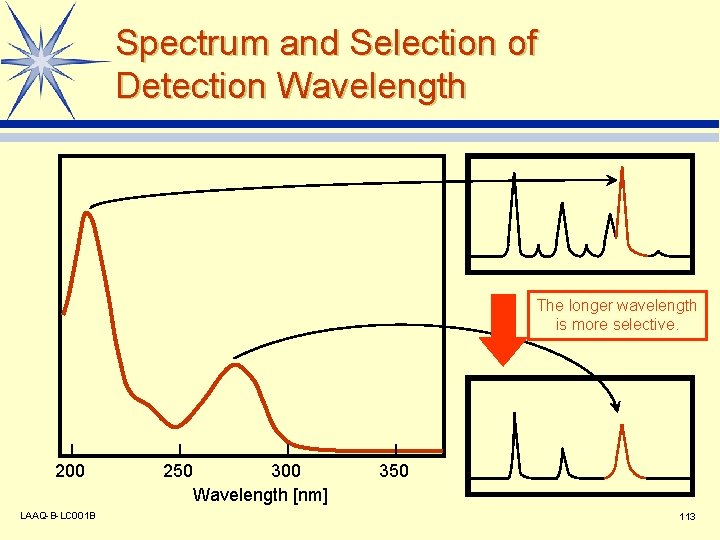
Spectrum and Selection of Detection Wavelength The longer wavelength is more selective. 200 LAAQ-B-LC 001 B 250 300 Wavelength [nm] 350 113
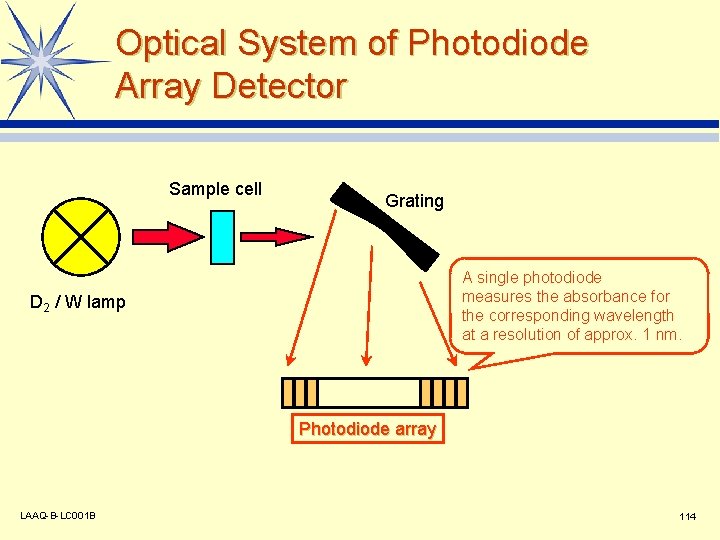
Optical System of Photodiode Array Detector Sample cell Grating A single photodiode measures the absorbance for the corresponding wavelength at a resolution of approx. 1 nm. D 2 / W lamp Photodiode array LAAQ-B-LC 001 B 114
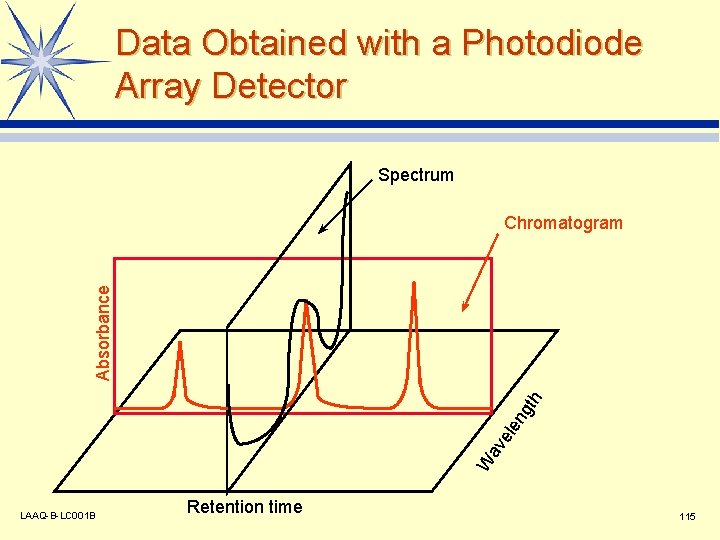
Data Obtained with a Photodiode Array Detector Spectrum W av ele ng th Absorbance Chromatogram LAAQ-B-LC 001 B Retention time 115

Advantages of Photodiode Array Detectors l Peak Identification Using Spectra v Complementation retention time v Library searches l Evaluation of identification based on of Peak Purity v Purity evaluation performed by comparison of the shape of spectra from the peak detection start point to the peak detection end point LAAQ-B-LC 001 B 116
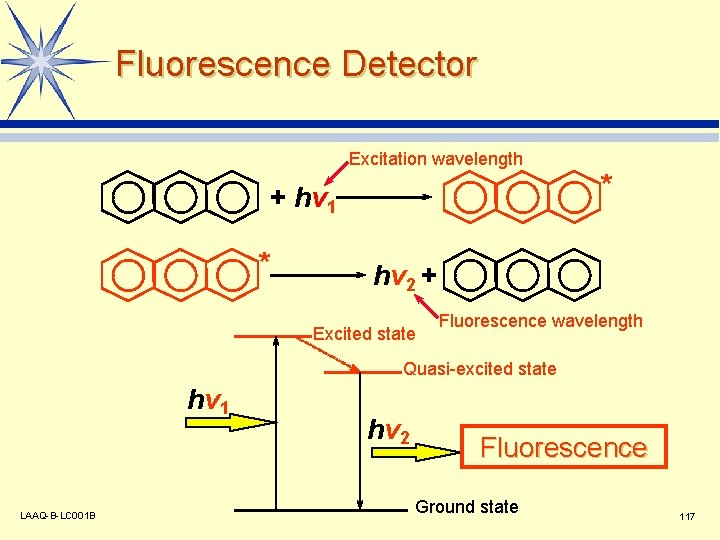
Fluorescence Detector Excitation wavelength + hv 1 * * hv 2 + Excited state Fluorescence wavelength Quasi-excited state hv 1 LAAQ-B-LC 001 B hv 2 Fluorescence Ground state 117
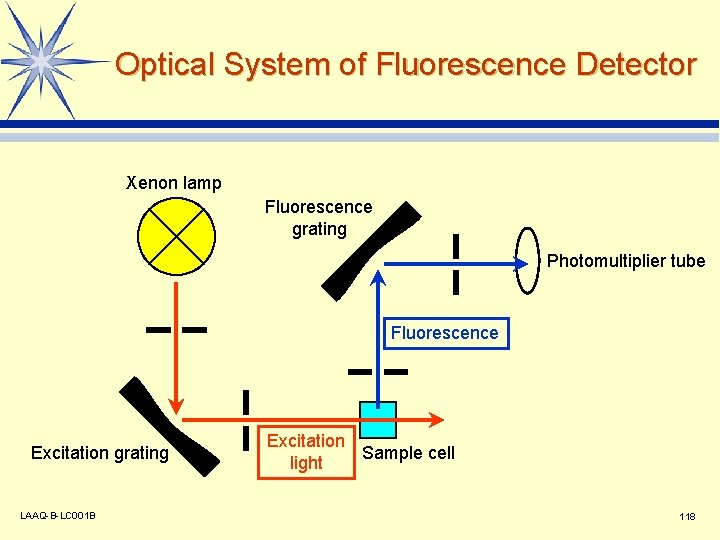
Optical System of Fluorescence Detector Xenon lamp Fluorescence grating Photomultiplier tube Fluorescence Excitation grating LAAQ-B-LC 001 B Excitation Sample cell light 118
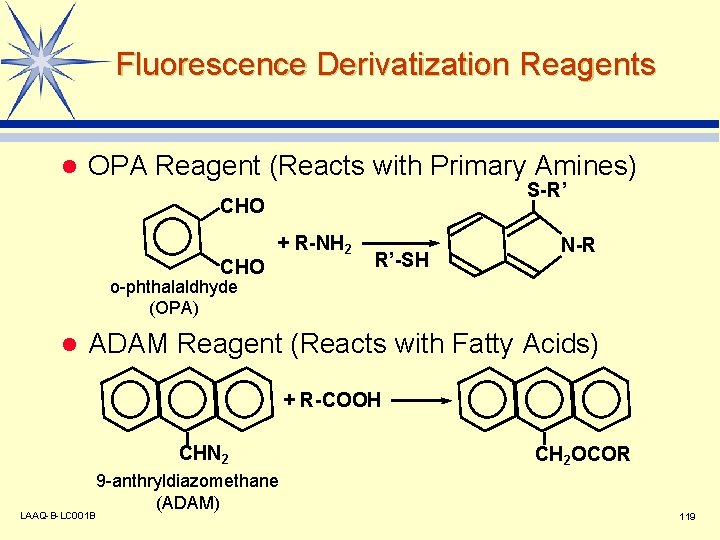
Fluorescence Derivatization Reagents l OPA Reagent (Reacts with Primary Amines) S-R’ CHO + R-NH 2 R’-SH N-R o-phthalaldhyde (OPA) l ADAM Reagent (Reacts with Fatty Acids) + R-COOH CHN 2 LAAQ-B-LC 001 B 9 -anthryldiazomethane (ADAM) CH 2 OCOR 119
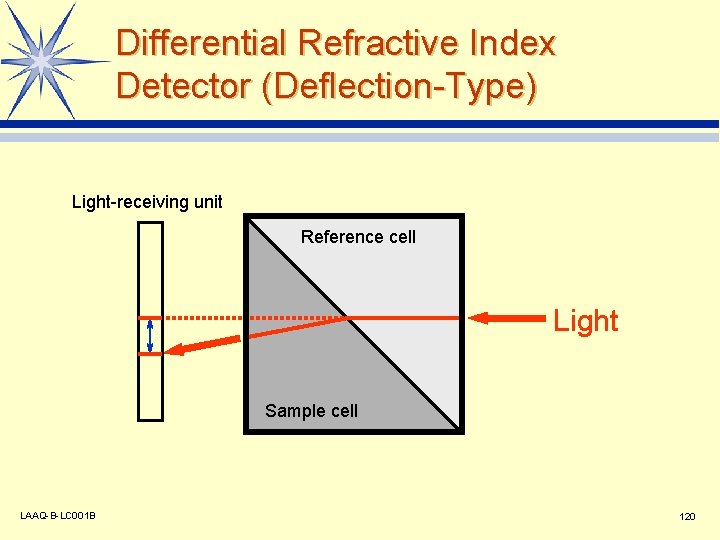
Differential Refractive Index Detector (Deflection-Type) Light-receiving unit Reference cell Light Sample cell LAAQ-B-LC 001 B 120
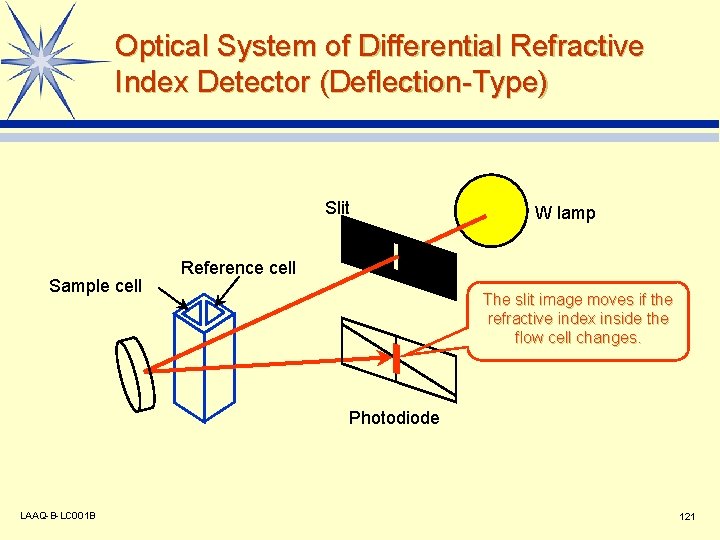
Optical System of Differential Refractive Index Detector (Deflection-Type) Slit Sample cell W lamp Reference cell The slit image moves if the refractive index inside the flow cell changes. Photodiode LAAQ-B-LC 001 B 121
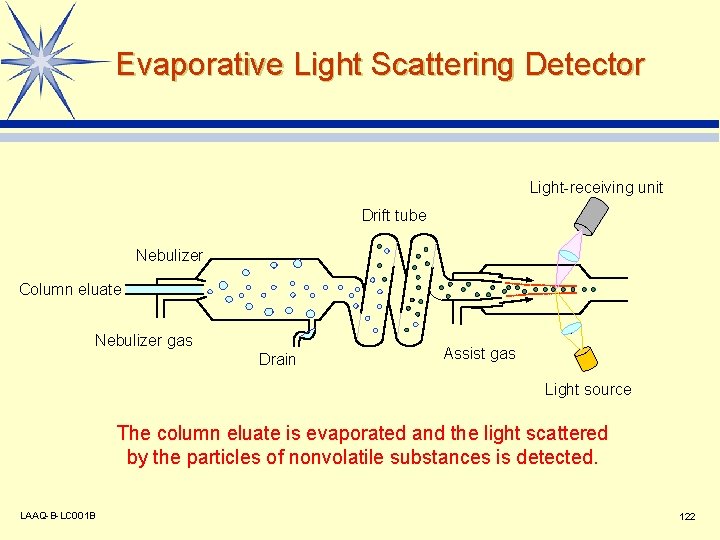
Evaporative Light Scattering Detector Light-receiving unit Drift tube Nebulizer Column eluate Nebulizer gas Drain Assist gas Light source The column eluate is evaporated and the light scattered by the particles of nonvolatile substances is detected. LAAQ-B-LC 001 B 122
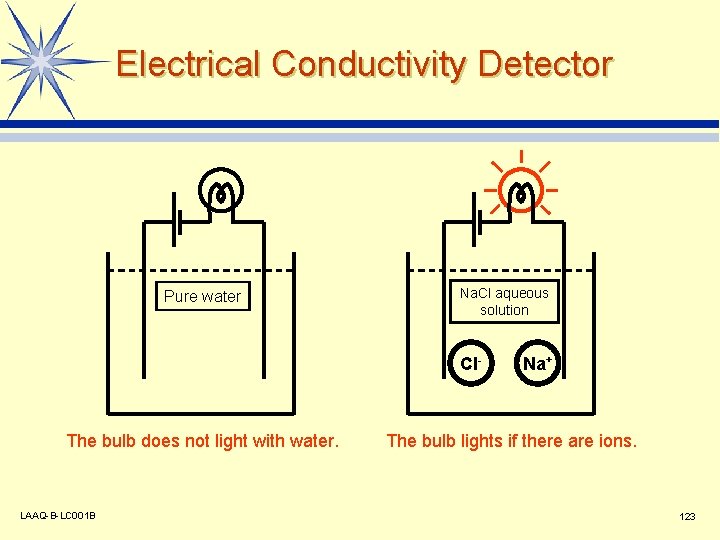
Electrical Conductivity Detector Pure water Na. Cl aqueous solution Cl- The bulb does not light with water. LAAQ-B-LC 001 B Na+ The bulb lights if there are ions. 123
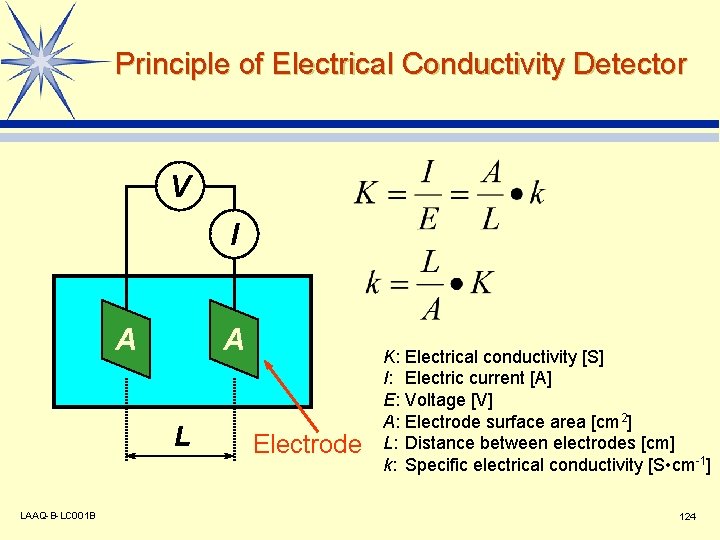
Principle of Electrical Conductivity Detector V I A A L LAAQ-B-LC 001 B Electrode K: I: E: A: L: k: Electrical conductivity [S] Electric current [A] Voltage [V] Electrode surface area [cm 2] Distance between electrodes [cm] Specific electrical conductivity [S • cm-1] 124
![Limiting Equivalent Ion Conductance, l [S • cm 2/mol], in Aqueous Solution (25ºC) LAAQ-B-LC Limiting Equivalent Ion Conductance, l [S • cm 2/mol], in Aqueous Solution (25ºC) LAAQ-B-LC](http://slidetodoc.com/presentation_image/26e5333a36f3b33d6d6980cc7d5507ed/image-125.jpg)
Limiting Equivalent Ion Conductance, l [S • cm 2/mol], in Aqueous Solution (25ºC) LAAQ-B-LC 001 B 125
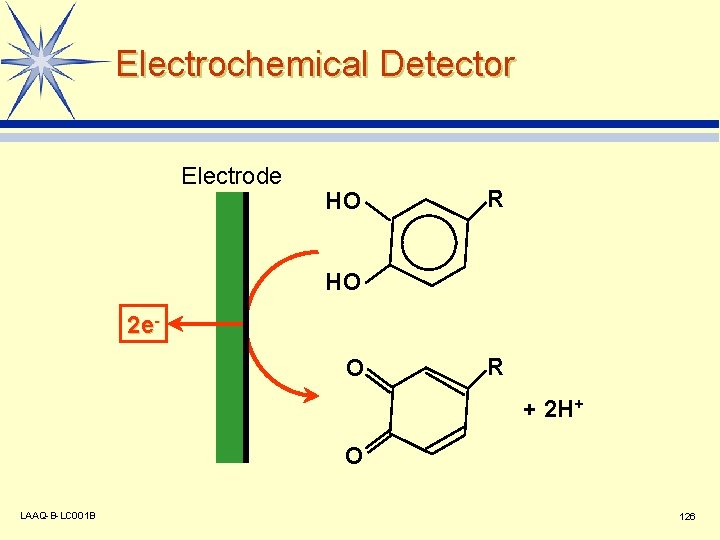
Electrochemical Detector Electrode HO R HO 2 e. O R + 2 H+ O LAAQ-B-LC 001 B 126
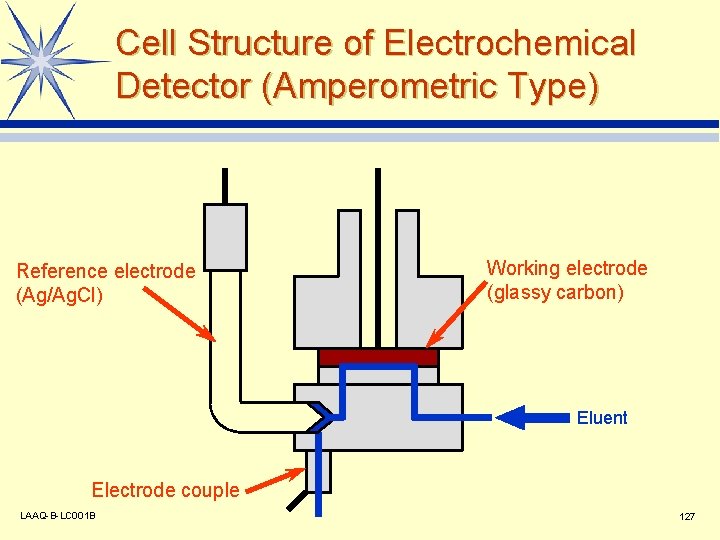
Cell Structure of Electrochemical Detector (Amperometric Type) Reference electrode (Ag/Ag. Cl) Working electrode (glassy carbon) Eluent Electrode couple LAAQ-B-LC 001 B 127
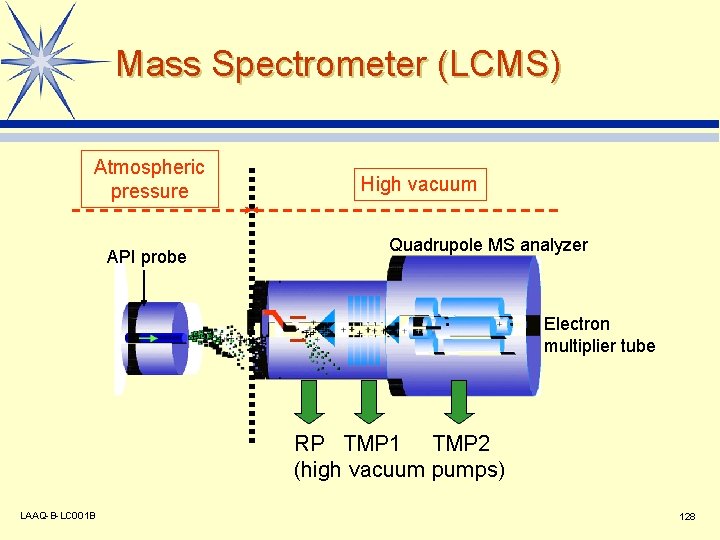
Mass Spectrometer (LCMS) Atmospheric pressure API probe High vacuum Quadrupole MS analyzer Electron multiplier tube RP TMP 1 TMP 2 (high vacuum pumps) LAAQ-B-LC 001 B 128
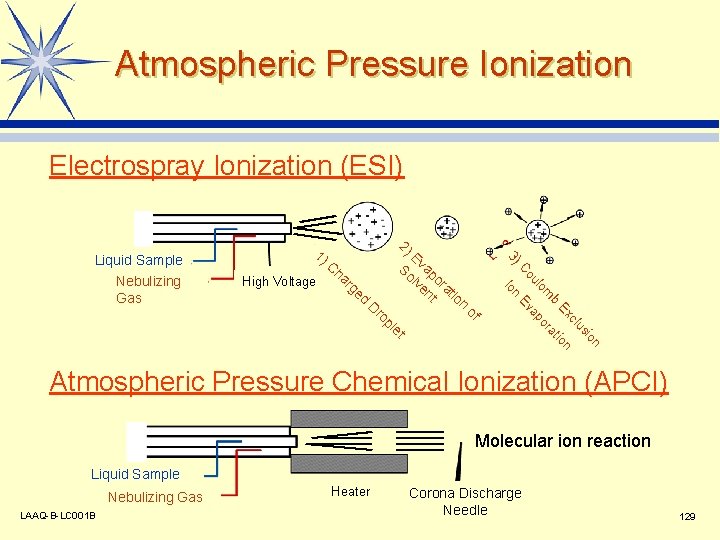
Atmospheric Pressure Ionization Electrospray Ionization (ESI) pl e of n io us cl Ex tion b a m or lo ap ro Ev D n ge d ou ha r C High Voltage E So vap lv ora en t t ion Io Nebulizing Gas C 2) 3) Liquid Sample 1) t Atmospheric Pressure Chemical Ionization (APCI) Molecular ion reaction Liquid Sample Nebulizing Gas LAAQ-B-LC 001 B Heater Corona Discharge Needle 129
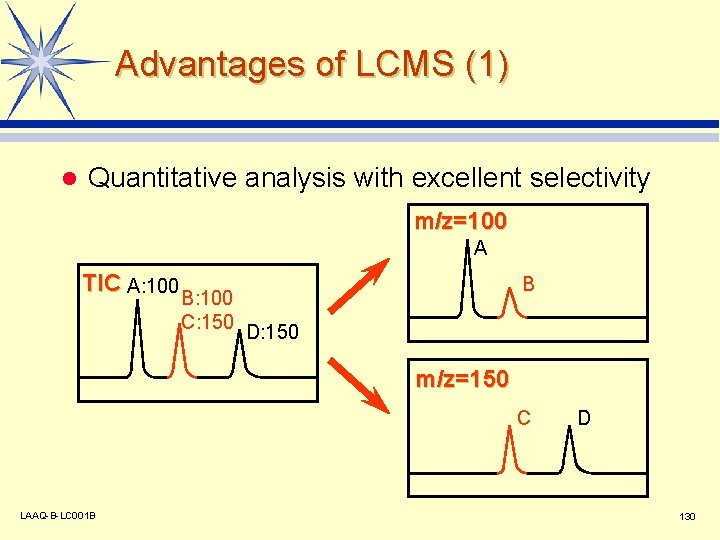
Advantages of LCMS (1) l Quantitative analysis with excellent selectivity m/z=100 A TIC A: 100 B B: 100 C: 150 D: 150 m/z=150 C LAAQ-B-LC 001 B D 130
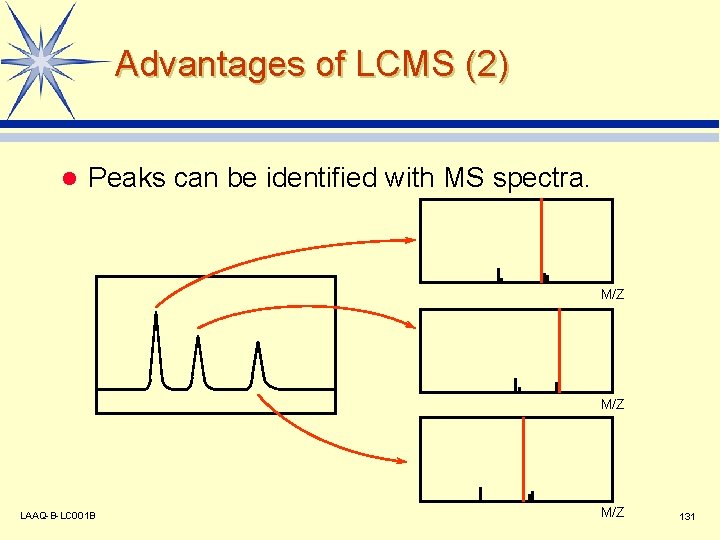
Advantages of LCMS (2) l Peaks can be identified with MS spectra. M/Z LAAQ-B-LC 001 B M/Z 131
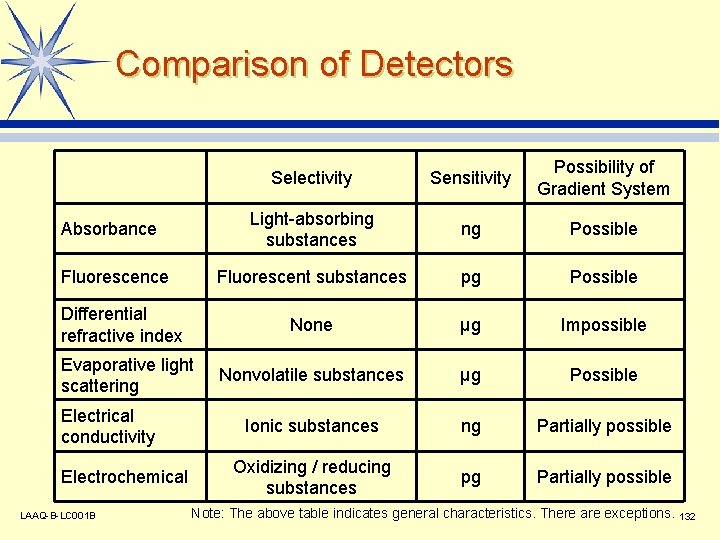
Comparison of Detectors Absorbance Fluorescence Differential refractive index Evaporative light scattering Electrical conductivity Electrochemical LAAQ-B-LC 001 B Selectivity Sensitivity Possibility of Gradient System Light-absorbing substances ng Possible Fluorescent substances pg Possible None µg Impossible Nonvolatile substances µg Possible Ionic substances ng Partially possible Oxidizing / reducing substances pg Partially possible Note: The above table indicates general characteristics. There are exceptions. 132
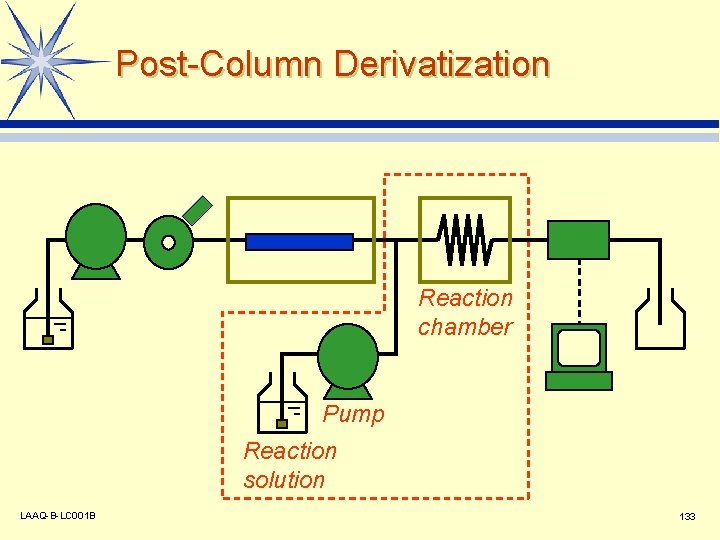
Post-Column Derivatization Reaction chamber Pump Reaction solution LAAQ-B-LC 001 B 133

Application Examples of Post. Column Methods l Amino Acids v v l l Arginine (fluorescence) v l LAAQ-B-LC 001 B Alkaline hydrolysis - OPA (fluorescence) l Tribromide ionization (ultraviolet absorption) o-Dianisidine (visible absorption) Cyanide Ions v Carbamate Pesticides v Bromate Ions v Orthophthalic acid, OPA (fluorescence) Ninhydrin (visible absorption) Reducing Sugars v l Chlorination - pyrazolone (visible absorption) Transition Metal Ions v 4 -(2 -Pyridylazo) resorcinol, PAR (visible absorption) 134

Quantitative Analysis Absolute Calibration Curve Method and Internal Standard Method LAAQ-B-LC 001 B 135

Qualitative Analysis l Identification based on retention time l Acquisition of spectra with detector v UV spectra v MS spectra l Transfer to other analytical instruments after preparative separation LAAQ-B-LC 001 B 136

Quantitative Analysis l Quantitation performed with peak area or height. l Calibration curve created beforehand using a standard. v Absolute calibration curve method v Internal standard method v Standard addition method LAAQ-B-LC 001 B 137
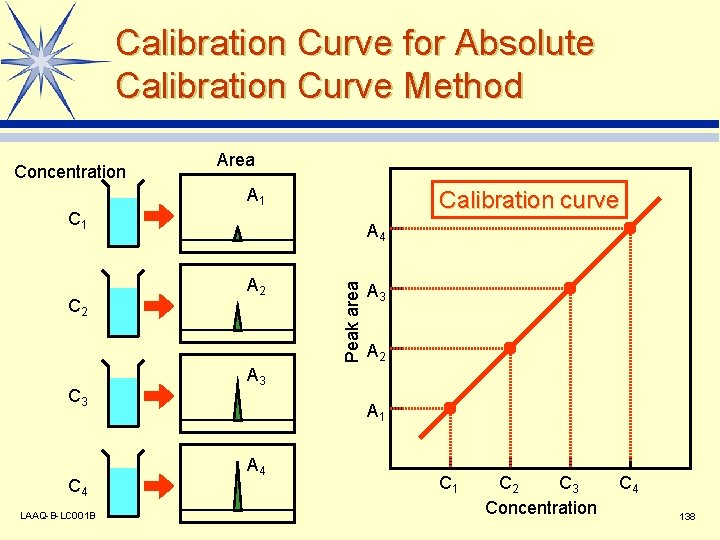
Calibration Curve for Absolute Calibration Curve Method Concentration Area A 1 Calibration curve C 1 C 3 C 4 LAAQ-B-LC 001 B A 2 Peak area C 2 A 4 A 3 A 2 A 3 A 1 A 4 C 1 C 2 C 3 Concentration C 4 138
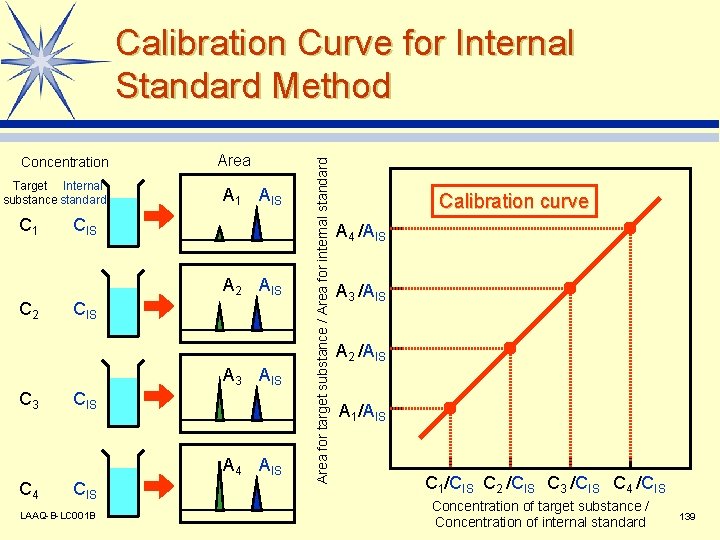
Concentration Target Internal substance standard C 1 Area A 1 AIS CIS A 2 AIS C 2 CIS A 3 AIS C 3 CIS A 4 AIS C 4 CIS LAAQ-B-LC 001 B Area for target substance / Area for internal standard Calibration Curve for Internal Standard Method Calibration curve A 4 /AIS A 3 /AIS A 2 /AIS A 1/AIS C 1/CIS C 2 /CIS C 3 /CIS C 4 /CIS Concentration of target substance / Concentration of internal standard 139
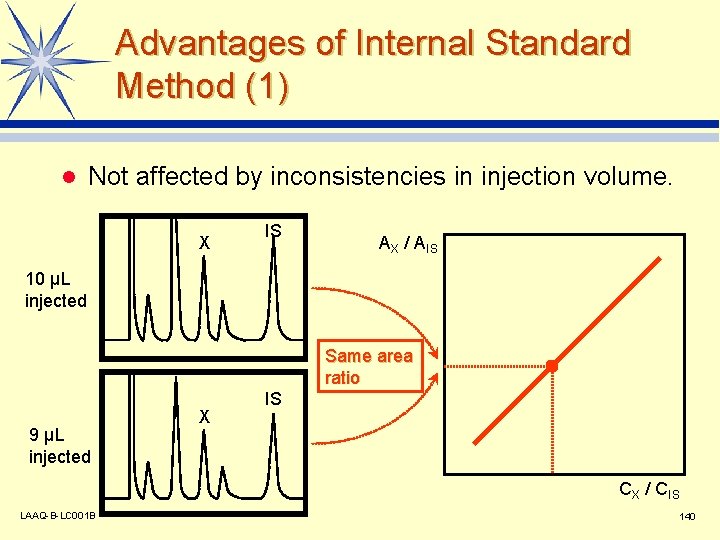
Advantages of Internal Standard Method (1) l Not affected by inconsistencies in injection volume. X IS AX / AIS 10 µL injected Same area ratio 9 µL injected X IS CX / CIS LAAQ-B-LC 001 B 140
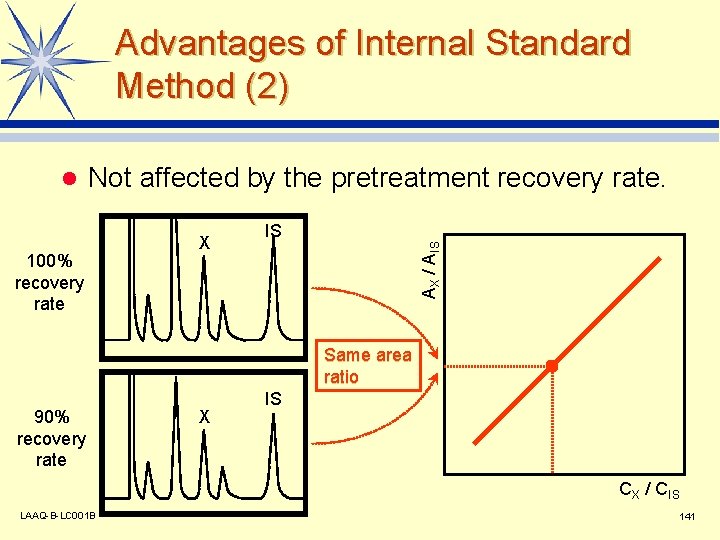
Advantages of Internal Standard Method (2) Not affected by the pretreatment recovery rate. 100% recovery rate X IS AX / AIS l Same area ratio 90% recovery rate X IS CX / CIS LAAQ-B-LC 001 B 141

Selection Criteria for Internal Standard l l l It must have similar chemical properties to the target substance. Its peak must appear relatively near that of the target substance. It must not already be contained in the actual samples. Its peak must be completely separated from those of other sample components. It must be chemically stable. LAAQ-B-LC 001 B 142

Sample Pretreatment Tasks Performed Before Injection LAAQ-B-LC 001 B 143

Objectives of Pretreatment To improve the accuracy of quantitative values l To improve sensitivity and selectivity l To protect and prevent the deterioration of columns and analytical instruments l To simplify measurement operations and procedures l To stabilize target substances l LAAQ-B-LC 001 B 144

Substances That Must Not Be Injected into the Column Insoluble substances (e. g. , microscopic particles and precipitation) l Substances that are precipitated in the eluent l Substances that irreversibly adsorb to the packing material l Substances that dissolve, or chemically react, with the packing material l LAAQ-B-LC 001 B 145

Filtration and Centrifugal Separation In general, filter every sample before injection! l It is convenient to use a disposable filter with a pore diameter of approx. 0. 45 µm. l Centrifugal separation is applicable for samples that are difficult to filter. l LAAQ-B-LC 001 B Filter Syringe 146

Deproteinization l Precipitation v Addition of organic solvent (e. g. , acetonitrile) v Addition of acid (e. g. , trichloroacetic acid, perchloric acid) v Addition of heavy metal or neutral salt l Ultrafiltration LAAQ-B-LC 001 B 147
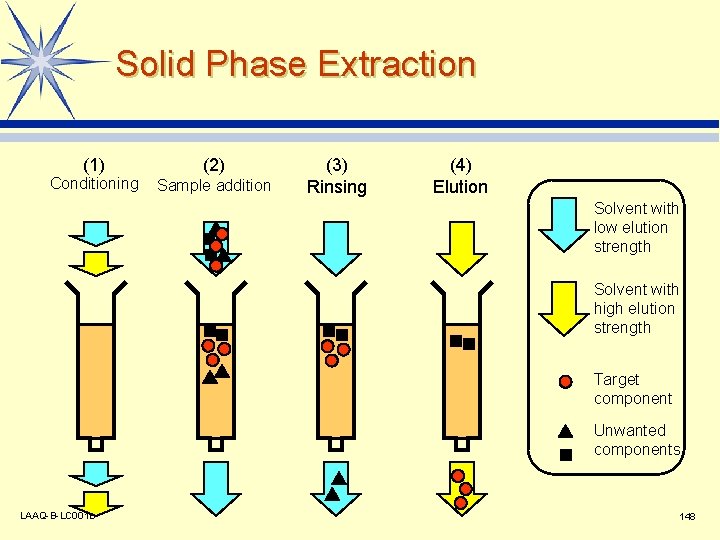
Solid Phase Extraction (1) Conditioning (2) Sample addition (3) Rinsing (4) Elution Solvent with low elution strength Solvent with high elution strength Target component Unwanted components LAAQ-B-LC 001 B 148
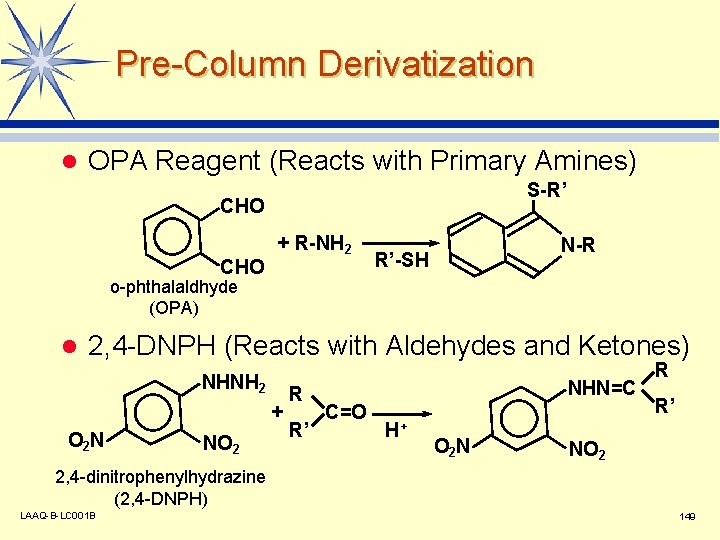
Pre-Column Derivatization l OPA Reagent (Reacts with Primary Amines) S-R’ CHO + R-NH 2 N-R R’-SH o-phthalaldhyde (OPA) l 2, 4 -DNPH (Reacts with Aldehydes and Ketones) NHNH 2 + O 2 N NO 2 R R’ NHN=C C=O H+ O 2 N R R’ NO 2 2, 4 -dinitrophenylhydrazine (2, 4 -DNPH) LAAQ-B-LC 001 B 149

Evaluation of the Reliability of Analysis Validation of Analytical Methods LAAQ-B-LC 001 B 150

What Is “Validation of Analytical Methods”? Scientifically demonstrating that the analytical methods concur with the intended purpose (i. e. , that errors are within a permissible range) l Evaluating required items from the validation characteristics l LAAQ-B-LC 001 B l Validation characteristics v v v v Accuracy / trueness Precision Specificity Detection limit Quantitation limit Linearity Range (Robustness) 151
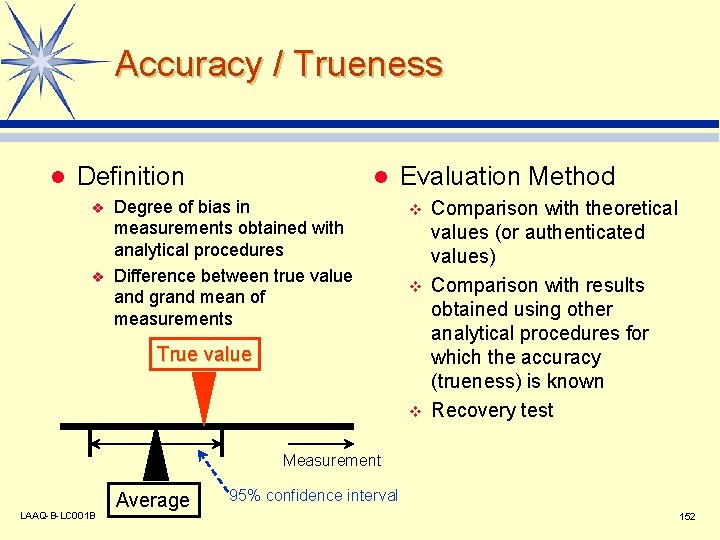
Accuracy / Trueness l Definition v v l Degree of bias in measurements obtained with analytical procedures Difference between true value and grand mean of measurements Evaluation Method v v True value v Comparison with theoretical values (or authenticated values) Comparison with results obtained using other analytical procedures for which the accuracy (trueness) is known Recovery test Measurement LAAQ-B-LC 001 B Average 95% confidence interval 152

Precision l Definition v v LAAQ-B-LC 001 B l Repeatability / Intra. Assay Precision Degree of coincidence of v Precision of series of measurements taken over obtained by repeatedly a short time period under analyzing multiple the same conditions samples taken from a l Intermediate Precision homogenous test substance l Reproducibility Variance, standard deviation, or relative standard deviation of measurements 153

Specificity l Definition v v v LAAQ-B-LC 001 B l The ability to accurately analyze the target substance in the presence of other expected substances The discrimination capability of the analytical methods Multiple analytical procedures may be combined in order to attain the required level of discrimination Evaluation Method v v Confirmation that the target substance can be discriminated (separated) from co-existing components, related substances, decomposition products, etc. If reference standards for impurities cannot be obtained, the measurement results for samples thought to contain the impurities are compared. 154

Detection Limit l Definition v v The minimum quantity of a target substance that can be detected. Quantitation is not absolutely necessary. l Evaluation Method v Calculated from the standard deviation of measurements and the slope of the calibration curve. v Calculated from the signal -to-noise ratio. LAAQ-B-LC 001 B DL = 3. 3 /slope ( : Standard deviation of measurements) (Slope: Slope of calibration curve) Concentration for which S/N = 3 or 2 155

Quantitation Limit l Definition v v LAAQ-B-LC 001 B The minimum quantity of a target substance that can be quantified Quantitation with an appropriate level of accuracy and precision must be possible. (In general, the relative standard deviation must not exceed 10%. ) l Evaluation Method v Calculated from the standard deviation of measurements and the slope of the calibration curve. v QL = 10 /slope ( : Standard deviation of measurements) (Slope: Slope of calibration curve) Calculated from the signal-tonoise ratio. Concentration for which S/N = 10 156

Linearity l Definition v v LAAQ-B-LC 001 B The ability of the analytical method to produce measurements for the quantity of a target substance that satisfy a linear relationship. Values produced by converting quantities or measurements of the target substance using a precisely defined formula may be used. l Evaluation Method v v Samples containing different quantities of the target substance (usually 5 concentrations) are analyzed repeatedly, and regression equations and correlation coefficients are obtained. Residuals obtained from the regression equations of the measurements are plotted, and it is confirmed that there is no specific slope. 157
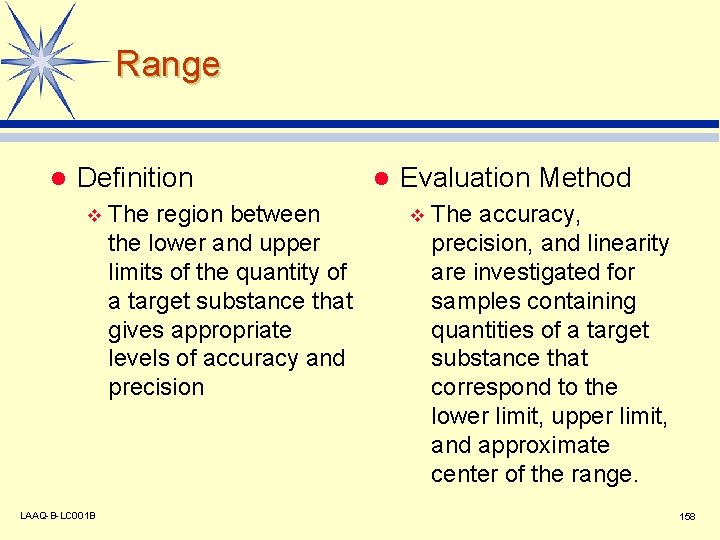
Range l Definition v LAAQ-B-LC 001 B The region between the lower and upper limits of the quantity of a target substance that gives appropriate levels of accuracy and precision l Evaluation Method v The accuracy, precision, and linearity are investigated for samples containing quantities of a target substance that correspond to the lower limit, upper limit, and approximate center of the range. 158

Robustness l Definition v LAAQ-B-LC 001 B The ability of an analytical procedure to remain unaffected by small changes in analytical conditions. l Evaluation Method v Some or all of the variable factors (i. e. , the analytical conditions) are changed and the effects are evaluated. 159

Maintenance of Separation Column Extending the Column’s Service Life LAAQ-B-LC 001 B 160
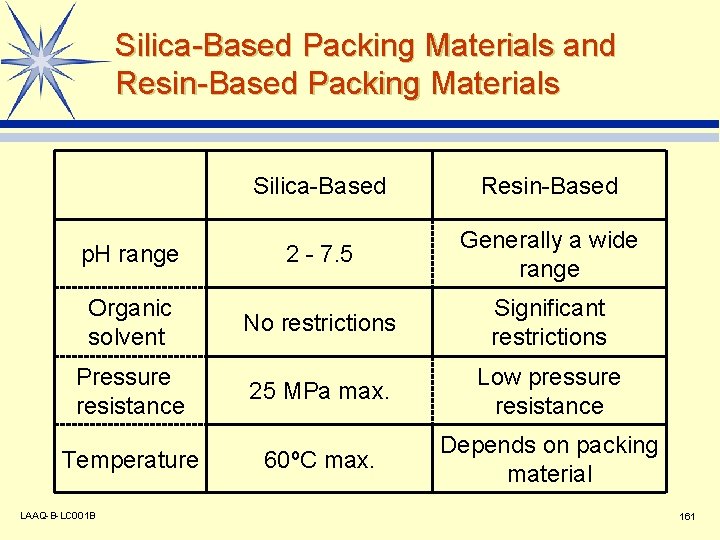
Silica-Based Packing Materials and Resin-Based Packing Materials Silica-Based Resin-Based p. H range 2 - 7. 5 Generally a wide range Organic solvent No restrictions Significant restrictions Pressure resistance 25 MPa max. Low pressure resistance Temperature 60ºC max. Depends on packing material LAAQ-B-LC 001 B 161

General Handling of Columns Observe restrictions l Use as low a load related to solvents and pressure as possible. the p. H range. v Do not exceed the upper l Never allow the pressure limit. packing material to dry. v Do not subject the column to sudden pressure l Do not allow solids or changes. microscopic particles to enter the column. l Do not subject the v Filter samples. column to intense shocks. l LAAQ-B-LC 001 B 162

Typical Problems (1) Column Clogging l Preventive Measures v v v Filter samples. Check that samples dissolve in the eluent. Get in the habit of observing pressure values. l Corrective Action v v LAAQ-B-LC 001 B Check for clogging in parts other than the column. Rinse with an appropriate solvent. Connect the column in reverse and flush out the insoluble substances at a low flow rate. Open the column end and perform ultrasonic cleaning of the filter. 163
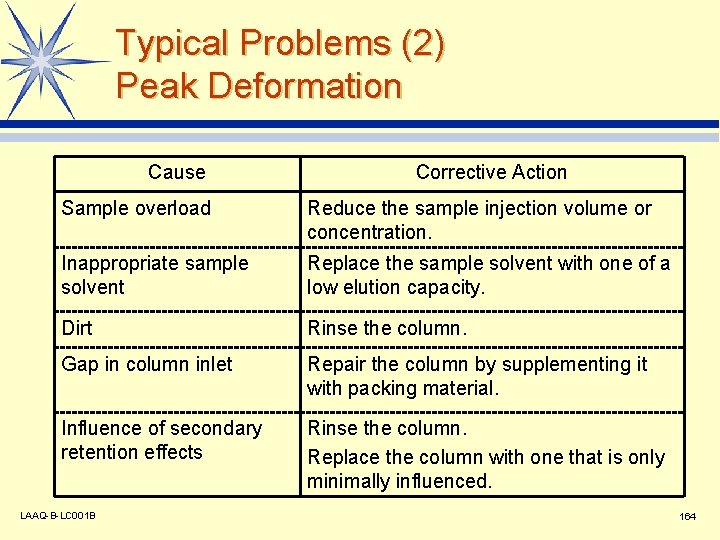
Typical Problems (2) Peak Deformation Cause Corrective Action Sample overload Reduce the sample injection volume or concentration. Inappropriate sample solvent Replace the sample solvent with one of a low elution capacity. Dirt Rinse the column. Gap in column inlet Repair the column by supplementing it with packing material. Influence of secondary retention effects Rinse the column. Replace the column with one that is only minimally influenced. LAAQ-B-LC 001 B 164

Typical Problems (3) Decrease in Retention Time l Check whether the cause of the problem is not the column. Eluent composition v Eluent flow rate v Column temperature v LAAQ-B-LC 001 B l If the column is identified as the cause. . . Rinsing v Replacement v 165

Typical Problems (4) Baseline Drift l Check whether the l If the column is cause of the problem is identified as the not the column. cause. . . v LAAQ-B-LC 001 B If the problem persists when the column is removed, it is caused by the eluent, the solvent delivery system (pump or degasser), or the detector. Rinsing v Review of temperature control v Replacement v 166
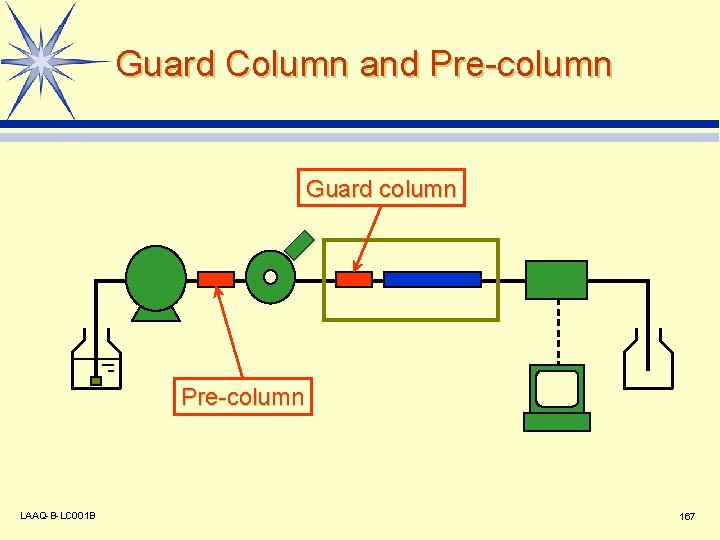
Guard Column and Pre-column Guard column Pre-column LAAQ-B-LC 001 B 167

Column Rinsing l Use an eluent with a high elution capacity v v l Reversed phase mode: Solution with a high proportion of organic solvent Ion exchange mode: Solution with a high salt concentration Consider secondary retention effects v v LAAQ-B-LC 001 B To remove basic substances from a reversed phase column Use an acidic solution and add an ion pair reagent. To remove hydrophobic substances from an ion exchange column Add an organic solvent. 168
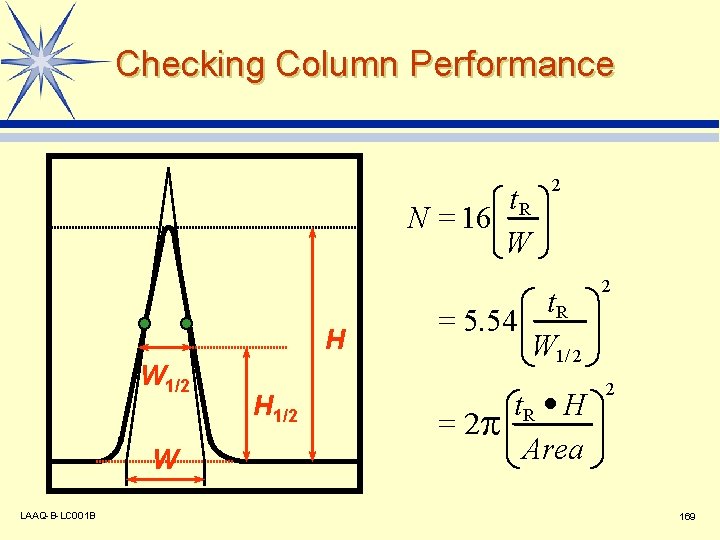
Checking Column Performance t. R N = 16 W H W 1/2 W LAAQ-B-LC 001 B H 1/2 2 t. R = 5. 54 W 1/ 2 2 t. R · H Area 2 = 2 169

Column Storage l Storage Solution v v It is generally safe to use the same storage solution as used at shipment. In order to prevent putrefaction, alcohol or some other preservative substance may be added. l Storage Conditions v v v LAAQ-B-LC 001 B Insert an airtight stopper in the column end. Never allow the packing material to dry. Make a record of the storage solution and final usage conditions and store it together with the column. Store the column in a location not subject to shocks or sudden temperature changes. 170
- Slides: 170