What is Density The ratio between the mass

What is Density? • The ratio between the mass of an object and the volume of the same object • In other words, it tells you how much matter an object has within a given amount of space (volume) • Each element has a specified density. For example: platinum = 21. 4 g/ml and silver = 10. 5 g/cm 3 • 1 ml = 1 cm 3

More on Density • Density is a ‘physical’ physical property of matter. It is a characteristic that applies to a specific substance, such as odor, color, and texture. • The density of a pure substance never changes, except for gases. • Physical properties that do not depend on ‘how much’ you have of the substance are called intensive properties, such as density • A physical property that depend on the amount of substance is called an extensive property.

Definitions • Mass = how much matter a substance has • Volume = how much space the substance takes up • Density = how packed the matter is

Determining Density Take the mass of the object and divide by the volume Mass volume Example: An object with a mass of 20 grams and a volume of 10 milliliters has a density of 2 g/ml. 20 g = 20 g ÷ 10 ml = 2 g/ml 10 ml The answer 2 g/ml is read 2 grams per one milliliter.
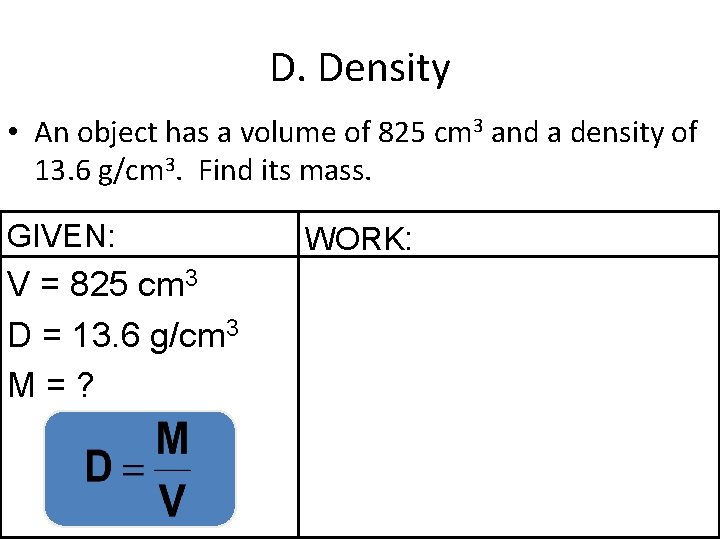
D. Density • An object has a volume of 825 cm 3 and a density of 13. 6 g/cm 3. Find its mass. GIVEN: V = 825 cm 3 D = 13. 6 g/cm 3 M=? WORK:
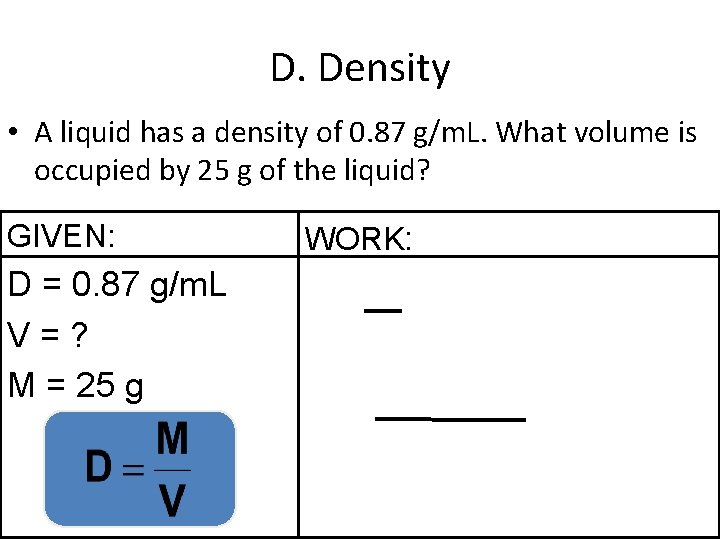
D. Density • A liquid has a density of 0. 87 g/m. L. What volume is occupied by 25 g of the liquid? GIVEN: D = 0. 87 g/m. L V=? M = 25 g WORK:

Calculate 1. Mass= 4. 53 kg and volume= 225 cm 3 2. Mass= 122. 4 g and volume=5. 5 ml 3. Find the mass if the density is 2. 14 g/cm 3 and the volume is 8 cm 3 2. 14 g = 1 cm 3
- Slides: 7