What is a covalent bond A bond formed
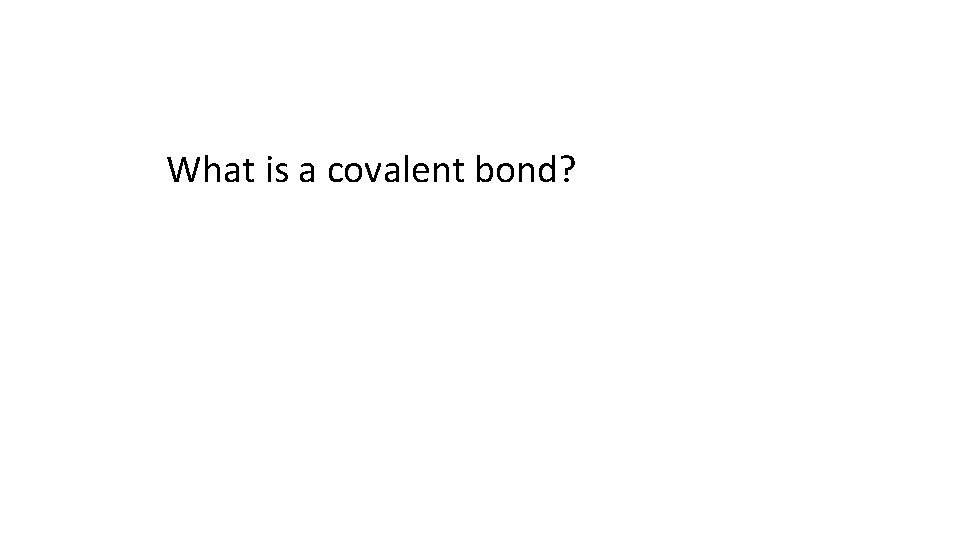
What is a covalent bond?

A bond formed by sharing a pair of electrons between two non metals attain a Nobel gas structure

Draw a dot cross diagram to illustrate the bonding in H 2
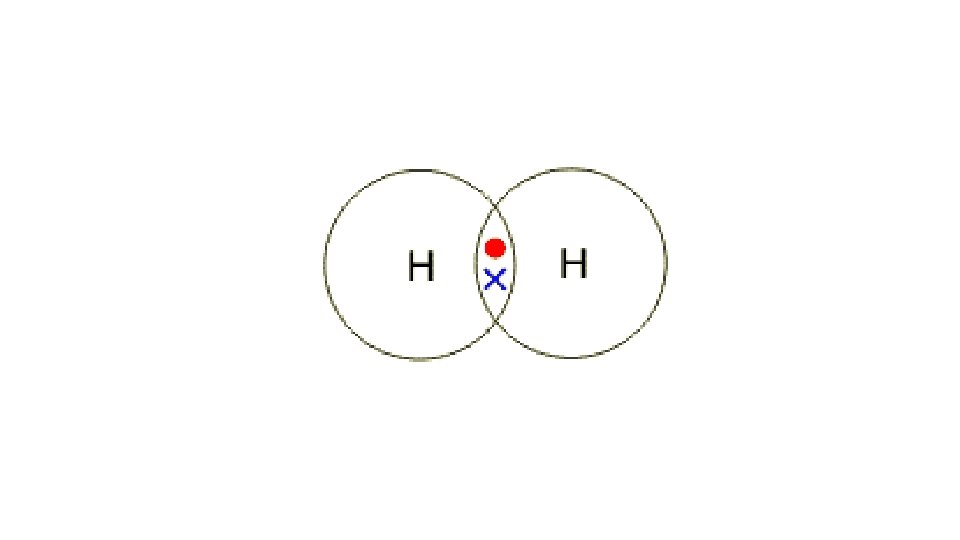

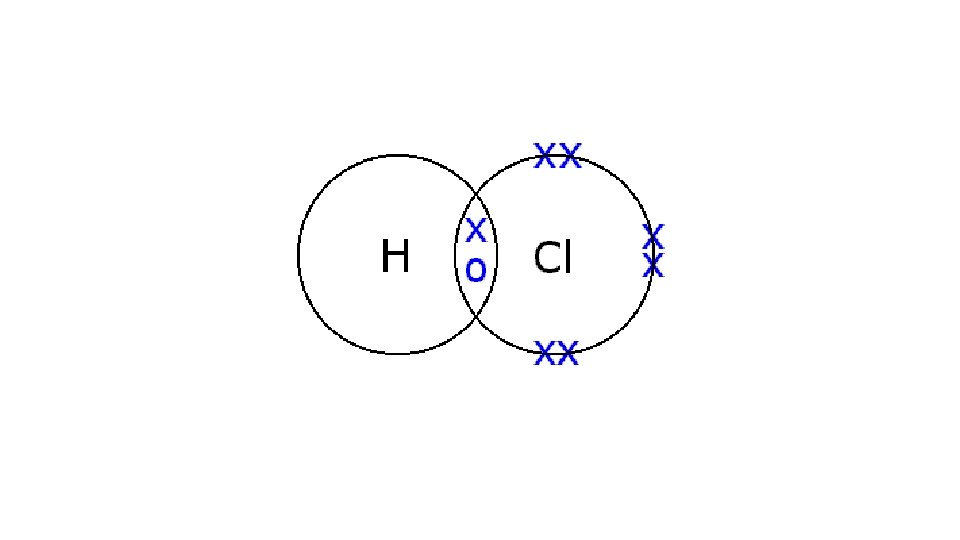
Draw a dot cross diagram to illustrate the bonding in Cl 2
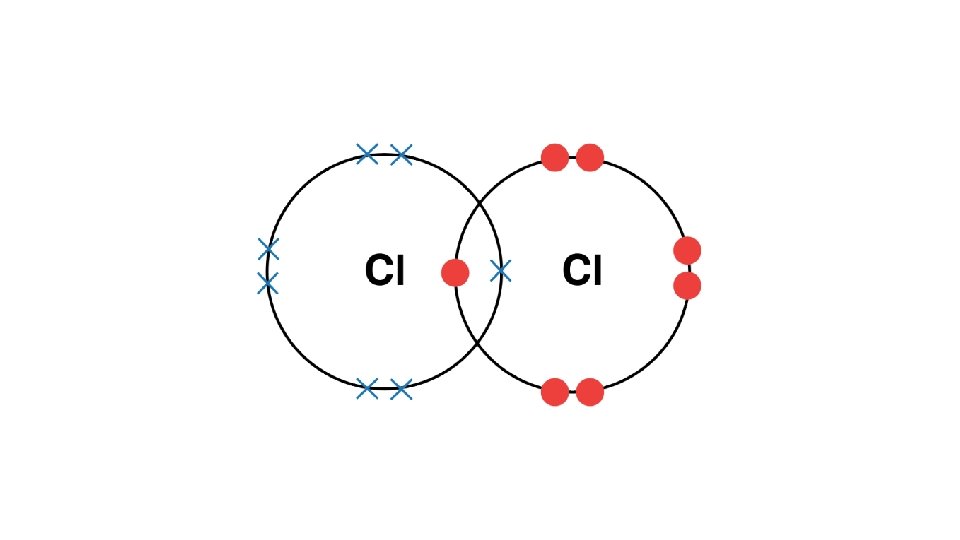

Draw a dot cross diagram to illustrate the bonding in H 2 O
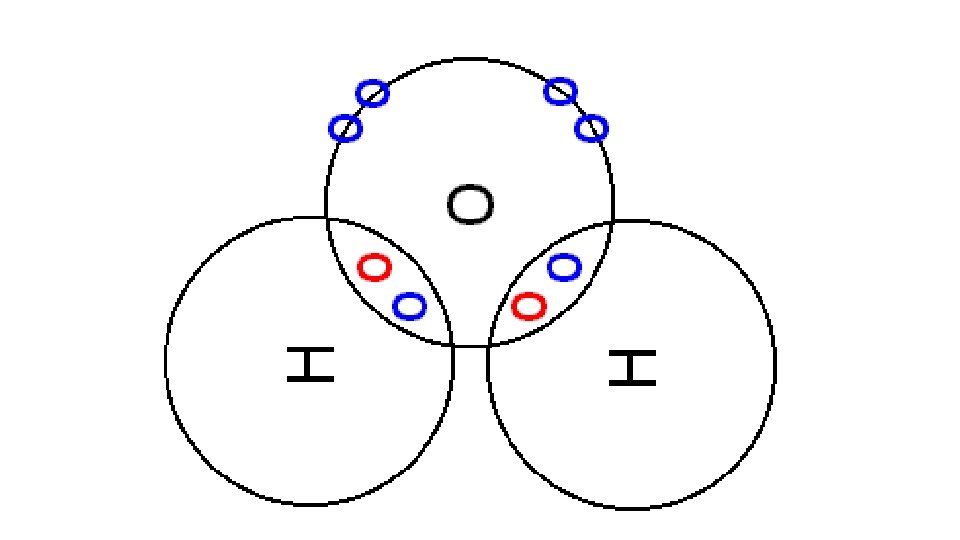

Draw a dot cross diagram to illustrate the bonding in CH 4
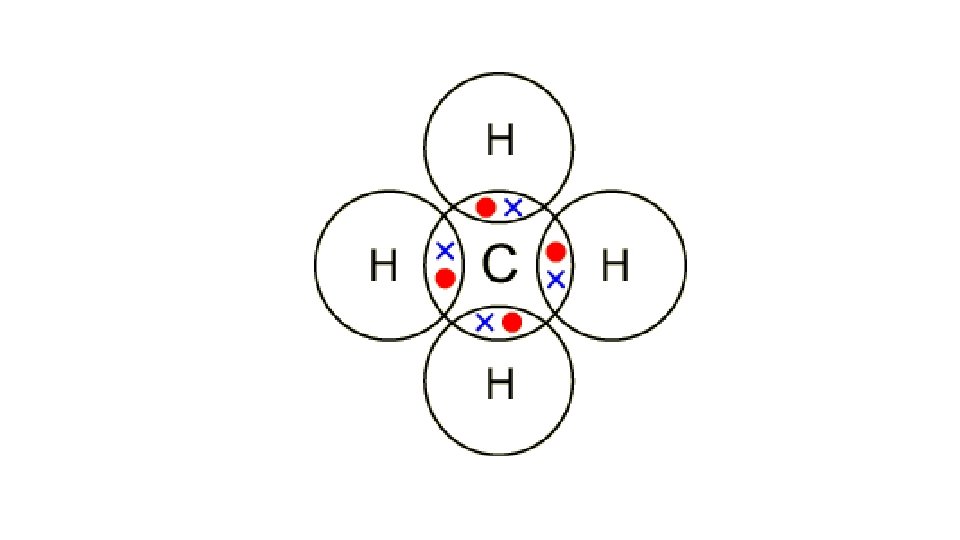

Draw a dot cross diagram to illustrate the bonding in NH 3
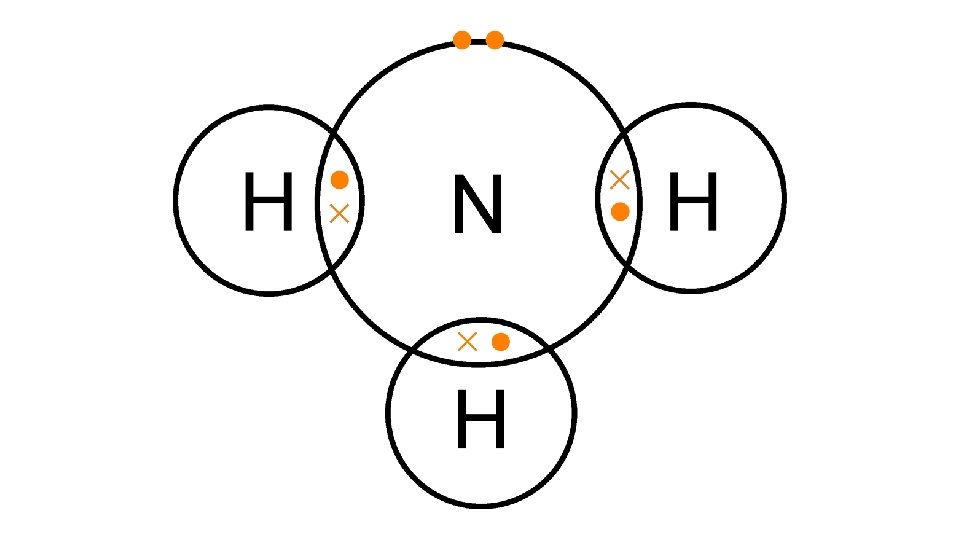

Draw a dot cross diagram to illustrate the bonding in HCl

Describe the differences in volatility, solubility and electrical conductivity between ionic and covalent compounds
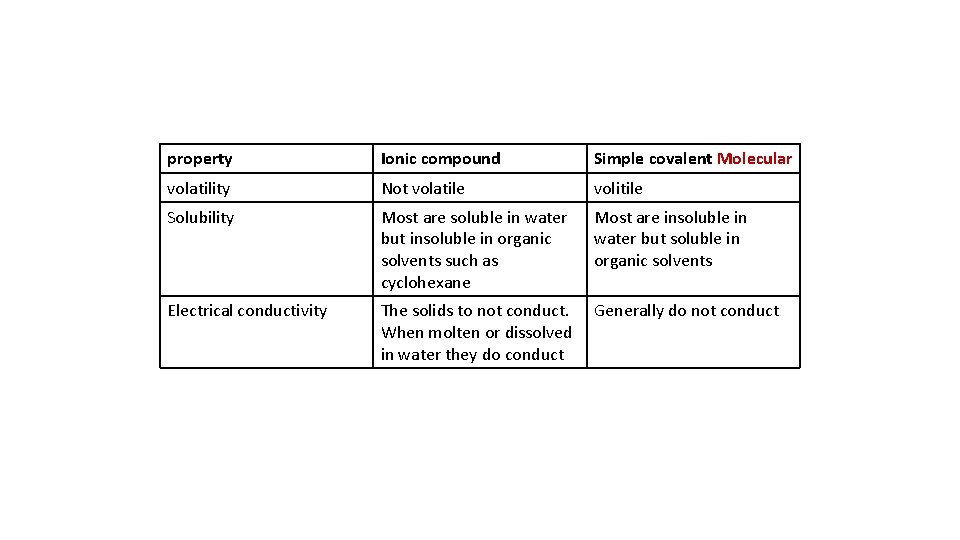
property Ionic compound Simple covalent Molecular volatility Not volatile volitile Solubility Most are soluble in water but insoluble in organic solvents such as cyclohexane Most are insoluble in water but soluble in organic solvents Electrical conductivity The solids to not conduct. When molten or dissolved in water they do conduct Generally do not conduct

Draw a dot cross diagram to illustrate the bonding in N 2

Draw a dot cross diagram to illustrate the bonding in C 2 H 4

Draw a dot cross diagram to illustrate the bonding in CH 3 OH

Draw a dot cross diagram to illustrate the bonding in CO 2

Explain the differences in melting point and boiling point of ionic and covalent compounds in terms of attractive forces

• Because there are only weak forces of attraction between simple covalent molecules, they can easily beak free of the solid structure, so melting points are low. • Particles in the liquid are only weakly held together so boiling points are also low • In ionic compounds melting points are high because of the strong attraction between ions

Explain the term ionic lattice

• In solids ions are regularly arranged in a lattice of alternating positive and negative ions. • Ionic compounds are said to have giant ionic structure

What are giant covalent structures

Macromolecules are giant covalent structures where many atoms are held together by strong covalent bonds, unlike simple covalent molecules such as chlorine and water

Describe the giant covalent structures in diamond and its uses

• Diamond is a giant macromolecule , it is a form of carbon in which every carbon atom is bonded to four other carbon atoms by strong covalent bonds • Because of its giant structure and strong covalent bonds, diamond: 1. Is the hardest natural substance 2. Has a high melting point 3. Does not conduct electricity • It is used in cutting tools and drill bits

Describe the giant covalent structures in graphite and its uses

• Graphite is another form of carbon • Each carbon atom is bonded by three strong covalent bonds to make a giant structure consisting of layers of hexagons • The layers can slide over each other because there are no covalent bonds between them, just weak forces of attraction between the layers • This makes graphite soft and a good lubricant • Graphite conducts electricity because the fourth electron of every carbon atom is unbounded and moves along the sea of delocalised electrons
Describe the macromolecular structure of silicon(IV) oxide (silicon dioxide)

• Silicon oxide has a structure similar to diamond, except instead of carbon atoms, there alternating silicon and oxygen atoms • This means it has similar properties to diamond

Describe the similarity in properties between diamond and silicon(IV) oxide related to their structures

Hard High melting point
Describe metallic bonding
Describe metallic bonding

Metals form a lattice of positive ions in a ‘sea of electrons’

Why are metals good conductors of electricity?

Why do metals have high melting points

• Strong electrostatic forces • Between positive ions and negative electrons • Require a lot of energy to overcome

Many metals conduct electricity because the electrons are free to move in the delocalised sea of electrons
Why are metals malleable

They are malleable because the metals ions are arranged in layers which can slide over each other
- Slides: 43