What happens to thermal heat energy When objects
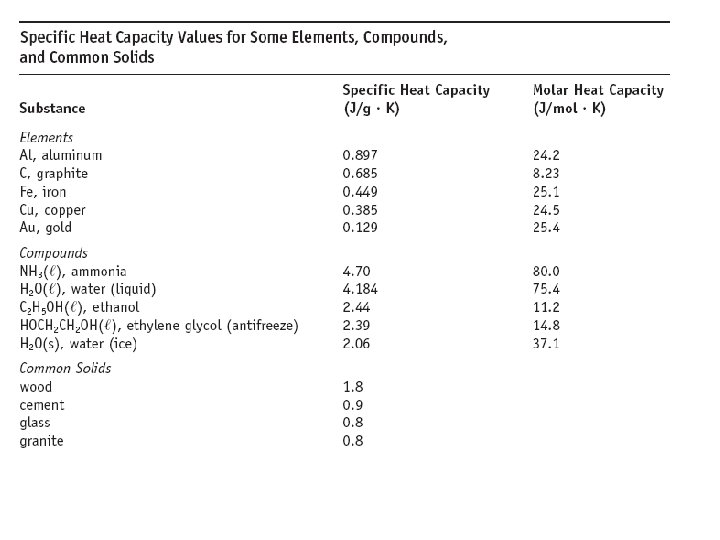

What happens to thermal (heat) energy? When objects of different temperature meet: • Warmer object cools • Cooler object warms • Thermal energy is transferred • qwarmer = -qcooler specific heat x mass x T = specific heat x mass x T warmer object cooler object

Heat transfer between substances:

Thermal Energy and Phase Changes First: What happens?

But what’s really happening? Warming: • Molecules move more rapidly • Kinetic Energy increases • Temperature increases Melting/Boiling: • Molecules do NOT move more rapidly • Temperature remains constant • Intermolecular bonds are broken • Chemical potential energy (enthalpy) increases

Energy and Phase Changes: Quantitative Treatment Melting: Heat of Fusion (DHfus) for Water: 333 J/g Boiling: Heat of Vaporization (DHvap) for Water: 2256 J/g

Total Quantitative Analysis Convert 40. 0 g of ice at – 30 o. C to steam at 125 o. C Warm ice: (Specific heat = 2. 06 J/g-o. C) Melt ice: Warm water (s. h. = 4. 18 J/g-o. C)

Total Quantitative Analysis Convert 40. 0 g of ice at – 30 o. C to steam at 125 o. C Boil water: Warm steam (s. h. = 1. 92 J/g-o. C)

Enthalpy Change and Bond Energies H = energy needed to break bonds – energy released forming bonds Example: formation of water: H = [498 + (2 x 436)] – [4 x 436] k. J = -482 k. J
- Slides: 9