What Happened in Activity 3 Solid Aluminum was
































- Slides: 37


What Happened in Activity 3? Solid Aluminum was added to a solution of Copper (II) chloride Al(s) + Cu. Cl 2(aq) → ? Name a physical change that occurred? ? ? How do you know it was a physical change? ? ? Name a chemical change that occurred? ? How do you know it was a chemical change? ? ?

Activity Series of Metals Decreasing Activity Li K Ba Sr Ca Na Mg Al Mn Zn Al(s) + Cu. Cl 2(aq) → Al. Cl 3(aq) + Cu(s) Fe Cd Co Ni Sn Pb H Cu Ag Hg Au

What do you see?

Law of Conservation of Matter can not be created or destroyed! Matter can only change forms!

Physical change The material itself is the same before and after the change. The change can usually be “undone. ” Ice melting: an example of a physical change.

Physical Change You still have bronze! Melting bronze into a liquid

Physical Change Ripping Paper But this one can’t be undone so easily.

Origami The form/shape may change, but you still have paper

Cracking an Egg Another example of an irreversible physical change. Beating an Egg

Ghost Crystals (crosslinked polyacrylamide) + water Physical change!! Given time the water will evaporate away and leave the crystals

Sodium Polyacrylate Theses crystals are If left to dry long found in the "super enough, it would absorbent layer" in return to its original most disposable state or appearance. diapers. It can absorb 200 to 300 times its weight in tap water

Physical Change Review 1. Only the state of matter changes 2. All Phase changes are physical changes (Solid---Liquid---Gas)! 3. NO new substances are formed! 4. Not all physical changes are reversible (crushing a rock).

Chemical Change The substances present at the beginning of the change are not present at the end; new substances are formed. The change cannot be “undone. ” A nail rusting: an example of chemical change.

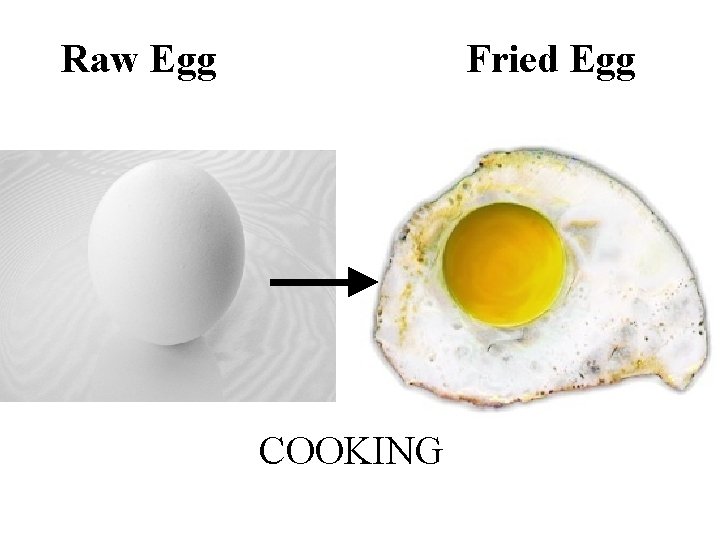
Raw Egg Fried Egg COOKING

Chemical Change! The proteins in the egg have changed! The cooked egg cannot change back to a raw egg.


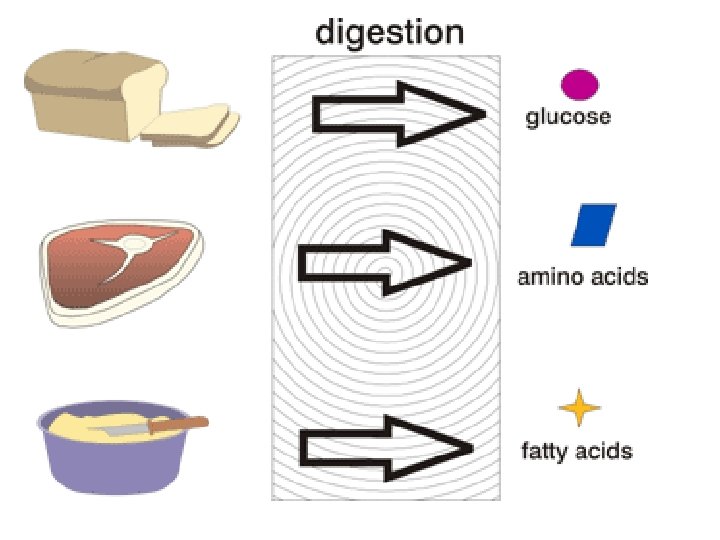
Chemical Reaction! Your body chemically reacts with the food you eat to make nutrients and energy!

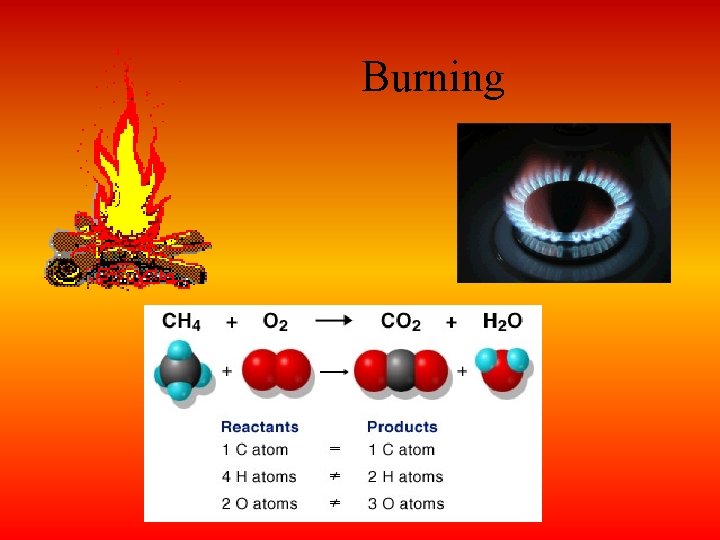
Burning

Chemical Change! Rust is a new substance! (color change) Car with rust =

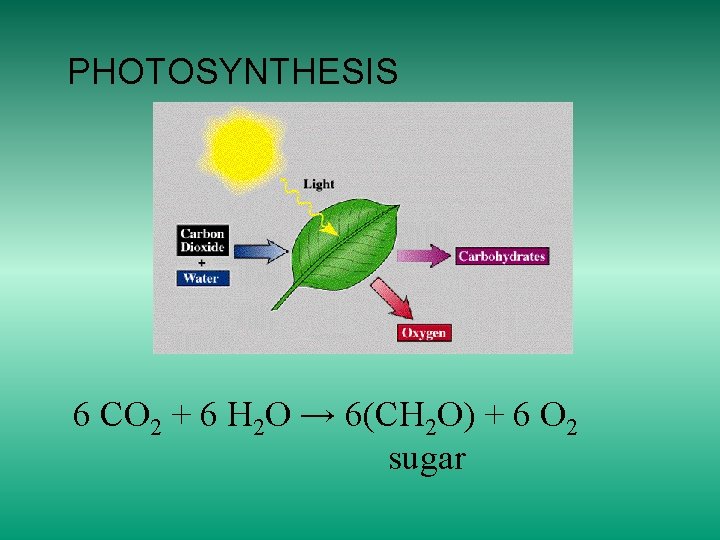
PHOTOSYNTHESIS 6 CO 2 + 6 H 2 O → 6(CH 2 O) + 6 O 2 sugar

Evidence of a Chemical Change • Color Change • Bubbling (formation of a gas) • Light given off • Temperature change • Precipitate formed. • Sound

Chemical Reaction! Formation of a Precipitate lead iodide (Pb. I 2), forms from the mixture of two clear liquids, potassium iodide (KI) and lead nitrate (Pb(NO 3)2).

Chemical Change Review 1. A chemical reaction occurs 2. New substance(s) are formed with NEW properties 3. It can not be reversed!


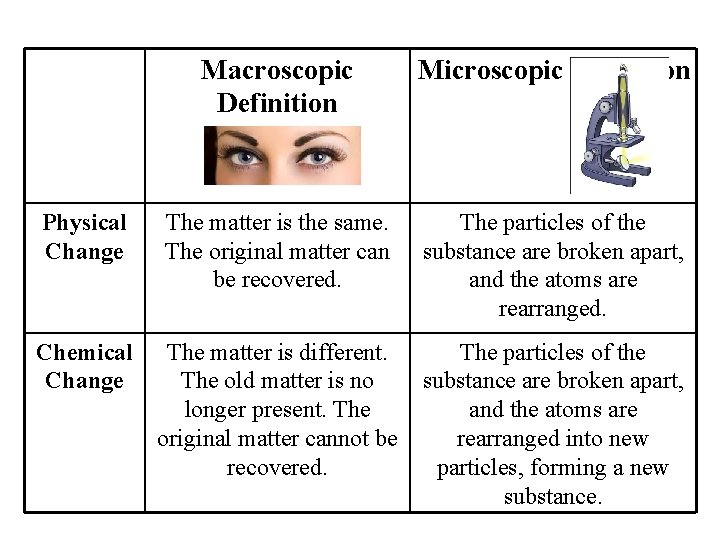
Physical Change Chemical Change Macroscopic Definition Microscopic Definition The matter is the same. The original matter can be recovered. The particles of the substance are broken apart, and the atoms are rearranged. The matter is different. The particles of the The old matter is no substance are broken apart, longer present. The and the atoms are original matter cannot be rearranged into new recovered. particles, forming a new substance.

Physical vs. Chemical • Examples: – melting point physical – flammable chemical – density physical – magnetic physical – tarnishes in air chemical

Physical vs. Chemical • http: //vital. cs. ohiou. edu/steamwebsite/downlo ads/Change. Lab. swf • http: //www. quia. com/quiz/303980. html

Yellow & Blue Switcheroo

Yellow & Blue Switcheroo Oscillating Reaction Chemical! Color Change indicates the chemical properties of the indicator were changed! New product formed! Can’t be reversed!

Pour a Rainbow

Acids to a Base Reactions Color change & Polymerization A Chemical Change Occurs!! We have now made our own slime!

Old Foamy

Old Foamy

Chemical Change Old Foamy Hydrogen Peroxide & Sodium Iodide Solution With Dishwashing Detergent New product formed! Can’t be reversed!

Whoosh Bottle

Chemical Change Exothermic Reaction: O 2 + Combustion CO 2 & H 2 O Color & Heat released New product formed! Can’t be reversed!