What factors determine the types of chemical bonds

What factors determine the types of chemical bonds that form? Chemistry

What factors determine the types of chemical bonds that form? Day 1 Introduction

Why do chemical bonds form? Stability in Bonding Notes A. Some elements combine chemically and no longer have the same properties they did before forming the compound.
Why do chemical bonds form? B. A chemical formula is composed of symbols and subscripts indicating the number of atoms of an element in a compound. Ex. H 2 O

Why do chemical bonds form? Compounds C. Atoms form compounds when the compound is more stable than the separate atoms

Why do chemical bonds form? Compounds l l Noble gases are more chemically stable b/c they have a complete outer energy level. Stable elements have 8 electrons in their outer energy level.
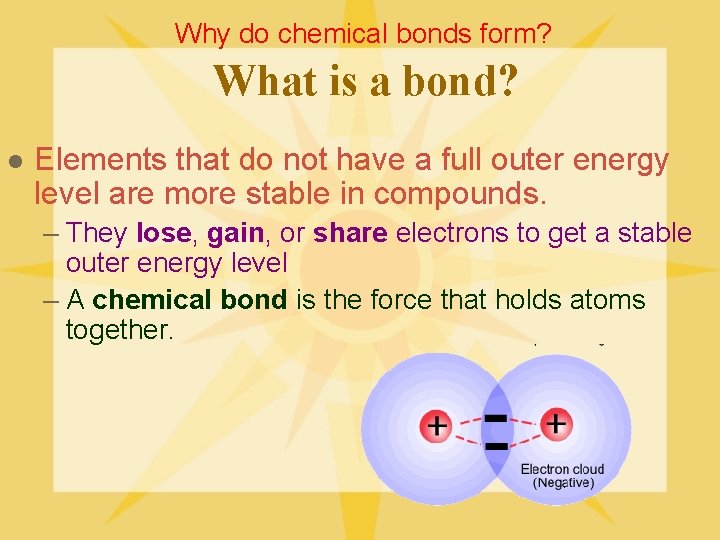
Why do chemical bonds form? What is a bond? l Elements that do not have a full outer energy level are more stable in compounds. – They lose, gain, or share electrons to get a stable outer energy level – A chemical bond is the force that holds atoms together.

What factors determine the types of chemical bonds that form? Day 2 Daily

How are ionic and covalent bonds different? Let’s reflect on yesterdays lab: l Baking soda + Vinegar Na. HCO 3 + HC 2 H 3 O 2 --> H 2 O + CO 2 + Na. C 2 H 3 O 2 l Copper chloride + Water Cu. Cl 2 + H 2 O --> _______ Why is the first one a chemical reaction? ? ?

How are ionic and covalent bonds different? What is an Ion? l A charged particle that has more or less electrons than protons. – If an atom loses an electron -- POSITIVE ION – If an atom gains an electron -- NEGATIVE ION l Superscripts indicate these changes (O 2 -) Ex: – Potassium loses an electron so the ion is K 1+ – Iodine gains an electron so ion the is I 1 -

How are ionic and covalent bonds different? Ions combine in Ionic Bonds l When ions bond together, they form compounds. – The result of the ionic bond is a neutral compound. (no charge) – Usually formed by bonding of metals and nonmetals

How are ionic and covalent bonds different? Ionic Bond Example l Mg. Cl 2 – Mg loses 2 electrons (Mg 2+) – Cl gains 1 electron (Cl 1 -) Draw Mg. Cl 2

How are ionic and covalent bonds different? Covalent Bonds l Molecules are neutral particles formed when electrons are shared. – Single bond - 2 shared electrons (H 2 O) – Multiple bond - 4 or 6 electrons shared (N 2) – Formed by nonmetals and nonmetals

How are ionic and covalent bonds different? Dot Diagrams l l H 2 O Na. Cl

What factors determine the types of chemical bonds that form? Day 3 Daily

EQ: How do you balance a chemical equation? Why do chemical equations need balancing? l l Make H 2 O using the atoms from the kit. Yellow is hydrogen and red is oxygen. Make an H 2 and an O 2. Does H 2 + O 2 = H 2 O? Explain Make a balanced equation using the model…. extra points for the group that gets it first.

What factors determine the types of chemical bonds that form? Day 4 Daily

How do you balance chemical equations? Parts of a Chemical Equation Cl 2 + 2 Na. Br → Br 2 + 2 Na. Cl l l l Chemical Symbols - Represent the elements involved Chemical Formulas - Represent the compounds involved. Subscripts - Identify the number of atoms of each element in a chemical formula or symbol. Reactants - Present before the reaction Products - Produced in the reaction Coefficients - the numbers in front of the chemical formulas or symbols that are used to balance the equation. Arrow - means “yields”, “produces”, or “react to produce”

What factors determine the types of chemical bonds that form? Day 5 Daily
What information can you obtain from a chemical equation? Balance and Reaction Type l __ Na + __ Cl 2 → __ Na. Cl l __ Na. Cl → __ Na + __ Cl 2 l __ HCl + __ Na. OH → __ HOH + __ Na. Cl l __ Cl 2 + __ Na. Br → __ Br 2 + __ Na. Cl l __ C 2 H 6 + __ O 2 → __ CO 2 + __ H 2 O

What information can you obtain from a chemical equation? Balance and Identify the following Chemical Reactions l 2 Na + Cl 2 → 2 Na. Cl l 2 Na. Cl → 2 Na + Cl 2 l HCl + Na. OH → HOH + Na. Cl l Cl 2 + 2 Na. Br → Br 2 + 2 Na. Cl l 2 C 2 H 6 + 7 O 2 → 4 CO 2 + 6 H 2 O

What information can you obtain from a chemical equation? Law of Conservation of Mass Matter is neither created nor destroyed in a chemical reaction! The mass of the reactants equals the mass of the products

How do you identify the type of chemical reaction? Decomposition Reaction aka “The Don Massey - ‘I think it is time I should be moving on’” reaction / The Break up Reaction The Opposite of a Synthesis Reaction AB → A + B compound → 2 elements or compounds • • • H 2 O → H 2 + O 2 Na. OH → Na 2 O + H 2 O Na. Cl. O 3 → O 2 + Na. Cl

How do you identify the type of chemical reaction? Synthesis Reaction aka “The Don Massey - ‘I think you & I should start dating’” reaction / The Dating Reaction A + B → AB 2 elements or compounds → compound – H 2 + O 2 → H 2 O

How do you identify the type of chemical reaction? Single Displacement Reaction aka “The Don Massey - ‘I’m gonna sweet talk you out of dating your boyfriend’” reaction A+ BC → B + AC A + BC → C + BA An element replaces an element in a compound • Zn + 2 HCl → H 2 + Zn. Cl 2 • F 2 + 2 HCl → Cl 2 + 2 HF

How do you identify the type of chemical reaction? Combustion Reaction aka “The Don Massey - ‘I’m gonna light your world on fire’” reaction C? H? + O 2 → CO 2 + H 2 O An organic substance + O 2 → CO 2 + H 2 O l C 3 H 8 + O 2 → CO 2 + H 2 O

How do you identify the type of chemical reaction? Double Displacement Reaction aka “The Don Massey ‘I’ve got to make happy all the ladies in this room. ’” reaction AB + CD → CB + AD 2 compounds → 2 Compounds • K 2 Cr. O 4 + Pb(NO 3)2 → 2 KNO 3 + Pb. Cr. O 4
- Slides: 27