What do you mean by a state function

What do you mean by a state function. Define intensive properties, isolated system, enthalpy. Show relationship between E and H, G, H and S. Distinguish between exothermic and endothermic reactions. Show equilibrium state graphically. When is the equilibrium of a reaction achieved? Does the concentrations of the reactants and products remain equal at equilibrium? Give an example of a reaction of a & b moles of the reactants A & B yielding c & d moles products C & D respectively, in equilibrium. Show the value of K in the reaction. Does pressure affect equilibrium without any gas in the system? In which direction will a reaction move if its reaction quotient is less than its equilibrium constant (Q < K eq)? Show the conjugate acid and base pairs in the following ionization : HCN(l) + H 2 O CN–(aq) + H 3 O+(aq). Define p. H and p. OH. How could you measure p. H of a solution ? In which range of p. H a solution is said to be acidic? Define Kw
![Explain i) Distribution law, ii) The p. H of a solution where [H+]=[HO-] and Explain i) Distribution law, ii) The p. H of a solution where [H+]=[HO-] and](http://slidetodoc.com/presentation_image_h2/4f786da044acbe18219f3bc0b0bc88ec/image-2.jpg)
Explain i) Distribution law, ii) The p. H of a solution where [H+]=[HO-] and iii) common ion effect. Explain saturated solution and solubility product. Explain i) Dissociation constant, ii) Degree of dissociation, iii) Weak electrolyte and iv) Electrophoretic effect. Give an example for each of an endothermic and exothermic reaction. Show that enthalpy is a state function. Distinguish between specific heat and heat capacity. Describe i) Partition coefficient, ii) Value of Ka for a moderate acid and a weak acid. Explain ionic atmosphere and relaxation effect. State Ostwald’s dilution law. What is a buffer solution? Describe Law of mass action and explain Le Chattelier’s principle.

Indicate the correct form of statement in each of the following: Due to common ion effect the degree of dissociation of a salt is increased / suppressed. There will be precipitation / no precipitation when Q > Ksp. An acid can accept / donate proton. In an exothermic reaction Hproducts <Hreactants / Hproducts >Hreactants / Hproducts =Hreactants. Volume of a system is its intensive / extensive / chemical property. Kw at 250 C is 10 -14 / 10 -7 / 7. In a system at constant V, DH=q / DH < q p. H of an acid will be > / = / < that of a base. At equilibrium, reaction is in static / dynamic condition. In an spontaneous reaction DG < 0 / DG > 0 / DG = 0. At equilibrium Keq > Q / Keq = Q / Keq < Q. Reluxation effect and asymmetric effect are same / different A closed system can accept mass / energy / both. Solubility product and ion product is the same for a saturated /unsaturated/ dilute solution.

Indicate the correct form of statement in each of the following: Due to common ion effect the degree of dissociation of a salt is increased / suppressed. There will be precipitation / no precipitation when Q > Ksp. An acid can accept / donate proton. In an exothermic reaction Hproducts <Hreactants / Hproducts >Hreactants / Hproducts =Hreactants. Volume of a system is its intensive / extensive property. Kw at 250 C is 10 -14 / 10 -7 / 7. In a system at constant V, DH=q / DH < q p. H of an acid will be > / = / < that of a base. At equilibrium, reaction is in static / dynamic condition. In an spontaneous reaction DG < 0 / DG > 0 / DG = 0. At equilibrium Keq > Q / Keq = Q / Keq < Q. Reluxation effect / asymmetric effect are same / different A closed system can accept mass / energy / both. Solubility product and ion product is the same for a saturated /unsaturated/ dilute solution.

For 5 gm of a substance of specific heat S its heat capacity is S / 5 S /S 5. Conjugate base of HCl is a weak / strong base. Endothermic reaction is an example of a spontaneous /nonspontaneous process. At equilibrium concentrations of the reactants and products are equal /unequal /irrelevant. Equilibrium of an exothermic reaction shifts toward products / reactants /none. Acids with Ka > 1 are suitable / not applicable in buffer solution. p. H + p. OH = 14 / 10 -7 / 7 H 2 O is conjugate base of OH-/ H 3 O+ / acetic acid.

For 5 gm of a substance of specific heat S its heat capacity is S / 5 S /S 5. Conjugate base of HCl is a weak / strong base. Endothermic reaction is an example of a spontaneous /nonspontaneous process. At equilibrium concentrations of the reactants and products are equal/unequal/irrelevant. Equilibrium of an exothermic reaction shifts toward products / reactants /none. Acids with Ka > 1 are suitable / not applicable in buffer solution. p. H + p. OH = 14 / 10 -7 / 7 H 2 O is conjugate base of OH-/ H 3 O+ / acetic acid.

What do you mean by a state function. Define intensive properties, isolated system, enthalpy. Show relationship between E and H, G, H and S. State functions are thermodynamic functions, independent of time factor, rate of change, path of change, depends only on the initial & final states of the system. H, S, E, G, these are state functions. Intensive properties: (P, T, ρ) These are intrinsic properties, independent of quantity of the substance. Extensive properties: (V, moles no. H, S, E, G) Changes with change in quantity. System is the specific part of the universe that is of interest in the study. In an isolated system entry or exit of both mass and energy is restricted. (E): At constant volume (say in a sealed tube), the heat content of a system is its internal energy. The total heat content of a system at constant pressure is equivalent to the internal energy E plus the PV energy. This is called the enthalpy H of the system. Thus enthalpy is defined by the equation: H= E + PV (relationship between E and H)

State Hess’s law and show that enthalpy is a state function. Hess’s law : For any chemical change made in several steps, the net DH is equal to the sum of the DH values of the separate steps. The path taken from reactants to products for DH is irrelevant. The rate of reaction for DH is also irrelevant. So enthalpy is a state function. C(s) + O 2(g) ® C(s) + 2 O(g) DH = +498 k. J/mol C(s) + 2 O(g) ® CO 2(g) DH = -891 k. J/mol C(s) + O 2(g) ® CO 2(g) DH = -393 k. J/mol

Relationship between G, H and S. For a constant-temperature process: Gibbs free energy (G) DG < 0 DG = DHsys -TDSsys The reaction is spontaneous in the forward direction. DG > 0 The reaction is spontaneous in the reverse direction. DG = 0 The reaction is at equilibrium.
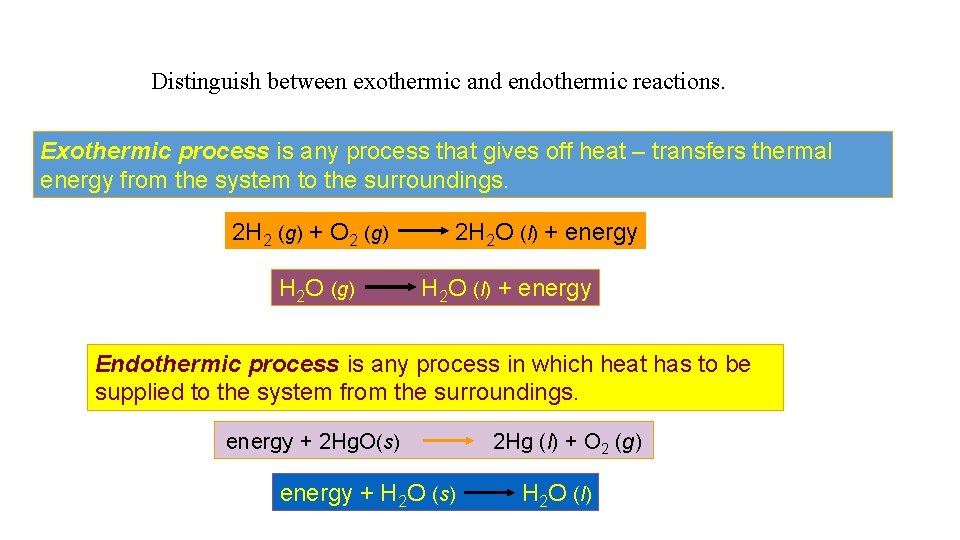
Distinguish between exothermic and endothermic reactions. Exothermic process is any process that gives off heat – transfers thermal energy from the system to the surroundings. 2 H 2 (g) + O 2 (g) H 2 O (g) 2 H 2 O (l) + energy Endothermic process is any process in which heat has to be supplied to the system from the surroundings. energy + 2 Hg. O(s) energy + H 2 O (s) 2 Hg (l) + O 2 (g) H 2 O (l)
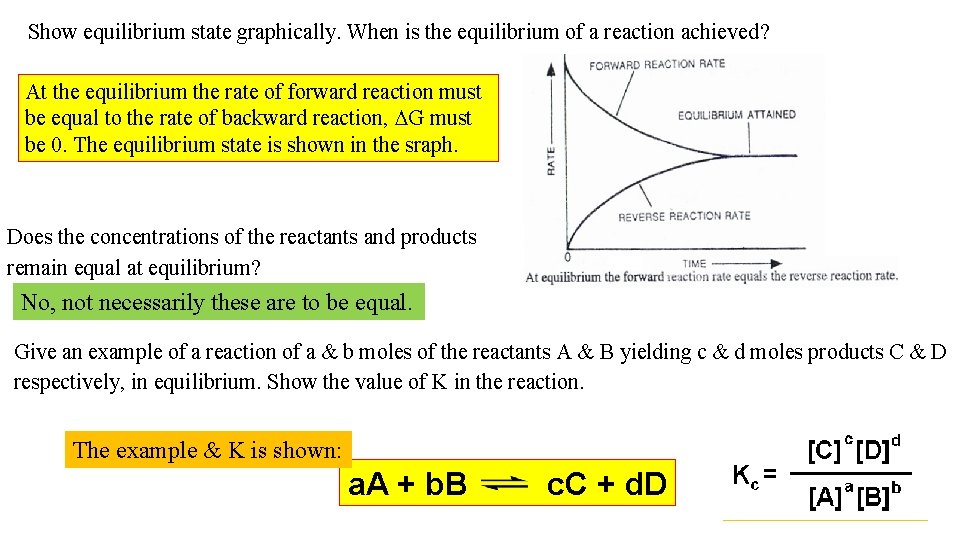
Show equilibrium state graphically. When is the equilibrium of a reaction achieved? At the equilibrium the rate of forward reaction must be equal to the rate of backward reaction, DG must be 0. The equilibrium state is shown in the sraph. Does the concentrations of the reactants and products remain equal at equilibrium? No, not necessarily these are to be equal. Give an example of a reaction of a & b moles of the reactants A & B yielding c & d moles products C & D respectively, in equilibrium. Show the value of K in the reaction. The example & K is shown: a. A + b. B c. C + d. D

Distinguish between Keq & Q. Explain the states Keq > Q, Keq = Q & Keq < Q. As the products form, the rate in the reverse direction increases. When the two rates are the same equilibrium is achieved. At DG = 0, the reaction is at equilibrium. At DG < 0, the reaction is spontaneous. Chemical equilibrium occurs in chemical reactions that are reversible. In a reaction such as: CH 4(g) + H 2 O(g) CO(g) + 3 H 2 (g) , equilibrium constant = equilibrium quotient. If Q > Keq , the product side is too high and the equilibrium will shift to the left to restore equilibrium. If Q < Keq , the product side is too low and the equilibrium will shift to the right to restore equilibrium, at equilibrium Q = Keq.

Describe Law of mass action and explain Le Chattelier’s principle. Law of mass action : Law of mass action states that reaction rate is proportional to the product of the concentrations of the reactants. Describe Le Chatelier's Principle states: When a system in chemical equilibrium is disturbed by a change of temperature, pressure, or a concentration, the system shifts in equilibrium composition in a way that tends to counteract this change of variable. A change imposed on an equilibrium system is called a stress. The equilibrium always responds in such a way so as to counteract the stress.

Distinguish between specific heat and heat capacity. The specific heat (s) of a substance is the amount of heat (q) required to raise the temperature of one gram of the substance by one degree Celsius. The heat capacity (C) of a substance is the amount of heat (q) required to raise the temperature of a given quantity (m) of the substance by one degree Celsius. C = ms Heat (q) absorbed or released: q = ms. Dt q = CDt Dt = tfinal - tinitial

Does pressure affect equilibrium without any gas in the system? Not at all, since there is no change in volume, no effect of pressure is there. In which direction will a reaction move if its reaction quotient is less than its equilibrium constant (Q < Keq)? The product is less, so the reaction will move in forward direction to give more products. Show the conjugate acid and base pairs in the following ionization : HCN(l) + H 2 O CN–(aq) + H 3 O+(aq). The conjugate base of the acid HCN(l) is CN–(aq) and conjugate acid of the base H 2 O is H 3 O+(aq).
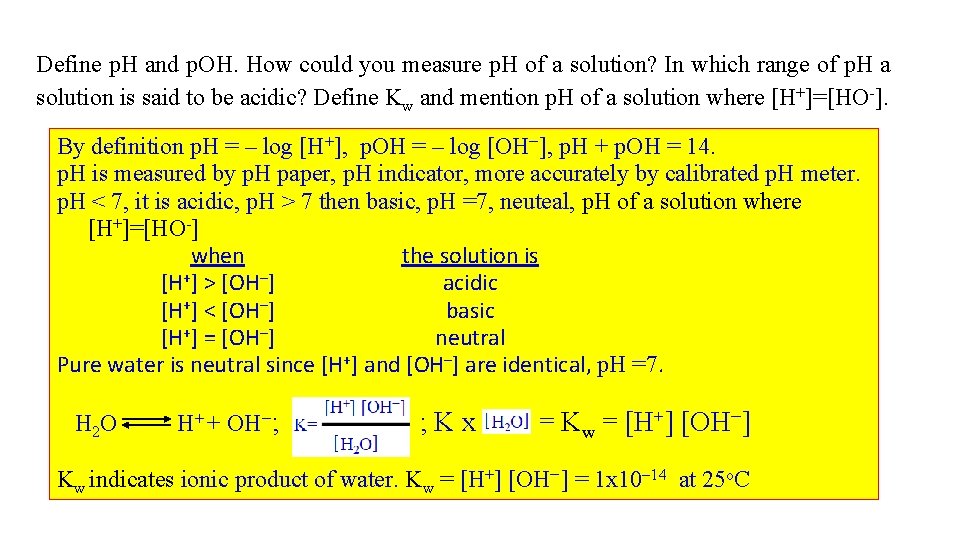
Define p. H and p. OH. How could you measure p. H of a solution? In which range of p. H a solution is said to be acidic? Define Kw and mention p. H of a solution where [H+]=[HO-]. By definition p. H = – log [H+], p. OH = – log [OH-], p. H + p. OH = 14. p. H is measured by p. H paper, p. H indicator, more accurately by calibrated p. H meter. p. H < 7, it is acidic, p. H > 7 then basic, p. H =7, neuteal, p. H of a solution where [H+]=[HO-] when the solution is [H+] > [OH–] acidic [H+] < [OH–] basic [H+] = [OH–] neutral Pure water is neutral since [H+] and [OH–] are identical, p. H =7. H 2 O H+ + OH-; ; Kx = Kw = [H+] [OH-] Kw indicates ionic product of water. Kw = [H+] [OH-] = 1 x 10 -14 at 25 o. C

Explain Distribution law or Partition coefficient.

Explain common ion effect. COMMON-ION EFFECT: Adding a common ion to solution decreases dissociation of acid or base.

Explain saturated solution and solubility product. A solid always dissolves until no more can dissolve - called molar solubility (mol/L or M) An equilibrium is established when the amount dissolving equals the amount precipitating A solution equilibrium with solid remaining is called “saturated”

Explain Dissociation constant. When a weak electrolyte AB is dissociated to its constituent ions A+ + B-, the ratio of the product of concentrations of the dissociated ions to the concentration of associated AB is known as dissociation constant of AB. AB A+ + B - [AB] = C(1 -α) mol litre-1 [A+] = Cα mol litre-1 [B-] = Cα mol litre-1 Rate of dissociation = k 1 x C(1 -α) Rate of combination = k 2 x Cα At equilibrium: k 1 x C(1 -α) = k 2 x Cα or or or mol per liter, , This Kc is known as dissociation constant of AB.

Explain i) Degree of dissociation, ii) Weak electrolyte and iii) Electrophoretic effect. Compound that is hardly dissociated and thereby its both degree of dissociation and dissociation constant are very small, is said to be a weak electrolyte. eg CH 3 COOH CH 3 COO- + H+. Since solvent molecules attached to the ionic atmosphere moving in the opposite direction produce friction hence reduce the motion of central ion.

Explain ionic atmosphere and relaxation effect. The relaxation effect: According to this effect, each cation is surrounded by a number of anions and vice versa in solution; which is called ionic atmosphere of the oppositely charged ions. On applying e. m. f. , the ion moves towards oppositely charged electrode leaving behind the ionic atmosphere. To form a new ionic atmosphere some time is taken which is called relaxation time and this effect of the ionic atmosphere is called relaxation effect.

Give the values of Ka for a moderate acid and a weak acid. In general: Ka < 10 – 3 Weak acid 10 – 3 < Ka < 1 Moderate acid Ka > 1 Strong acid State Ostwald’s dilution law.

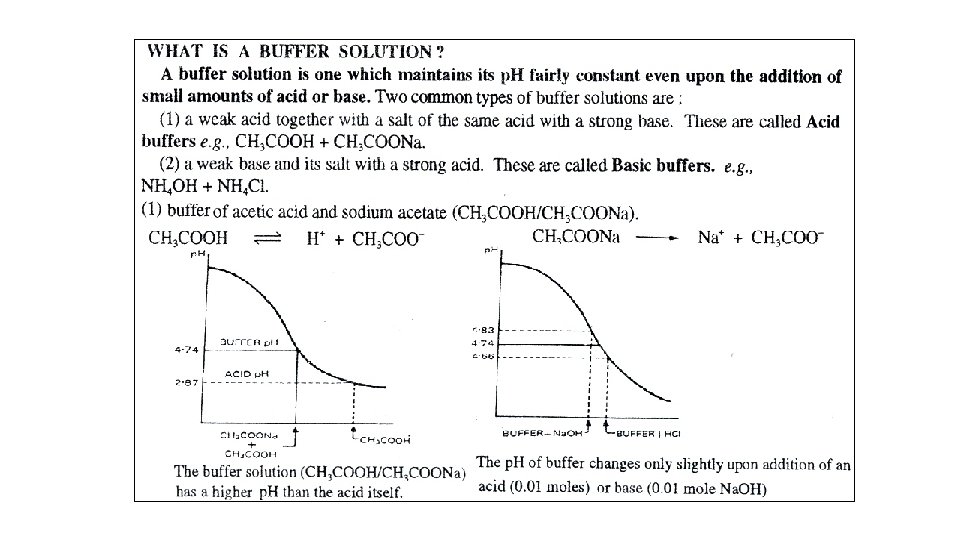
Buffer solution A buffer solution exhibits very small change in p. H changes when H 3 O+ or OH- is added. A buffer solution consists of relatively high concentration of the components of a conjugate weak acid base pair. Buffers change p. H slowly when acid or base is added. Buffers take advantage of common-ion effect, dissociate very little. When a strong acid is added to the solution, it neutralizes the base, When a strong base is added to the solution, it neutralizes the acid.
- Slides: 25