What data are collected How and who by

What data are collected? How, and who by?
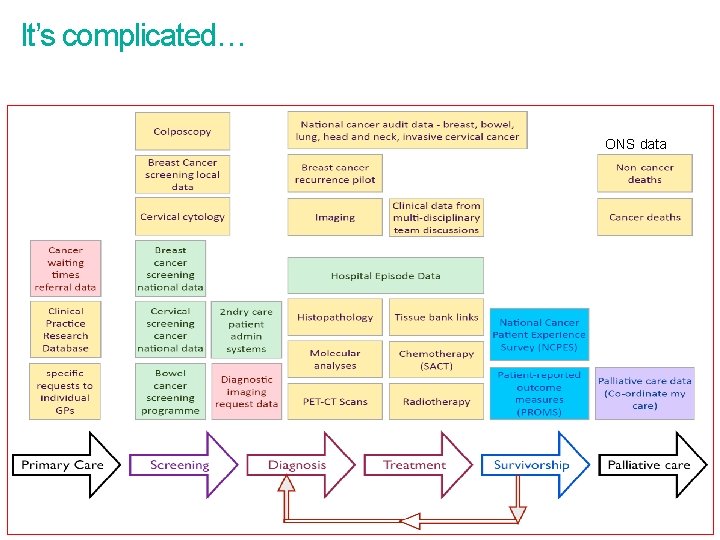
It’s complicated… ONS data

Focus on the main data collections by trusts • Two main types • Cancer Information Standards • National Cancer Audits • Five standards and four audits (at present) • Standards - mandated through NHS National contract and commissioning • National audits - monitored through CQC and Quality Surveillance Programme (formerly Peer Review) • Two main collection routes for cancer data from trusts • PHE/NCRS • HSCIC/CASU • Other routine data and data collections such as HES and ONS deaths • HSCIC collects most of the NHS data (including some cancer data) • PHE/NCRS collates all cancer related data (some directly, some indirectly) 3

National audits • Partnerships between clinical (Royal Colleges) and information services • Four audits • NBCA(NBOCAP) – Bowel • NOGCA – Oesophago-gastric • NLCA – Lung • NPCA – Prostate • DAHNO – Head & Neck currently no contract • Breast Cancer - likely to be commissioned in 2015 • Majority of audit data is also collected through COSD • Try to upload monthly for audits as well as COSD • Record in one place only for both COSD and audits 4

National Audits • Consultant Outcomes Publications based on audit data • COSD provides the routine data (Bowel audit only 4 additional items, lung all in COSD inc EGFR and smoking status) • Good case ascertainment in established audits ( eg 94% Bowel) • Developing PROMS/PREMS • Increasing focus on using the data rather than collecting the data 6

Data Standards • Five standards: • CWT - Cancer Waits • RTDS – Radiotherapy • SACT – Chemotherapy • DID – Diagnostic imaging • COSD – Cancer Outcomes and Services • COSD incorporates key audit fields and CWT fields 12

Cancer Waiting Times (CWT) – what’s new? • Guidance document being updated § Cancer Waiting Times: a Guide Version 9 in progress § New data items to be collected from March 2016 § NHS NUMBER STATUS INDICATOR CODE § REFERRAL REQUEST RECEIVED DATE (INTER-PROVIDER TRANSFER). § Change of submission type from March 2016 § CSV replaced by XML submissions 13

Radiotherapy dataset (RTDS) § Previously collected and compiled by Nat. Can. SAT (National Clinical Analysis and Specialised Applications Team) § Radiotherapy data will be collected and compiled by NCRS from April 2016 § Exploring whether Commissioning data can be omitted as available forom other sources - ? just the key radiotherapy data items 14

Diagnostic Imaging Dataset (DID) § Record-level data from NHS Radiology Information Systems (RIS) in England • Covers pathway – who, what, where, when etc • But not reports § Now three years data and ready to link and assess impact of diagnostiic imaging on cancer outcomes § NHS staff can apply for access to the DID summary data § § Further details: http: //www. hscic. gov. uk/DID Monthly provisional summary and annual reports by NHS England § http: //www. england. nhs. uk/statistics/statistical-work-areas/diagnostic-imaging-dataset/

Systemic Anti-Cancer Therapy (SACT) • Monthly submissions from all trusts providing chemotherapy • Currently to Chemotherapy Intelligence Unit in Oxford but moving into NCRS system • Monthly, quarterly and annual reports available through website http: //www. chemodataset. nhs. uk/home • Coverage not yet complete for all trusts and cases • Particularly haematology, paediatric and not hospital settings • Some poorly collected data items • eg Morphology, outcomes, performance status • But new information never previously available 17

SACT – new information on chemotherapy March 14 to February 15 April 14 to March 15 Patients Receiving Drug Treatments Regimens Commenced 165, 256 165, 883 282, 646 283, 384 Cycles Commenced 790, 868 805, 326

Cancer Outcomes and Services Dataset (COSD) – a reminder § Aligned and rationalised with other cancer datasets § Core subset incorporates registration data for national and international epidemiology etc § 12 site specific subsets include key audit items for service and outcomes analysis etc § Include all recurrences and MDT meetings by July 2016 § Pathology submissions in XML from January 2016 (structured data) – Plan by 1 st July 2015! § Key items: § § • • 20 Basis of diagnosis and morphology Stage and performance status CNS contact Site specific audit items

How do we know it’s right? • Clinicians are responsible for ensuring accurate data is recorded • Make sure that you know how and when your data is submitted • Agree a (manageable) system for checking accuracy • Use the COSD portal regularly (ask for reports to be sent to you) 21
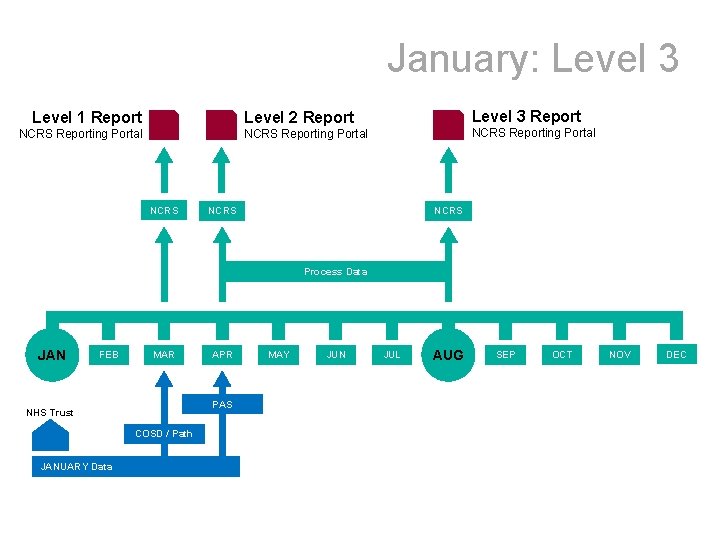
January: Level 3 Level 1 Report NCRS Reporting Portal NCRS Level 2 Report Level 3 Report NCRS Reporting Portal NCRS Process Data JAN FEB MAR PAS NHS Trust COSD / Path JANUARY Data APR MAY JUN JUL AUG SEP OCT NOV DEC

COSD portal – feedback reports 23
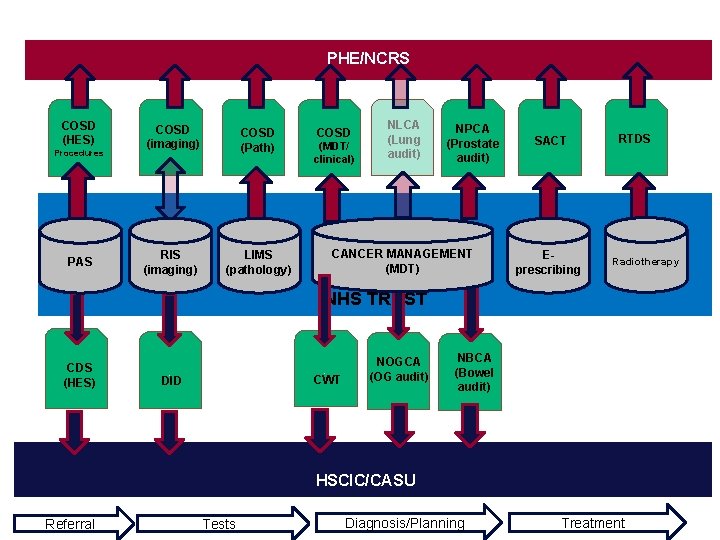
PHE/NCRS COSD (HES) Procedures PAS COSD (imaging) COSD (Path) RIS (imaging) LIMS (pathology) COSD (MDT/ clinical) NLCA (Lung audit) NPCA (Prostate audit) CANCER MANAGEMENT (MDT) SACT Eprescribing RTDS Radiotherapy NHS TRUST CDS (HES) . CWT . DID NOGCA (OG audit) NBCA (Bowel audit) HSCIC/CASU Referral Tests Diagnosis/Planning Treatment

“Do once and share” • Principle is to record once only on one system if possible • Try to avoid transcribing from paper to electronic systems • Try to use electronic systems where they are available • CWT and most audit items are also included in COSD • COSD submitted in multiple extracts from different systems (no need to duplicate entry) • Try to avoid double entering data (eg audits can all be uploaded from local systems)

A burden shared… • Most data is needed for clinical and/or patient pathway management • Decide who is the best person to record the different data items • Don’t leave it all to one person • See who records the data items at the point of care • Recording in MDT meetings can work – but needs to become routine 26

Thank you 27
- Slides: 19