WHAT ARE IONIC BONDS Section 15 5 IONIC

WHAT ARE IONIC BONDS? Section 15 -5

IONIC BOND Charged Neutral Atoms Particles In an atom, the When an atom gains number of protons (+) electrons (becomes equals the number of negative) or loses electrons (-). This is a electrons (becomes neutral atom. positive), it is called an ion.

IONIC BOND Compounds are held together by atomic bonds, either ionic or covalent. Ionic bonds involve the transfer of outer shell electrons from one element to another. The element giving up the electron becomes a positive ion and the element accepting the electron becomes a negative ion. As a result, they are attracted to each other because of opposite electrical charges. This force is what bonds the elements together in a compound.

IONIC BOND Sodium gives 1 electron away and chlorine takes on the electron. Na+ ion and Cl- ions are formed.

CRYSTALS Many compounds that contain ionic bonds are solid. A crystal lattice is a solid which has a pattern of positive and negative ions. The shape of a crystal depends on its crystal lattice. There are 6 types of crystal lattices.

WHAT ARE COVALENT BONDS? Section 15 -6

COVALENT BONDS Compounds are held together by atomic bonds, either ionic or covalent. Most atoms do not have complete outer energy levels. Covalent bonds involve the sharing of outer energy level electrons. Thus ions are not formed, the atoms remain neutral.
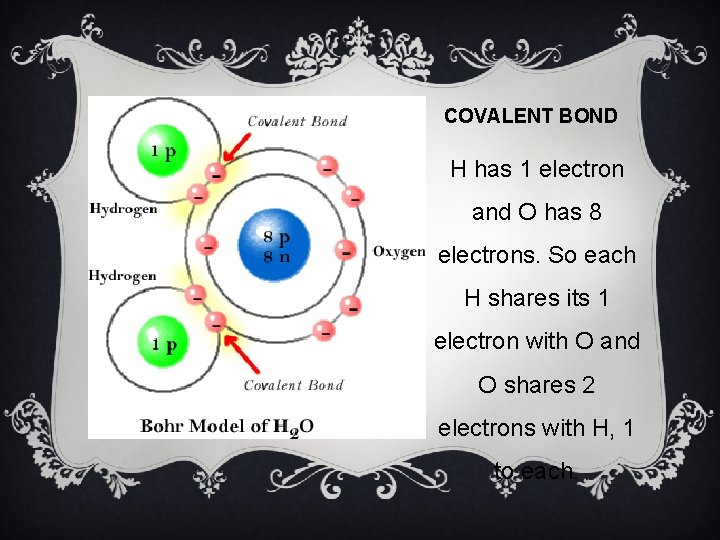
COVALENT BOND H has 1 electron and O has 8 electrons. So each H shares its 1 electron with O and O shares 2 electrons with H, 1 to each.

IONIC VS. COVALENT Covalent Ionic compounds v. Atoms complete outer energy levels v. Electrons are lost v. Electrons are or gained shared v. Atoms form ions v. Atoms remain neutral
- Slides: 9