Welcome to Grade 12 Biology Bios life Ology

Welcome to Grade 12 Biology! § Bios = life § Ology = the study of § Biology is the study of life and living processes! Introduction to biology video: http: //www. youtube. com/watch? v=Vg. TPg 99 V_J M

02/09/09 Chemistry Review- what? I thought it was Bio. .

Chemistry of Life

All life is hierarchically organized

What is matter? q All “stuff” is matter q matter has mass q Fundamental unit = atom q compounds – combinations of elements eg. Na. Cl

Elements essential to life q All life forms require 25 elements q The Big 4 a. CHON i. Carbon ii. Hydrogen iii. Oxygen iv. Nitrogen Makes up 96% of living matter

Elements essential to life q All life forms require 25 elements q The Little 4 b. PCa. SK i. Phosphorous ii. Calcium iii. Sulphur iv. potassium Makes up 3% of living matter

Elements essential to life q All life forms require 25 elements q Trace elements Essential in small amounts Makes up < 1%

Elements essential to life
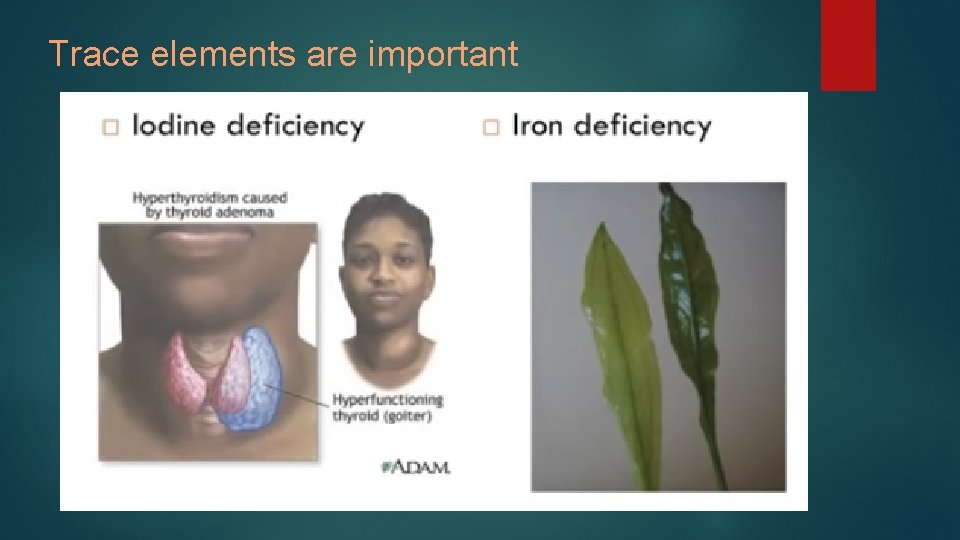
Trace elements are important

Trace elements are important

Calculating subatomic particles

Calculating subatomic particles
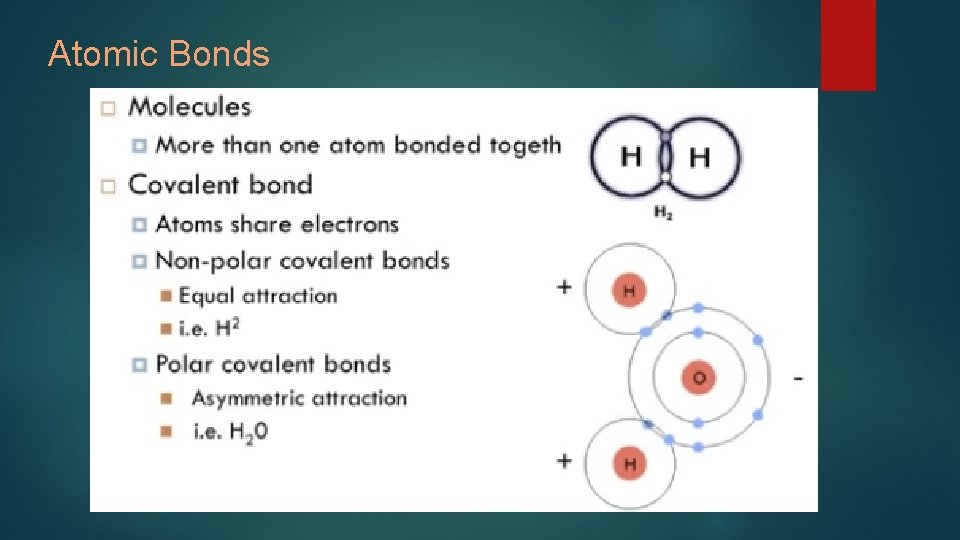
Atomic Bonds
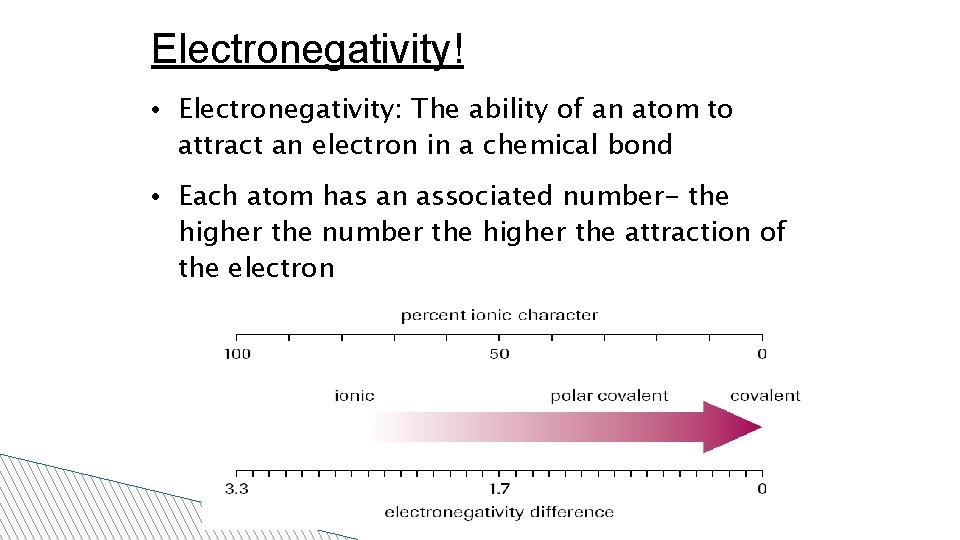
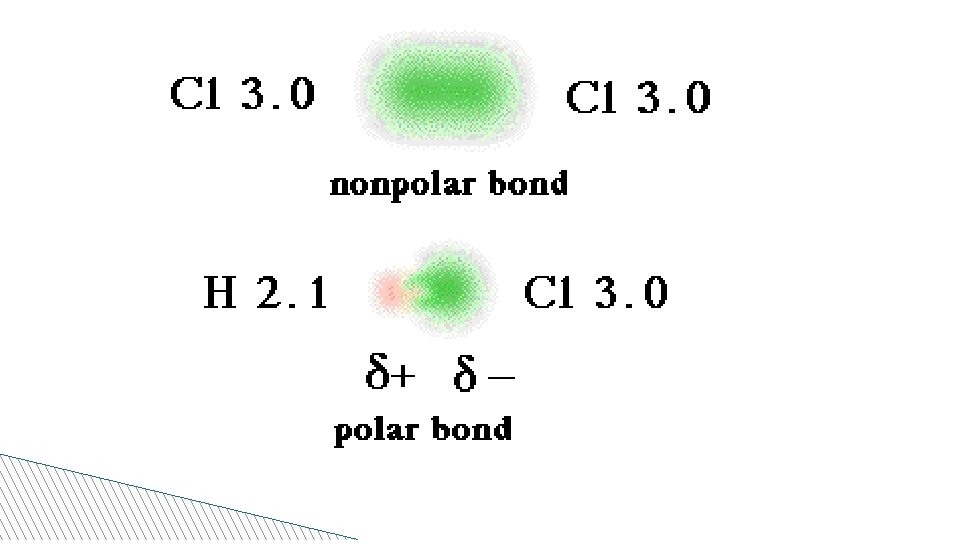
Electronegativity! • Electronegativity: The ability of an atom to attract an electron in a chemical bond • Each atom has an associated number- the higher the number the higher the attraction of the electron
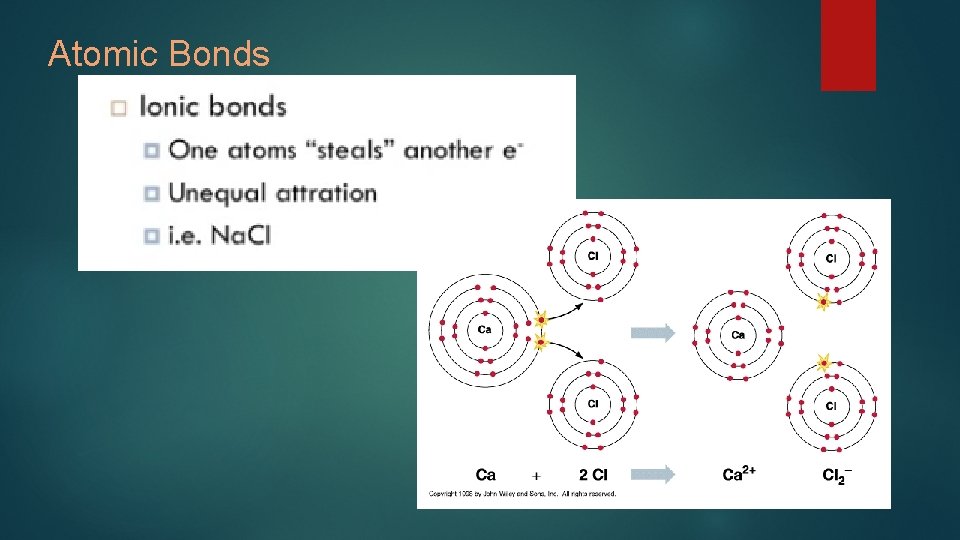
Atomic Bonds

Number of bonds

Chemical Reactions
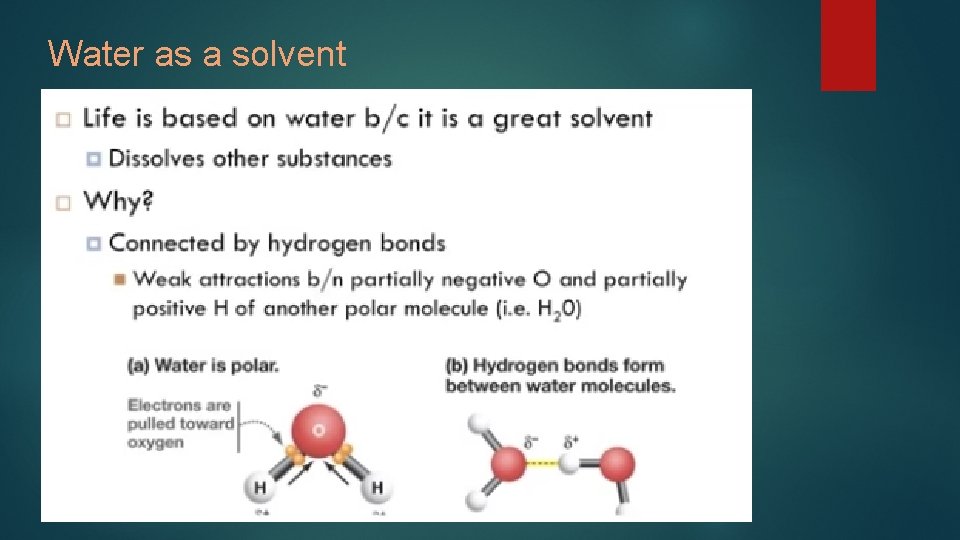
Water as a solvent
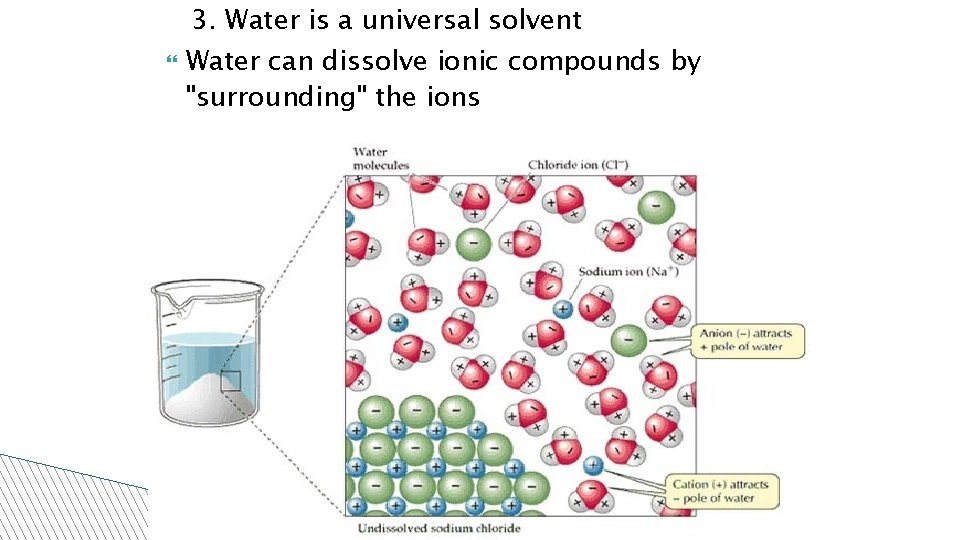
3. Water is a universal solvent Water can dissolve ionic compounds by "surrounding" the ions 02/09/09

Other properties of water

Other properties of water

Other properties of water
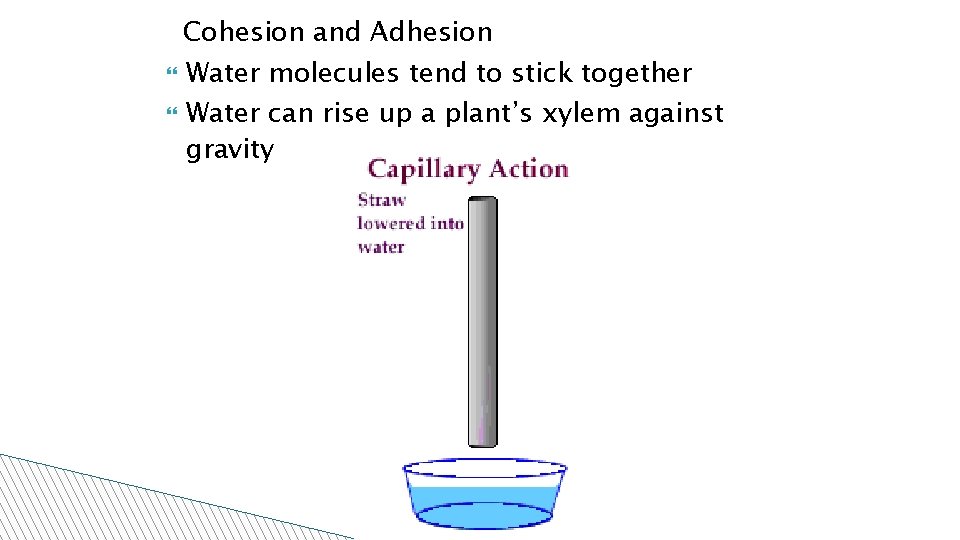
Cohesion and Adhesion Water molecules tend to stick together Water can rise up a plant’s xylem against gravity

Unique Properties of Water • Chemistry of Water Video Summary: • http: //www. youtube. com/watch? v=DAil. C 0 sjvy 0 • How do these unique properties make life possible? . . .

Hydrophilic vs. Hydrophobic • Ionic / polar compounds “love” water! • Nonpolar compounds “fear” water!

Properties of Acids • Low p. H • Sour taste • React with some metals to produce H 2 • A good conductor of electricity in solution • Release H+ ions in solution (actually, hydronium H 3 O+ ions) • Indicators: – Blue litmus turns red
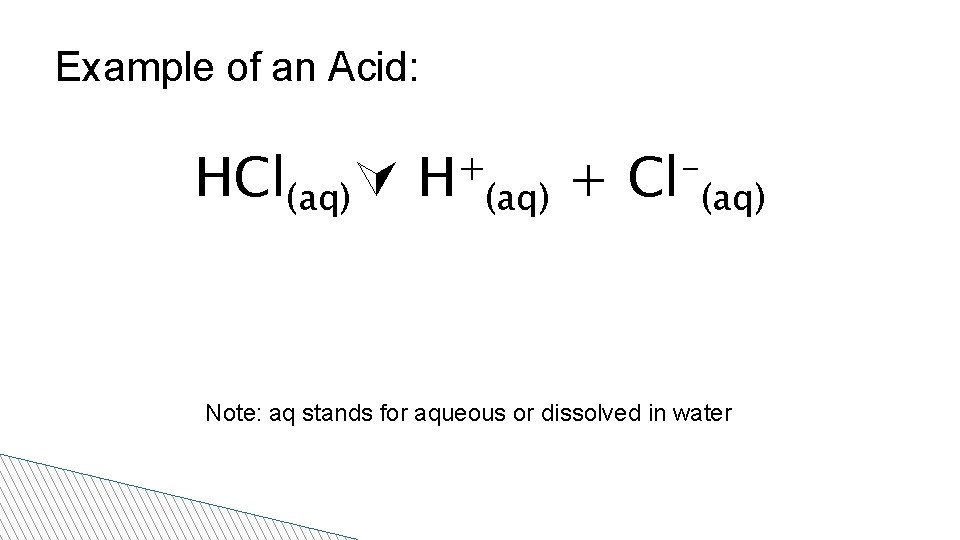
Example of an Acid: HCl(aq) + H (aq) + Cl (aq) Note: aq stands for aqueous or dissolved in water

Properties of Bases • High p. H • Bitter taste • Slippery and soapy to the touch • A good conductor of electricity in solution • Release OH- (hydroxide) ions in solution • Indicators: – Red litmus turns blue
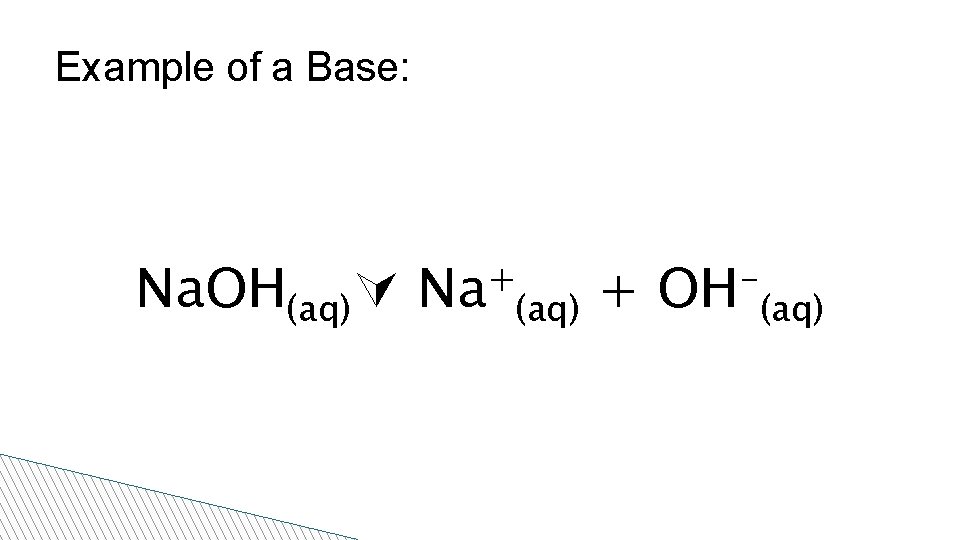
Example of a Base: Na. OH(aq) + Na (aq) + OH (aq)
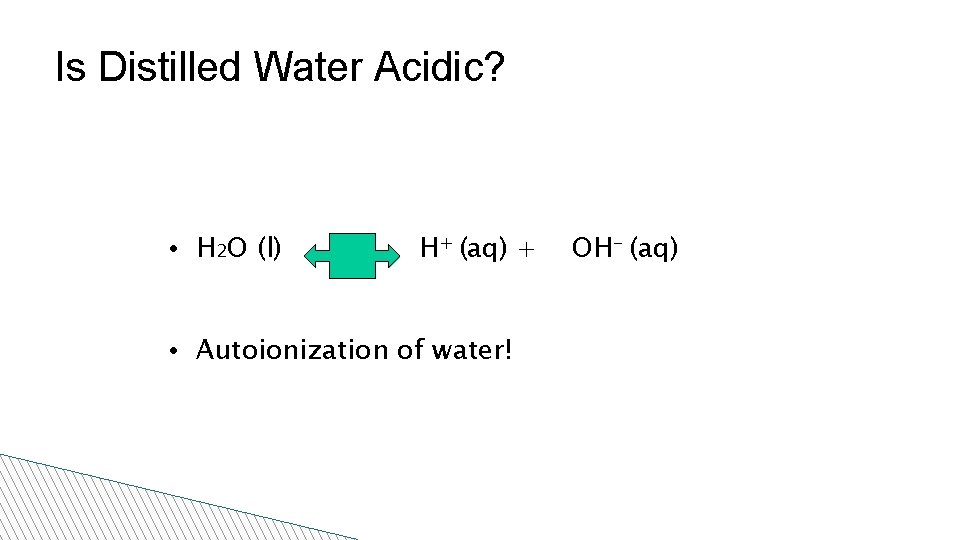
Is Distilled Water Acidic? • H 2 O (l) H+ (aq) + • Autoionization of water! OH- (aq)

The p. H Scale • Used to identify how acidic or basic a solution is based on a scale of 1 to 14 • A very acidic solution has a LOW p. H • A very basic solution has a HIGH p. H • A neutral solution has a p. H of 7 (water)
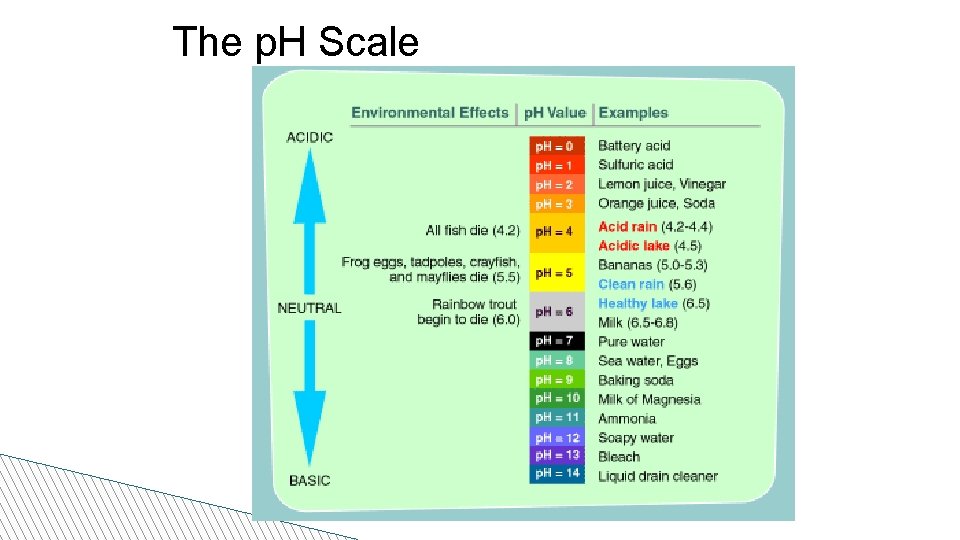
The p. H Scale

p. H • Is a measure of how acidic a substance is – Acid examples Hydrochloric acid, Sulfuric Acid, Citric Acid • It is measured on a scale (p. H scale) from 1 -14 7 (Neutral) = Water and Blood < 7 (Acidic) = e. g. Vinegar > 7 (Basic) = e. g. Ammonia (household cleaner)

Logarithims • The p. H scale is a logarithmic scale • One spot on the scale is a 10 fold difference in the concentration of the solution Examples: p. H 3 vs p. H 4 - the 1 st is 10 times more acidic p. H 3 vs p. H 5 - the 1 st is 100 times more acidic

NOTE: • As p. H INCREASES the solution becomes more BASIC • As p. H DECREASES the solution becomes more ACIDIC

Neutralization Reactions • When an acid reacts with a base • p. H moves towards 7 • Double displacement reactions – E. g. HCl(aq) + Na. OH(aq) HOH(l) (H 20)(l) + Na. Cl(aq) • Produces a salt and water Acid + Base Water+ Salt
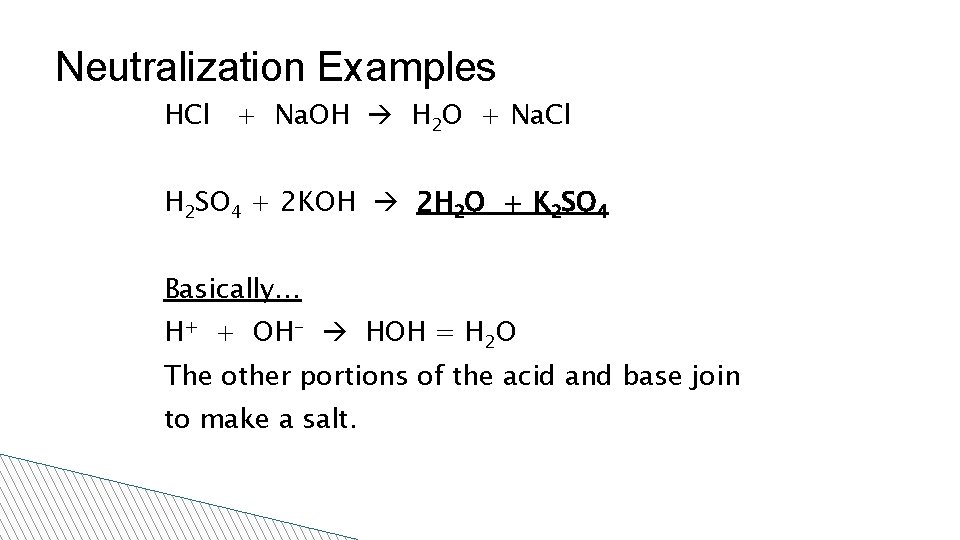
Neutralization Examples HCl + Na. OH H 2 O + Na. Cl H 2 SO 4 + 2 KOH 2 H 2 O + K 2 SO 4 Basically… H+ + OH- HOH = H 2 O The other portions of the acid and base join to make a salt.

What is a salt? • Any combination of positive and negative ions formed by the reaction of an acid and a base (ionic compound). • Not necessarily table salt (Na. Cl)!

What is a Buffer? • A chemical that resists p. H changes in a solution by accepting or donating H+ ions. e. g. carbonic acid in your blood helps to regulate the p. H within a narrow range of 7. 35 -7. 45 HCO 3 - + H+ H 2 CO 3 • If the blood is too acidic (too many H+ ions), they bind to the bicarbonate ions and get locked up as H 2 CO 3 • If blood is too basic (too few H+ ions) the reaction goes the other way (right to left)

Acids and Bases Song! • https: //www. youtube. com/watch? v=u 9 n. OIZD dv. Rw
- Slides: 42