Welcome to Class Silently Begin Do Now What

Welcome to Class Silently Begin Do Now What elements are we? 1) I have 26 electrons. Fe, Iron 2) My atomic number is 3. Li, Lithium 3) My chemical symbol is K. Potassium 4) I have an atomic mass of 14 amu and 8 neutrons. atomic mass = p+ + n 14 = p+ + 8 14 – 8 = p+ 6 = p+ C, Carbon

Homework / Announcements Due: Text 332 -334 Homework Isotope Worksheet Textbook 343 -349 Sign up for turnitin. com Test: Tuesday October 27, 2015 Ongoing: Science Fair Project and USA Test. Prep

Parts of a Lab Report Check List Title (underlined and on the top center of the page) Background Information Problem Statement: Identify the research question/problem and state it clearly. Variables and Control Test: Independent, Dependent, and Controlled variables Hypothesis: “if…, then…, because…” statement. Null Hypothesis: It is the opposite of the hypothesis. Materials: Procedure: Data: Ensure that all data is recorded in an organized way (data chart) Graphs: Remember TAILS Results: Data analysis statement Conclusion and Evaluation: Answer listed questions in at least three paragraphs A conclusion statement answers the following 7 questions in at least three paragraphs. Reference Page: 5 references, APA style http: //www. apastyle. org/ https: //owl. english. purdue. edu/owl/resource/560/01/

Parts of a Lab Report Check List Double spaced 12 point font Arial, Times New Roman 1” Margins Heading: Name (first and last)

Turnitin. com Class codes 1 st period: 10780813 2 nd period: 10780883 th 4 period: 10780893 th 6 period: 10780895

Table of Contents: Bohr’s model L. O. SWBAT identify and draw atoms of the periodic table using Bohr’s model. Standard: SC. 8. P. 8. 7
Quiz – Periodic Table Practice With your table group Use your notes

Turn to page 343 Class Read Aloud
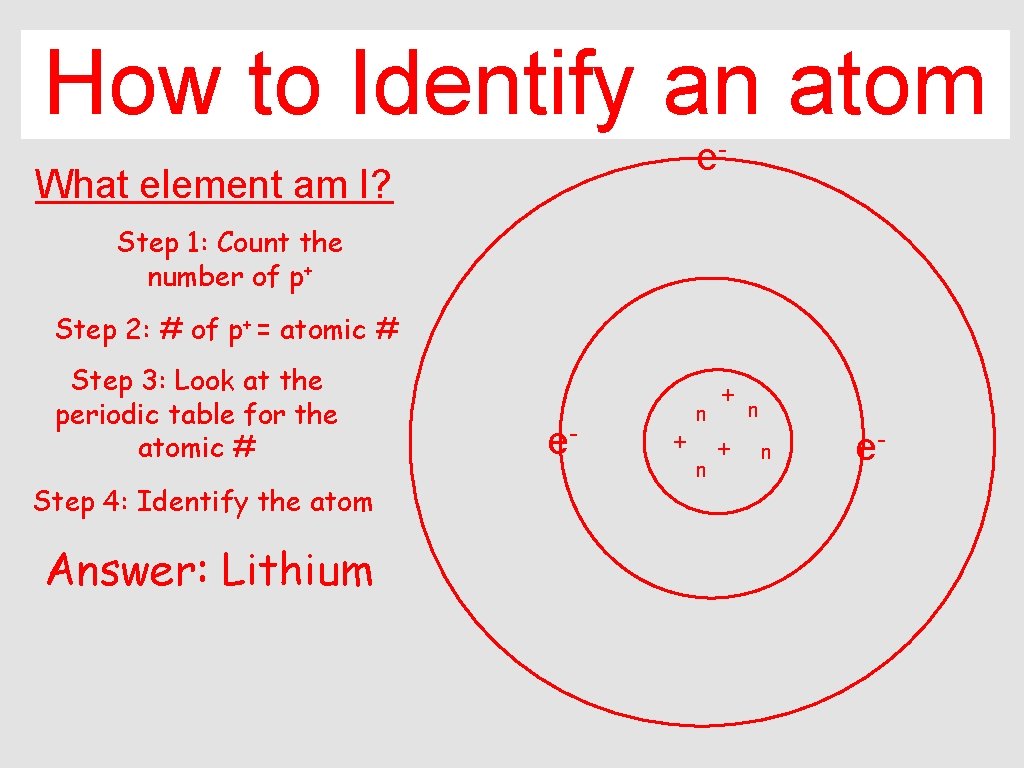
How to Identify an atom e- What element am I? Step 1: Count the number of p+ Step 2: # of p+ = atomic # Step 3: Look at the periodic table for the atomic # Step 4: Identify the atom Answer: Lithium e- n + + n n e-
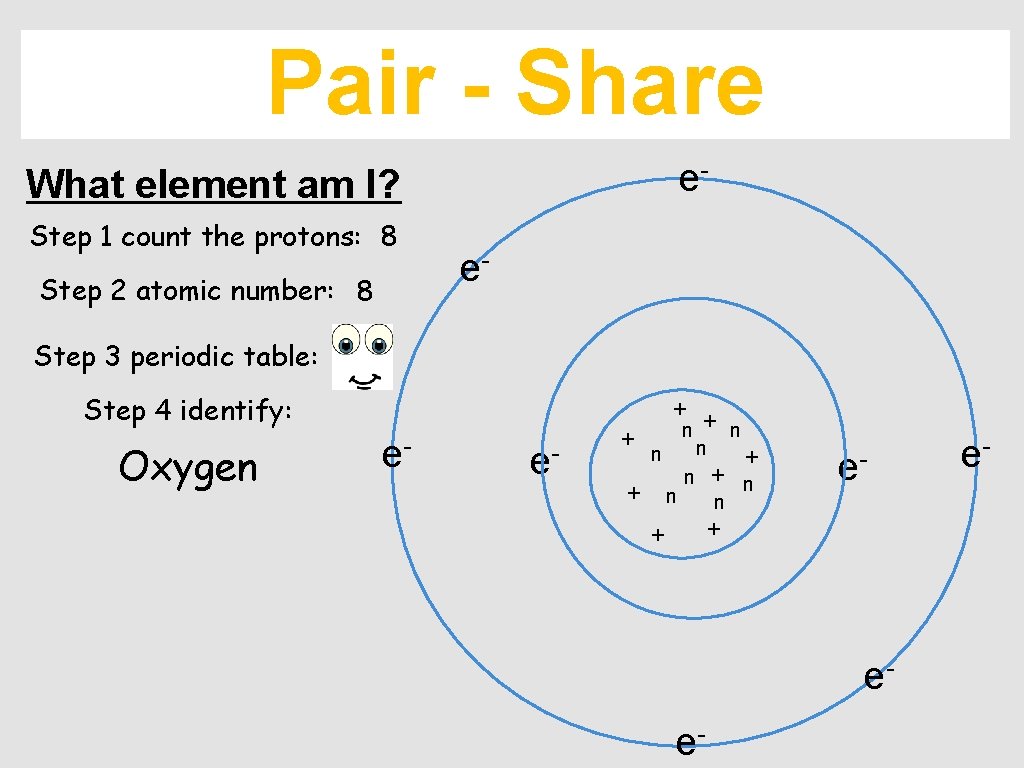
Pair - Share e- What element am I? Step 1 count the protons: 8 Step 2 atomic number: 8 e- Step 3 periodic table: Step 4 identify: Oxygen + e- e- + n n n + + n e- n + + ee- e-

How to draw an atom Step 1: Find the atom on the periodic table. Step 2: Determine the number of p+, e-, and n Step 3: Draw the nucleus (p+ and n) Step 4: Draw ring 1 (max. of 2 e-) If needed, …. Step 5: Draw ring 2 (max. of 8 e-) Step 6: Draw ring 3 (max. of 8 e-)
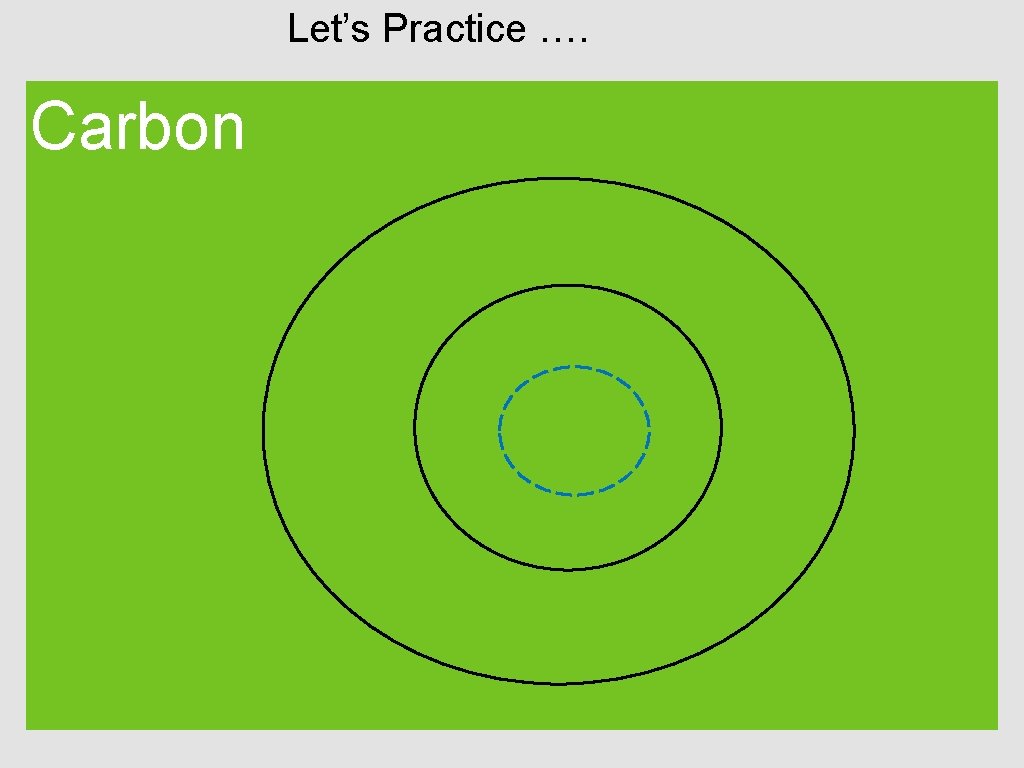
Let’s Practice …. Carbon

Now you do…. . Beryllium, Be

Independent Work 1. Isotope Worksheet 2. Textbook 343 -349, 351 -353 3. Sign up for turnitin. com 1: 10780813 4: 10780893 2: 10780883 6: 10780895
- Slides: 14