WELCOME Lecture on Melting Point and Boiling Point

WELCOME

Lecture on Melting Point and Boiling Point Class-IX (Chemistry) By: Ferdousi Ahsan Teacher Foundation School

Learning outcome • After this lesson, student will be able to know about: • Melting Point (M. P) and Boiling Point (B. P) • Heating curve • Cooling curve • Factors of M. P and B. P • Determination of M. P and B. P

What is Melting Point (M. P)? • Melting Point of a solid is the temperature at which it changes state from solid to liquid at atmospheric pressure. • At the melting point the solid and liquid phase exist in equilibrium. • Example: M. P of Iron is 1538 °C

What is Boiling Point (B. P)? • Boiling Point of a liquid is the temperature at which it changes state from Liquid to Gas at atmospheric pressure. • The B. P of a liquid varies depending upon the surrounding environmental pressure. • A liquid in a vacuum has a lower B. P than when that liquid is at atmospheric pressure. • A liquid at high pressure has a higher B. P than when that liquid is at atmospheric pressure. • Example: B. P of water is 99. 97 °C at a pressure of 1 atm (i. e. , 101. 325 k. Pa).

Difference between M. P and B. P M. P B. P
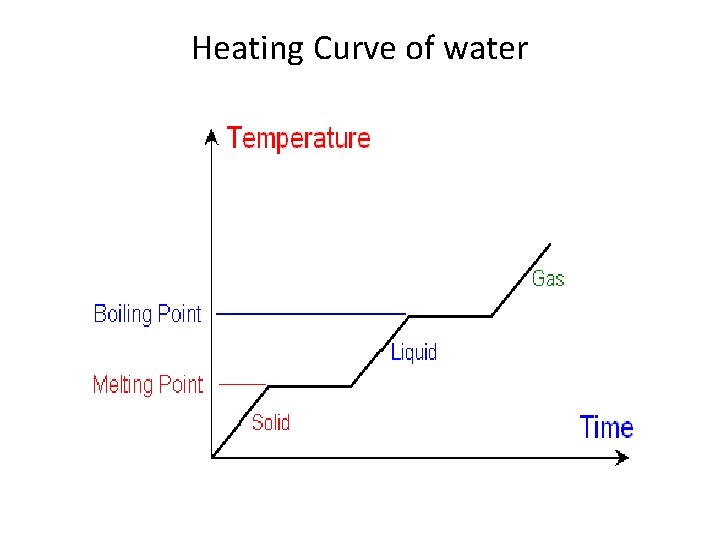
Heating Curve of water

• The first change of state is melting (changing from a solid to a liquid). The temperature remains the same while a substance melts. For water, this temperature is 0°C because the melting point for water is 0°C. • The second change of state is boiling (changing from a liquid to a gas). The temperature remains the same while a substance boils. For water, this temperature is 100°C because the boiling point for water is 100°C. • Different substances have different melting points and boiling points, but the shapes of their heating curves are very similar.
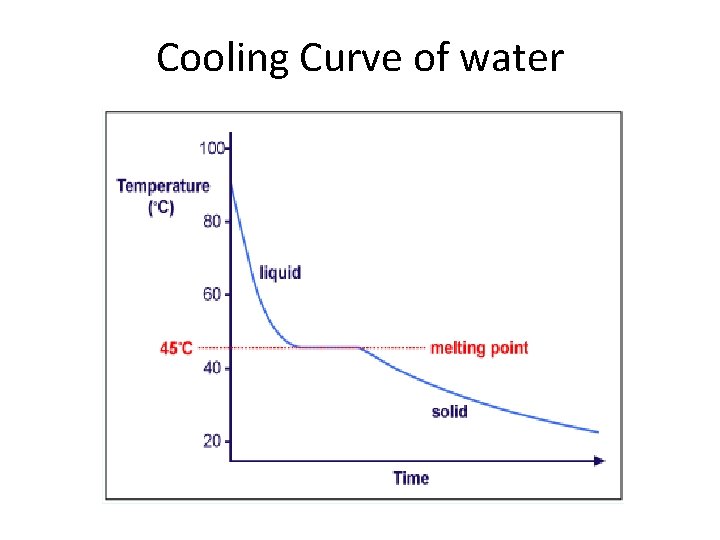
Cooling Curve of water

Factors of M. P • • Molecular size Impurities (solute added) Pressure Shape of the molecule

Factors of B. P • Pressure: when the external pressure is: – less than one atmosphere, the boiling point of the liquid is lower than its normal boiling point. – equal to one atmosphere, the boiling point of a liquid is called the normal boiling point. – greater than one atmosphere, the boiling point of the liquid is greater than its normal boiling point.

• Types of Molecules: the types of molecules that make up a liquid determine its boiling point. If the intermolecular forces between molecules are: • relatively strong, the boiling point will be relatively high. • relatively weak, the boiling point will be relatively low.


Group Task • What is latent heat of water? Explain with heating curve of water.

Home work • Make a list of boiling point of the following substances. • Gold, Iron, Aluminum, Sodium, Sulfur, Iodine , Phenol, Acetic acid anhydride, Petrol, Ether, Hydrogen, Nitrogen, Ammonia • Make a list of Melting point of the following substances. • Aluminum, Bromine, Copper, Ethyl Alcohol, Gold, Hydrogen, Iron, Mercury, Nitrogen, Oxygen, Sulfuric acid, Water

THAN KS
- Slides: 16