Welcome back Access to Science Chemistry 1 Periodicity

Welcome back Access to Science - Chemistry 1
Periodicity Analytical Chemistry Reacting amounts Chemistry Structure & Bonding Organic Chemistry 2

Reacting amounts unit � Balancing equations � Relating equations to chemical amounts � Taught with worksheets culminating in an exam in 4 weeks time. � Quantitative analysis: how we can work out how much we have of an unknown amount of one chemical by using a known amount of another chemical in a process known as titration � Assessed practically culminating in a report 3

Let’s break this down: � Learning aims for this session: � Today you will learn how to balance equations � After the break you will carry out your first titration 4

https: //quizlet. com/49783310/scatter 5

(1) Identify all the species This can often be done by using a word equation: SODIUM + CHLORINE SODIUM CHLORIDE The reactants (sodium and chlorine) on the left give (denoted by an arrow, sometimes seen as equals sign) the products on the right (sodium chloride) 6

(2) Write out the correct formulas At this stage, ignore the numbers just concentrate on getting the formula for each species correct Na + Cl 2 Na. Cl 7

(3) Insert numbers in front of all the formula you have written so that the total number of atoms of each type is the same on both sides of the equation 2 Na + Cl 2 2 Na. Cl The big numbers at the front apply to EVERY atom / ion in the formula after them. The small sub-scripts apply to ONLY the atoms / ions before them. 8

Balance the following equations Remember: The big numbers at the front apply to EVERY atom / ion in the formula after them. The small sub-scripts apply to ONLY the atoms / ions before them. 1 3 Pb 2 2 SO 2 + + 2 O 2 2 SO 3 3 N 2 4 Na 2 CO 3 + H 2 O + 5 6 CO 2 + 6 H 2 O + 3 H 2 Pb 3 O 4 2 NH 3 CO 2 C 6 H 12 O 6 2 Na. HCO 3 + 6 O 2 9

Balance the following equations 6 7 CH 4 + 2 O 2 C 2 H 6 + 3 Cl 2 CO 2 + 2 H 2 O C 2 H 3 Cl 3 + 3 HCl When there is a sub-script number in brackets it applies to every atom/ion in the brackets 8 Ba. Cl 2 + 2 Na. OH 9 Cl 2 + 2 Na. OH 10 2 Ag. NO 3 + Cu Ba(OH)2 + 2 Na. Cl Na. OCl + Na. Cl + H 2 O Cu(NO 3)2 + 2 Ag 10

State symbols: Solid or precipitate – (s) Solution / dilute = (aq) Liquid = (l) Gaseous = (g) 11

Balance using state signs 1 Ba. Cl 2 (aq) + H 2 SO 4(aq) Ba. SO 4(s)+ 2 HCl (aq) Barium chloride solution + (and) dilute sulphuric acid -- (gives) a precipitate of barium sulphate + (and) dilute hydrochloric acid 12

Balance using state signs Solid aluminium oxide and dilute hydrochloric acid gives aluminium chloride solution and water 2 Al 2 O 3 (s) + 2 Al. Cl 3(aq) + 3 Cl 2 (g) + Na. OCl(aq) + 6 HCl(aq) 3 H 2 O(l) 2 Na. OH (aq) Na. Cl (aq) + H 2 O (l) 13

Balance using state signs 4 4 NH 3 (g) + 5 3 KOH (aq) 3 O 2 (g) + Fe(OH)3 (s) + 2 N 2 (g) + Fe. Cl 3 (aq) 6 H 2 O (g) 3 KCl (aq) 14

Homework / extension work 1 Ca (s) + Cl 2 (g) Ca. Cl 2 (s) 2 2 Cu (s) + O 2(g) 2 Cu. O (s) 3 C (s) 4 2 Fe (s) + 3 Cl 2 (g) 5 + H 2 O (g) H 2(g) + CO (g) 2 Fe. Cl 3 (s) Pb. O (s) + H 2 (g) Pb (s) + H 2 O (g) 15

6 - 10 16

Break 17
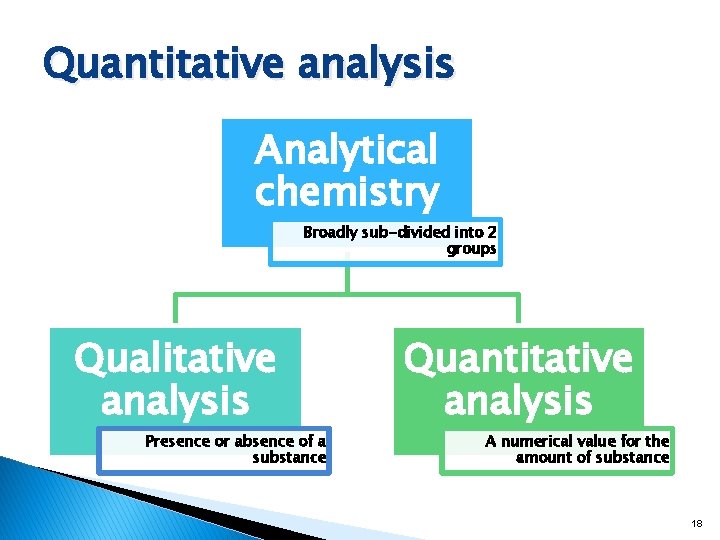
Quantitative analysis Analytical chemistry Broadly sub-divided into 2 groups Qualitative analysis Presence or absence of a substance Quantitative analysis A numerical value for the amount of substance 18

Quantitative analysis �A numerical value � Most common wet technique for analysing how much of a substance is present is by titration. � A titration uses a solution of known concentration and volume to find out the amount and / or concentration of an unknown substance. � The solution that we know the concentration of is called a Standard Solution! 19

Using a standard solution in a titration � Key points: standard solution in the conical flask, unknown in the burette, just a few drops of indicator. � An indicator is a chemical that has at least two distinct colours, which change after the equivalence point reached to show the end-point of the reaction. 20

Practical Activity Safety First: Wear safety goggles Stand up Tuck your chair & bags under your desk Tie back long hair

A standard solution: �A standard solution is a solution of accurately known concentration. � A standard solution can be prepared by dissolving a primary standard in a suitable solvent (such as distilled water). � A primary standard is a soluble solid compound that is very pure, with a consistent formula that does not change on exposure to the atmosphere, and has a relatively high molar mass (contains big atoms and /or a lot of them in its formula) 22

Equivalence and end points � The equivalence point is reached when enough standard solution has been added to the unknown concentration of solution so that the right proportions have reacted together in accordance to the chemical equation. � The term "end point" is where the indicator changes colour, this is what we document and it is slightly after the equivalence point, as the indicator changes colour due to a slight excess of the standard solution being added by the burette. 23
- Slides: 23