Week 9 Plan for today Laboratory Hoods and

Week 9; Plan for today: Laboratory Hoods and other Safety Equipment Laboratory Emergencies Risk assessment of laboratory operations from legal point of view. Hazards of Mercury spills Fire prevention in the scientific labs How to use Fire Extinguisher properly and efficiently Presentation by Committee #6 Last class announcements

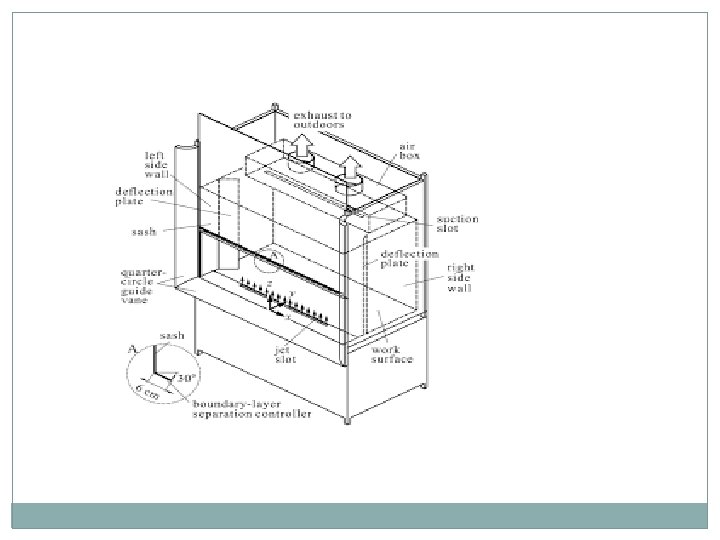
Laboratory Fume Hoods Can you name all parts? How do we know if the hood is working properly? What technical terms are used to determine hoods efficiency? Simple tests to check? Rules of using laboratory hoods.

Safety Showers and Eye Washes 29 CFR 1910. 151 OSHA Standard ANSI 2358. 1 When to use eye wash: Chemical splash Sharp objects in the eye(s) Thermal burns Dust and other foreign particles When to use a safety shower? 15 minutes continuously, always? ? Then what to do?

Eye wash OSHA requirements: Controlled flow to both eyes at low velocity to avoid eye injury Capacity: 1. 5 L /min for 15 minutes Nozzles protected from airborne contaminants Provide enough room to allow the eyelids to be held open with the hands Valve – water flow remains on, simple to operate, resistant to corrosion from potable water Units are easily accessible and visibly signed Maintenance and testing!

Other safety equipment: Respirators and masks – only if trained! First aid kits. Blankets for covering injured person Acid carriers, carts Spark free tools

Laboratory Emergencies …AND EMERGENCY EQUIPMENT.

Can you name any lab emergency circumstances? 1…………. 2…………. . 3…………

Emergency action how to Basic principle: #1. Save…… #2. Minimize…. Order of steps: 1……. 2……. . 3…….

CHEMICAL SPILLS Average ~12/year Key – to react quickly Who should clean up chemical spills? What are “Reportable spills” ? Spill clean up program should include: 1. Written procedures 2. Equipment/kits labeled: “Spill response kit” (gloves, goggles, pillows, pads, absorbent materials, bags, tongs, dust pan. . etc)

True or false? Chemical spill on a person should be treated differently than spill on a bench. The physical size of a chemical spill may not be a true indication of its severity. Evacuation is always necessary in case of every spill.

Specific spill-mix recipes: General spill-mix recipe: Na 2 CO 3/clay cat litter/dry sand (1: 1: 1) For acids: Na. HCO 3, Na 2 CO 3, very diluted Na. OH For bases: Acetic acid ~ 5% or less Br 2 and Iodine: absorbent first then 10% Na. HSO 3

3. Is your lab operation in compliance with the Regulations? REGULATORY EXPOSURE LIMITS OF “AIR-CONTAMINANTS”

Threshold Limit Values: PEL, TWA, STEL. . etc PEL = Permissible Exposure Limit (OSHA) The allowable maximum concentration (in ppm or mg/cu meter of air) which all workers may be repeatedly exposed every day (8 hours) without adverse health effect. TWA: Time Weighted Average = Average conc. For all workers /8 hours a day. STEL: Short Term Exposure Limit= max conc. of 15 minutes exposure no more than 4 times a day. Ceiling: max exposure, never to be exceed Units: ppm or mg/cu meter

Finding PEL-s in MSDS /SDS: Section 8 - Exposure Controls, Personal Protection Personal Protective Equipment Eyes: Wear chemical goggles. Skin: Wear appropriate protective gloves to prevent skin exposure. Clothing: Wear appropriate protective clothing to prevent skin exposure. Respirators: Follow the OSHA respirator regulations found in 29 CFR 1910. 134 or European Standard EN 149. Always use a NIOSH or European Standard EN 149 approved respirator when necessary. Exposure Limits : Chemical Name Acetone ACGIH 500 ppm TWA; 750 ppm STEL NIOSH OSHA - Final PELs 250 ppm TWA; 590 mg/m 3 TWA 1000 ppm TWA; 2400 mg/m 3 TWA

What do PEL-s tell us? The regulatory requirement by OSHA: for each substance with established PEL-s , a documented proof of sufficient protection for all workers is required. How is this done in reality? 2. All regulated substances (with listed OSHA PEL-s) need special precautions when used. How is it done in reality? 3. Using PEL-s in evaluating laboratory emergencies: accidental release, spills…etc. ? ? ? At which point the lab should be evacuated? Example: Let’s imagine Toluene spills on the bench in the lab of dimensions 6 x 5 x 3 m and there is no ventilation. Within 8 hours shift, all will be evaporated and workers will be breathing its vapors. Let’s calculate how much of this spill is “legally safe”/within OSHA exposure limit and would not necessarily required an evacuation. OSHA PEL (SDS) for exposure of Toluene: TWA 8= 200 ppm Conversion to mg/cu meter=ppm x MW / 24. 45; 200 ppm x 92. 14/24. 45= 753. 7 mg/cu meter Volume of the lab: 90 cu meters; Mass of Toluene, calculated: 90 x 754 mg/cu meter= 67860 mg=67. 9 g Volume of Toluene in ml: mass/density = 67. 9/0. 86=78. 95 ml ~ 80 ml 1.

Activity A theoretical case scenario: Acetone and Bromine spilled in our SU BA 605 lab. The ventilation system is down, the reagents totally evaporated and students are breathing the vapors. Let’s calculate how much (in ml) of each Acetone and Bromine (spilled and not cleaned up) would still be legally safe for 8 hours shift. What are your conclusions? Data provided: Volume of BA 605: 283 cu meters ( you are welcome to check if you wish) Acetone: FW = 58 g/mole; density ~0. 8 g/ml; Bromine: FW=180 g/mole; density~ 3. 1 g/ml

Is this the right order of a chemical spill response? 1. Containing a spill 2. Communicate w/others 3. Identify the spilled material 4. Neutralize or absorb the spill. 5. Eliminate additional hazard 6. Make decision about evacuation, if necessary. 7. Disposal of spilled material. 8. Placing warning sings

What would you do? A student stuck a paper clip into electrical receptacle on a lab bench and while falling down with severe shock symptoms, he/she broke 500 ml bottle full of Bromine. Imagine you are teaching this class of 20 students. What would you do and in which order?

Mercury spills – most painful!!! Mercury vapors are extremely poisonous! Average thermometer = 1 g of Hg If not cleaned up vaporize until reach equilibrium concentration = 20 mg/ m 3 This is ~ 200 times more than OSHA TLV= 0. 1 mg/m 3 !!!!!! Especially hazardous in confined spaces and small labs, without adequate ventilation.

Chemical and physical properties of Mercury: High density and surface tension, low viscosity = forms spheres, moving very quickly!!! Vaporizes easily at room temp, especially from small droplets!! Rate of evaporation rises with temperature never use mercury thermometers in ovens!!! Resistant to cleaning solution, insoluble in water, common solvents, alkalis and H 2 SO 4 Dissolves in conc. HNO 3 Forms amalgam with Zinc dust

Mercury spill clean-up methods: Let’s watch short video Specialized glass vacuum system ( never regular vacuum cleaner!!) very expensive! Small Hg clean up kits. Cover with Zinc dust or Sulfur, let it stand for 24 hours, scoop it –> Extremely Hazardous Waste

Control measures when using Hg Use hoods, glove boxes and secondary containers. When transferring from one container to another – cover w/oil or water. Always keep Hg in tightly closed container. Do we really need to use Mercury thermometers in laboratories? How to handle Hg thermometers?

Fires in laboratories PRESENTATION BY COMMITTEE #6

How to use fire extinguisher properly? Few things to consider before using it Do you know? P…………. A…………. S…………. What do we have in our labs? what’s in them?

Last class questions and messages Homework #3: due June 2, 4: 30 pm (I am off until May 31) Field trip on June 3, check Canvas often for details; (10 points) Class evaluation , need screen - shot or any other proof (5 points) Final: due June 8 ( Thursday), at midnight , there will be no extensions!!! And only one take!!! Questions ? ?
- Slides: 26