Week 6 Thermodynamics Properties Tables Self Reading Assignment
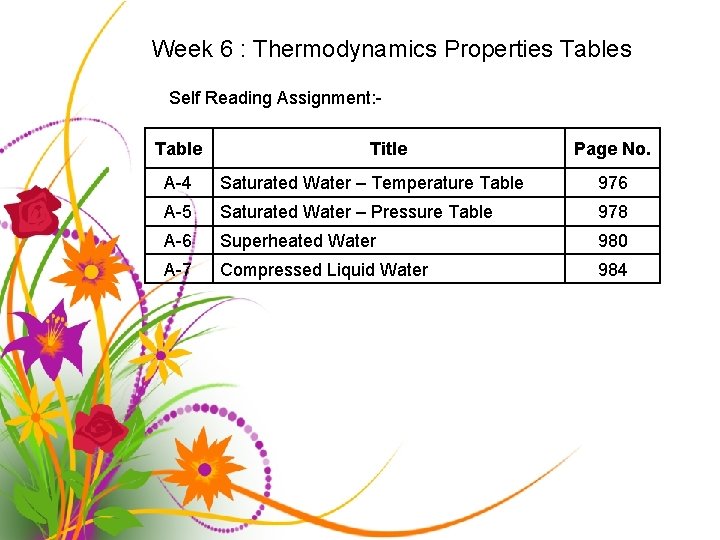
Week 6 : Thermodynamics Properties Tables Self Reading Assignment: Table Title Page No. A-4 Saturated Water – Temperature Table 976 A-5 Saturated Water – Pressure Table 978 A-6 Superheated Water 980 A-7 Compressed Liquid Water 984
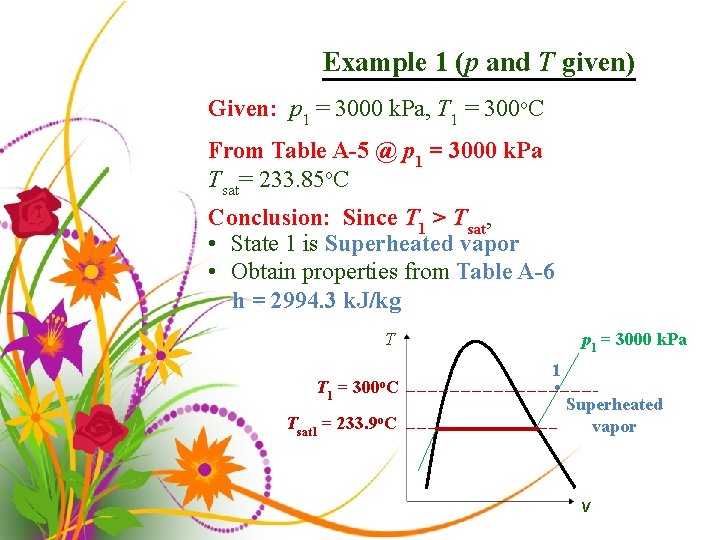
Example 1 (p and T given) Given: p 1 = 3000 k. Pa, T 1 = 300 o. C From Table A-5 @ p 1 = 3000 k. Pa Tsat= 233. 85 o. C Conclusion: Since T 1 > Tsat, • State 1 is Superheated vapor • Obtain properties from Table A-6 h = 2994. 3 k. J/kg p 1 = 3000 k. Pa T T 1 = Tsat 1 = 300 o. C 233. 9 o. C 1 • Superheated vapor v
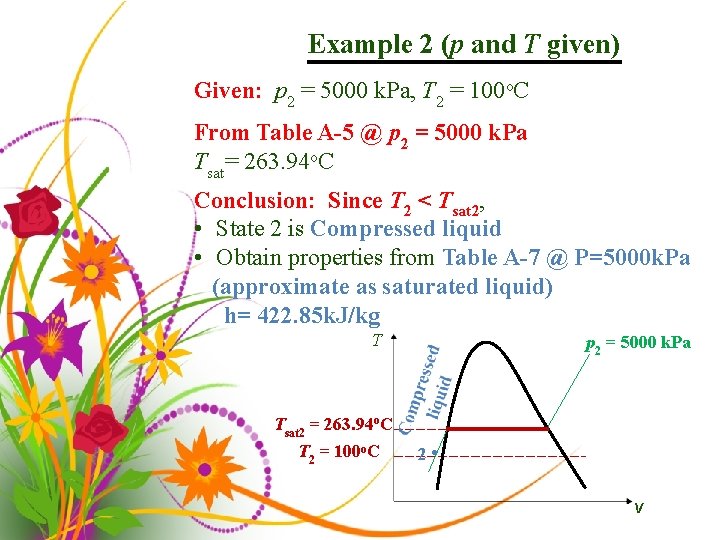
Example 2 (p and T given) Given: p 2 = 5000 k. Pa, T 2 = 100 o. C From Table A-5 @ p 2 = 5000 k. Pa Tsat= 263. 94 o. C Conclusion: Since T 2 < Tsat 2, • State 2 is Compressed liquid • Obtain properties from Table A-7 @ P=5000 k. Pa (approximate as saturated liquid) h= 422. 85 k. J/kg T p 2 = 5000 k. Pa Tsat 2 = 263. 94 o. C T 2 = 100 o. C 2 • v

Example 3 (p and T given) Given: p 3 = 500 k. Pa, T 3 = 151. 83 o. C From Table A-5 @ p 3 = 500 k. Pa Tsat= 151. 83 o. C Conclusion: Since T 3 = Tsat 3, • State 3 is Two-phase liquid-vapor mixture • Need another property to obtain properties p 3 = 500 k. Pa T Tsat=3 T== 151. 83 o. C Two-phase liquid-vapor mixture v
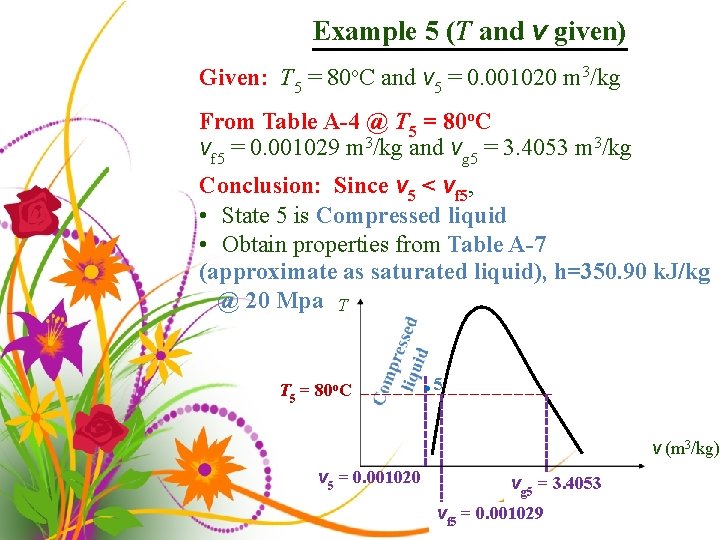
Example 5 (T and v given) Given: T 5 = 80 o. C and v 5 = 0. 001020 m 3/kg From Table A-4 @ T 5 = 80 o. C vf 5 = 0. 001029 m 3/kg and vg 5 = 3. 4053 m 3/kg Conclusion: Since v 5 < vf 5, • State 5 is Compressed liquid • Obtain properties from Table A-7 (approximate as saturated liquid), h=350. 90 k. J/kg @ 20 Mpa T T 5 = 80 o. C • 5 v (m 3/kg) v 5 = 0. 001020 vg 5 = 3. 4053 vf 5 = 0. 001029
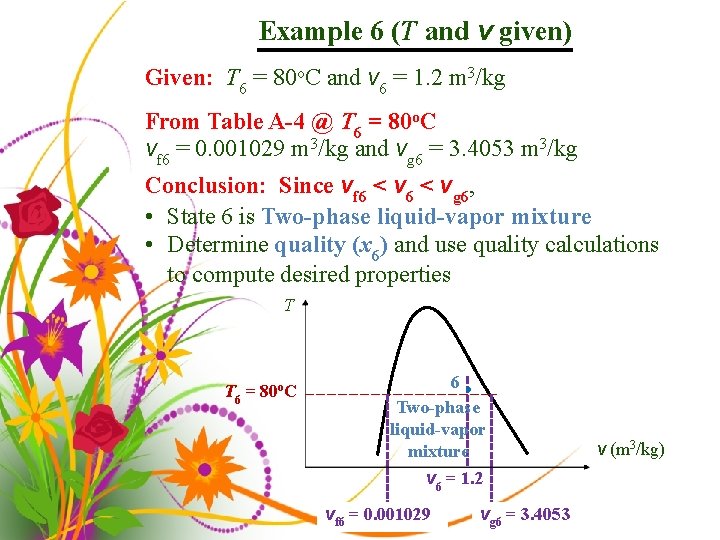
Example 6 (T and v given) Given: T 6 = 80 o. C and v 6 = 1. 2 m 3/kg From Table A-4 @ T 6 = 80 o. C vf 6 = 0. 001029 m 3/kg and vg 6 = 3. 4053 m 3/kg Conclusion: Since vf 6 < vg 6, • State 6 is Two-phase liquid-vapor mixture • Determine quality (x 6) and use quality calculations to compute desired properties T T 6 = 80 o. C 6 • Two-phase liquid-vapor mixture v 6 = 1. 2 vf 6 = 0. 001029 vg 6 = 3. 4053 v (m 3/kg)
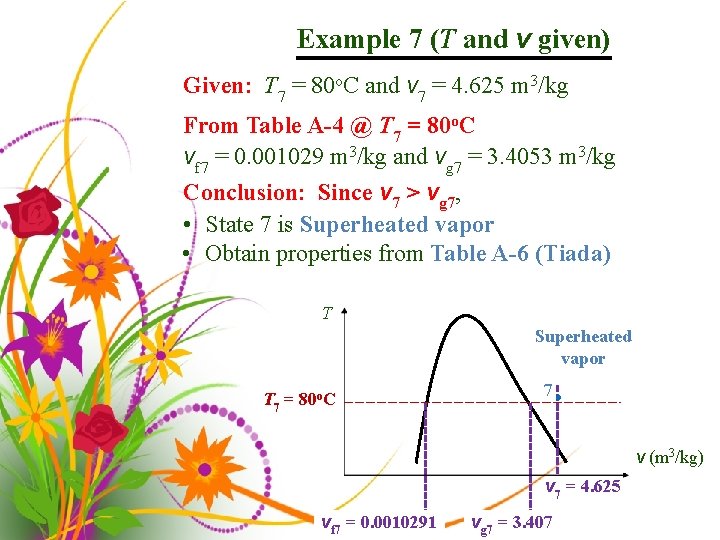
Example 7 (T and v given) Given: T 7 = 80 o. C and v 7 = 4. 625 m 3/kg From Table A-4 @ T 7 = 80 o. C vf 7 = 0. 001029 m 3/kg and vg 7 = 3. 4053 m 3/kg Conclusion: Since v 7 > vg 7, • State 7 is Superheated vapor • Obtain properties from Table A-6 (Tiada) T Superheated vapor T 7 = 80 o. C 7 • v (m 3/kg) v 7 = 4. 625 vf 7 = 0. 0010291 vg 7 = 3. 407

Example 11 (T and x given) Given: T 11 = 80 o. C and x 11 = 0. 6 Conclusion: • State 11 is Two-phase liquid-vapor mixture • Use quality calculations with saturated liquid (f) and saturated vapor (g) values from Table A-2 to compute desired properties From Table A-2 @ T 11 = 80 o. C vf 11 = 0. 001029 m 3/kg and vg 11 = 3. 4053 m 3/kg T V 11 = vf 11 +11 ( x 11 = 0. 6 T 11 = 80 o. C 11 • Two-phase liquid-vapor mixture V 11 =2. 044 m 3/kg v (m 3/kg) v 11 = 2. 044 vf 11 = 0. 001029 g 11 vg 11 = 3. 4053 x f 11 v)

Homework Please do your homework individually before the next class. 1. 2. 3. 4. 6 -22 6 -39 6 -42 6 -44 Study Smart & Do your homework
- Slides: 9