Week 22 Chemistry Percent Composition Empirical Formulas Molecular


![Agenda Warm Up [6 minutes] Pre-Lab/Expectations [7 minutes] Bubble Gum Lab [18 minutes] Answer Agenda Warm Up [6 minutes] Pre-Lab/Expectations [7 minutes] Bubble Gum Lab [18 minutes] Answer](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-3.jpg)














![Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-22.jpg)












![Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-35.jpg)












![Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-49.jpg)

















![Agenda Warm Up [6 minutes] Notes/Examples Guided Quiz [11 minutes] Practice [9 minutes] [20 Agenda Warm Up [6 minutes] Notes/Examples Guided Quiz [11 minutes] Practice [9 minutes] [20](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-74.jpg)

















- Slides: 93

Week 22 Chemistry Percent Composition, Empirical Formulas, Molecular Formulas

Warm Up: 3 Minutes Stay in your own seat Record your Learning Target You should be working SILENTLY A certain bag of M&M’s has 8 blue candies, 7 red candies, 4 green candies, and 6 yellow candies. What percentage of the bag is blue M&M’s? Do the warm up in One. Note
![Agenda Warm Up 6 minutes PreLabExpectations 7 minutes Bubble Gum Lab 18 minutes Answer Agenda Warm Up [6 minutes] Pre-Lab/Expectations [7 minutes] Bubble Gum Lab [18 minutes] Answer](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-3.jpg)
Agenda Warm Up [6 minutes] Pre-Lab/Expectations [7 minutes] Bubble Gum Lab [18 minutes] Answer Key [ 4 minutes] Modeling You Tracking [5 minutes] Track [10 minutes] Closing [3 Minutes]

Bubble Gum Lab

Problem What percentage of Double Bubble Gum is composed of sugar?

Pre-Lab Question According to the nutritional manufacturer, each piece of gum has a mass of 6 g with 5 g of sugar. Using this information, what percent of the gum is sugar by mass?

Lab Expectations Work in assigned groups Raise your hand if you have a question No gum should be left on the desks

General information: How to do the lab 1. Find the mass of your piece of gum before chewing (include the wrapper) 2. Chew your gum for ~5 minutes 3. Find the mass of your piece of gum after chewing Gum should be placed in wrapper before putting it on mass balance 4. Complete analysis and post-lab questions

Bubble Gum Lab You each need One Piece of Gum and your Brain Good Luck!!! 15 Minutes

Answer Key As Mr. Ghosh goes through the answers, highlight the ones you got wrong 1. A 12. B 2. C 13. B 3. A 14. A 4. D 15. C 5. D 16. A 6. C 17. C 7. A 18. A 8. D 19. A 9. C 20. B 10. B 21. C 11. B

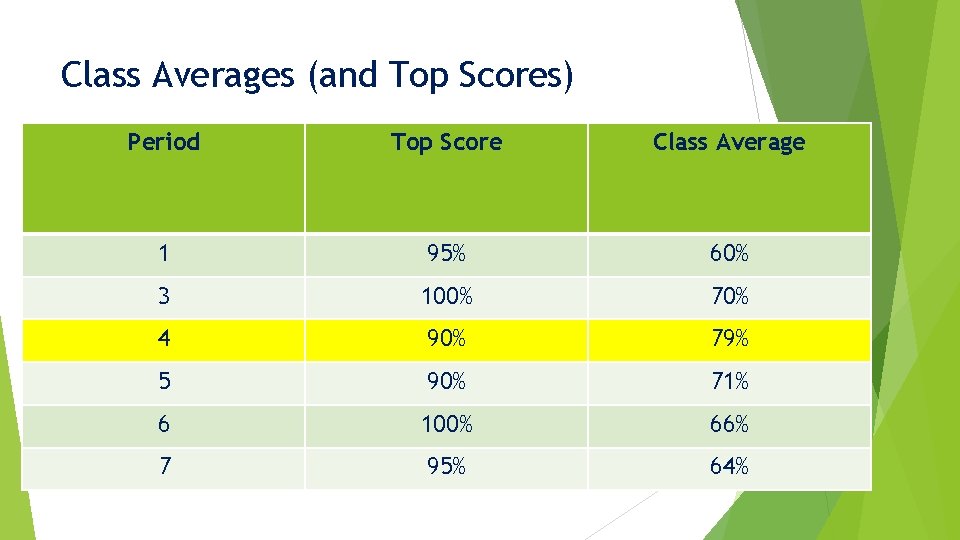
Class Averages (and Top Scores) Period Top Score Class Average 1 95% 60% 3 100% 70% 4 90% 79% 5 90% 71% 6 100% 66% 7 95% 64%

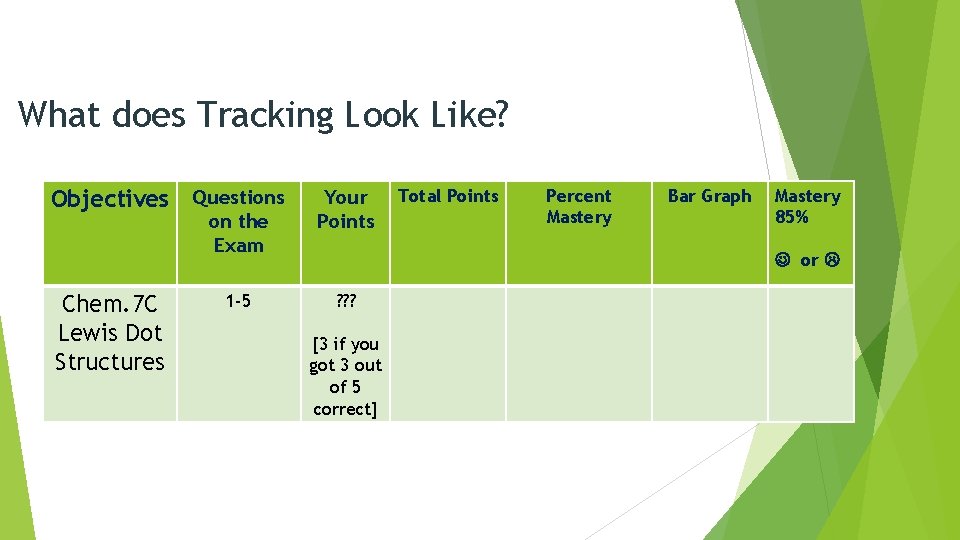
What does Tracking Look Like? Objectives Questions on the Exam Chem. 7 C Lewis Dot Structures Your Points Total Points Percent Mastery Bar Graph Mastery 85% or

What does Tracking Look Like? Objectives Questions on the Exam Chem. 7 C Lewis Dot Structures 1 -5 Your Points Total Points Percent Mastery Bar Graph Mastery 85% or

What does Tracking Look Like? Objectives Questions on the Exam Chem. 7 C Lewis Dot Structures 1 -5 Your Points ? ? ? [3 if you got 3 out of 5 correct] Total Points Percent Mastery Bar Graph Mastery 85% or

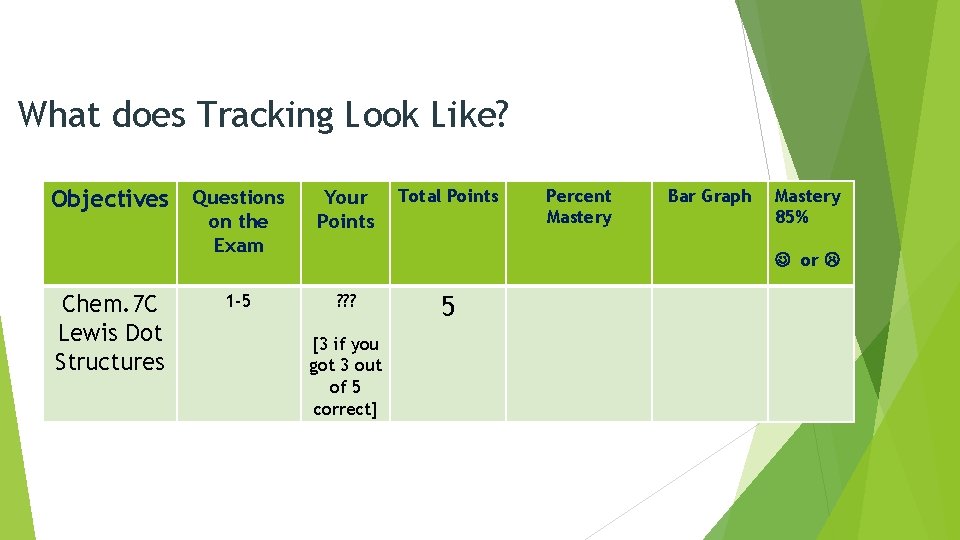
What does Tracking Look Like? Objectives Questions on the Exam Chem. 7 C Lewis Dot Structures 1 -5 Your Points Total Points ? ? ? 5 [3 if you got 3 out of 5 correct] Percent Mastery Bar Graph Mastery 85% or

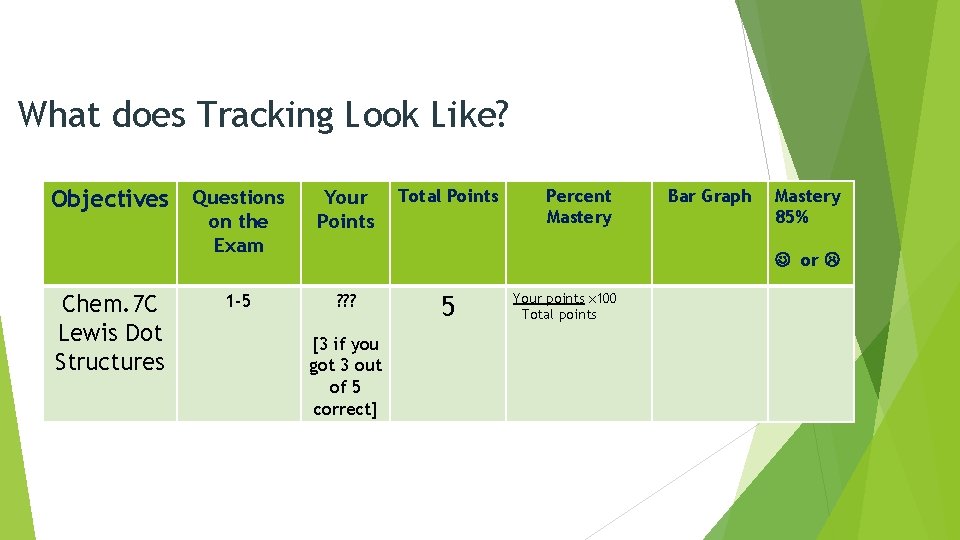
What does Tracking Look Like? Objectives Questions on the Exam Chem. 7 C Lewis Dot Structures 1 -5 Your Points ? ? ? [3 if you got 3 out of 5 correct] Total Points 5 Percent Mastery Your points x 100 Total points Bar Graph Mastery 85% or

Percent Mastery Cathy received 1 point out of 4 possible points. What is her percent mastery? 25% Your points x 100 Total points

What does Tracking Look Like? Objectives Questions on the Exam Chem. 7 C Lewis Dot Structures 1 -5 Your Points ? ? ? [3 if you got 3 out of 5 correct] Total Points 5 Percent Mastery Your points x 100 Total points Bar Graph Shade in your % mastery Mastery 85% or

Your Turn 1. Go to shschem. weebly. com (our class website) 2. Hover my page: Mr. Ghosh Homework Assignments ESL (or regular) Chemistry 3. Under Homework Assignments, there is a new one labeled “Unit 5 Tracking” 4. Download (and save) this and fill it out

Closing Explain the purpose of today’s lab

Warm Up: 4 Minutes Stay in your own seat You should be working SILENTLY 1. Log into your computer 2. Go to m. socrative. com 3. Enter room number: 230538 4. Finish all questions When you are finished, check your grades on PS Connect
![Agenda Warm Up 7 minutes NotesExamples Guided 15 minutes Practice 12 minutes Independent Closing Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-22.jpg)
Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing [3 Practice [15 minutes] Minutes]


What is % Composition? ? Percent by mass of each element in a compound

What is % Composition? ?

Example 1: A compound contains 2. 0 g of sodium, 3. 5 g of oxygen, and 0. 5 g of hydrogen. What is the percent composition of this compound?

Example 2: What is the percent composition by mass of the elements in the compound, Ag. NO 3?

Guided Practice Teacher: 1. Will show the problem on the board Students: 1. Take 24 seconds to identify your given and unknown. 2. Take 46 seconds to speak with your shoulder partner about the problem 3. Be ready to share when Mr. Ghosh says SWAG

Guided Practice 1: 9. 03 g Magnesium combines completely with 3. 48 g Nitrogen to form a compound. What is the percent composition of Nitrogen in this compound? 27. 82 % N

Guided Practice 2: What is the percent composition by mass of the elements in Mercury (II) Oxide? 92. 6% Hg 7. 4% O

Guided Practice 3: What is the percent composition by mass of the elements in K 2 CO 3? 56. 6% K 8. 7% C 34. 7% O

Independent Practice Take some time to practice finding the % composition of the following compounds Practice makes Perfect 85%

Closing How do you find the percent composition of an element in a compound?

Warm Up: 4 Minutes Stay in your own seat Write the Learning Target You should be working SILENTLY 1. Log into your computer 2. Go to m. socrative. com 3. Enter room number: 230538 4. Finish all questions When you are finished, check your grades on PS Connect
![Agenda Warm Up 7 minutes NotesExamples Guided 15 minutes Practice 12 minutes Independent Closing Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-35.jpg)
Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing [3 Practice [15 minutes] Minutes]

Goal For Today Student Given: Percent Composition: 79. 8% C, 20. 2% H Student Answer: CH 3

Empirical Formulas Shows the lowest number ratio of atoms in a compound empirical formula of glucose = CH 2 O

Example 1: What is the empirical formula for a chemical compound that is 79. 8% C and 20. 2% H?

How to Compose the Empirical Formula? Step 1: Convert Percent to Grams Step 2: Convert Grams to Moles by dividing grams by the atomic mass of each element. 79. 8% C and 20. 2% H

How to Compose the Empirical Formula? Step 3: Divide both molar quantities by the smallest mole quantity to determine the subscript for each element in the formula 79. 8% C and 20. 2% H

Example 2: What is the empirical formula for a compound that is 81. 7% Be and 18. 3% H?

Guided Practice Teacher: 1. Will show the problem on the board Students: 1. Take 22 seconds to identify the elements involved. 2. Take 68 seconds to solve the problem with your shoulder partner 3. Be ready to share when Mr. Ghosh says SWAG

Guided Practice 1: What is the Empirical Formula of a compound that is 67. 6% Hg, 10. 8% S, and 21. 6 % O by mass? Hg. SO 4

Guided Practice 2: What is the Empirical Formula of a compound that is 88. 2 % Oxygen and 11. 8% Hydrogen by mass? H 2 O

Guided Practice 3: What is the empirical formula of a compound that is made from 50% Cesium and 50% Iodine by mass? Cs. I

Independent Practice 85% Practice makes Perfect

Closing What How is the empirical formula? do you compose the empirical formula?

Warm Up: 4 Minutes Stay in your own seat You should be working SILENTLY 1. Log into your computer 2. Go to m. socrative. com 3. Enter room number: 230538 4. Finish all questions When you are finished, check your grades on PS Connect
![Agenda Warm Up 7 minutes NotesExamples Guided 15 minutes Practice 12 minutes Independent Closing Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-49.jpg)
Agenda Warm Up [7 minutes] Notes/Examples Guided [15 minutes] Practice [12 minutes] Independent Closing [3 Practice [15 minutes] Minutes]

More Empirical Formulas!!

Writing Empirical Formulas Most of the time, our mole ratios come out close to a whole number (~. 9, ~. 1): Ratio from Calculator Examples: What we assume 1. 992 2 16. 00412 16 4. 962 5 6. 071 6 But what if our mole ratio comes out to something like 1. 501? What do we do then?

Example 1: What is the empirical formula for a chemical compound that is 49. 1% Be and 50. 9% N?

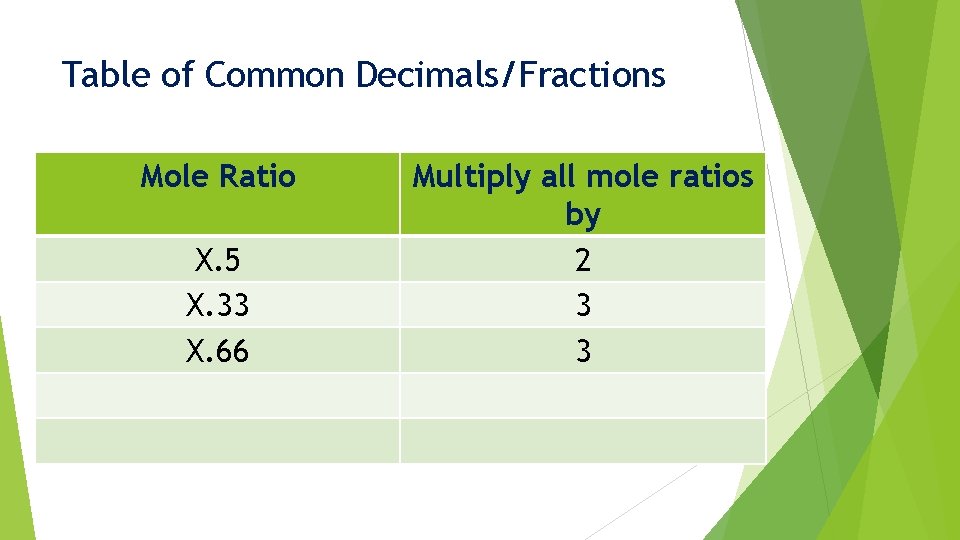
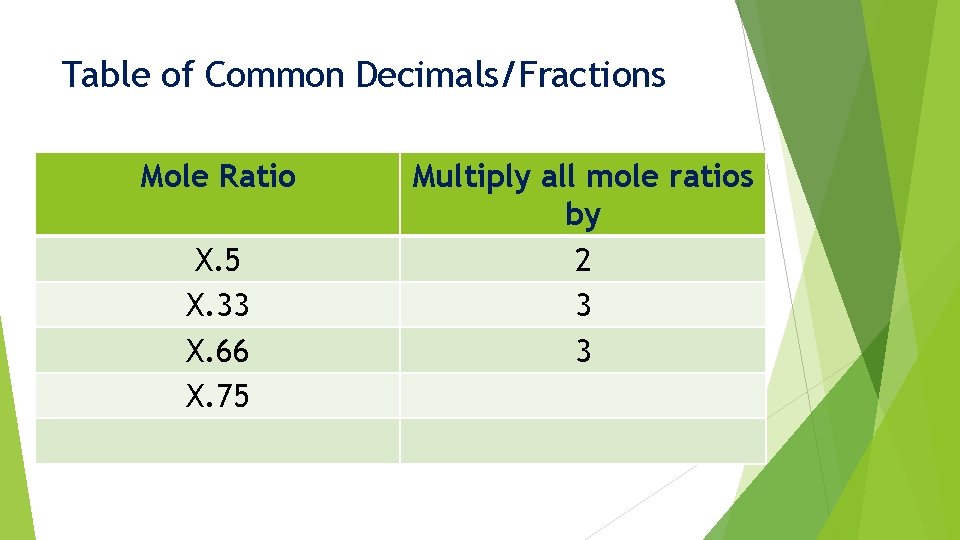
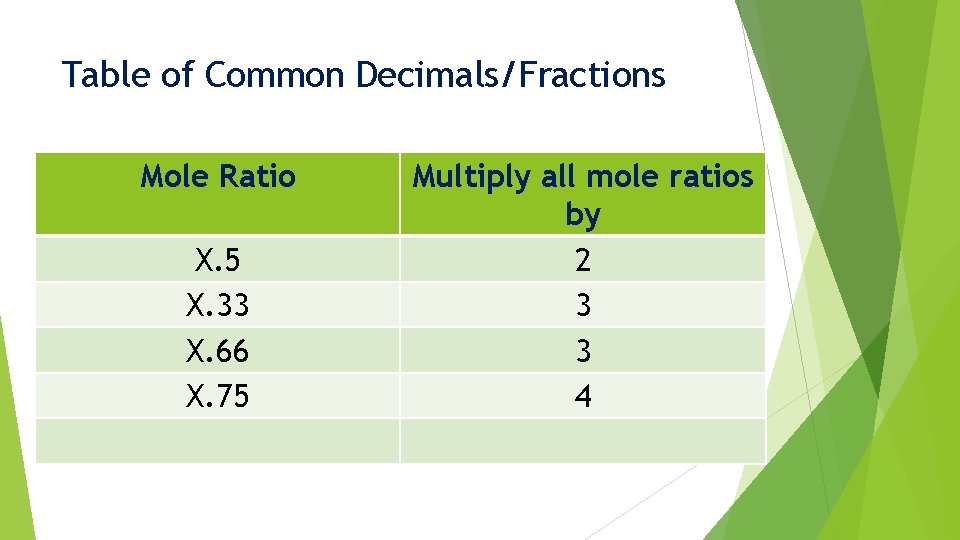
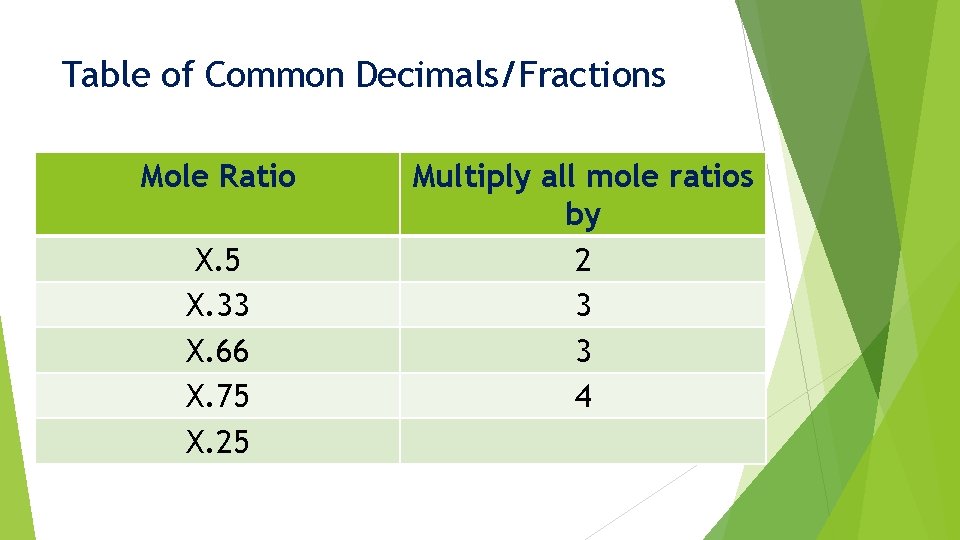
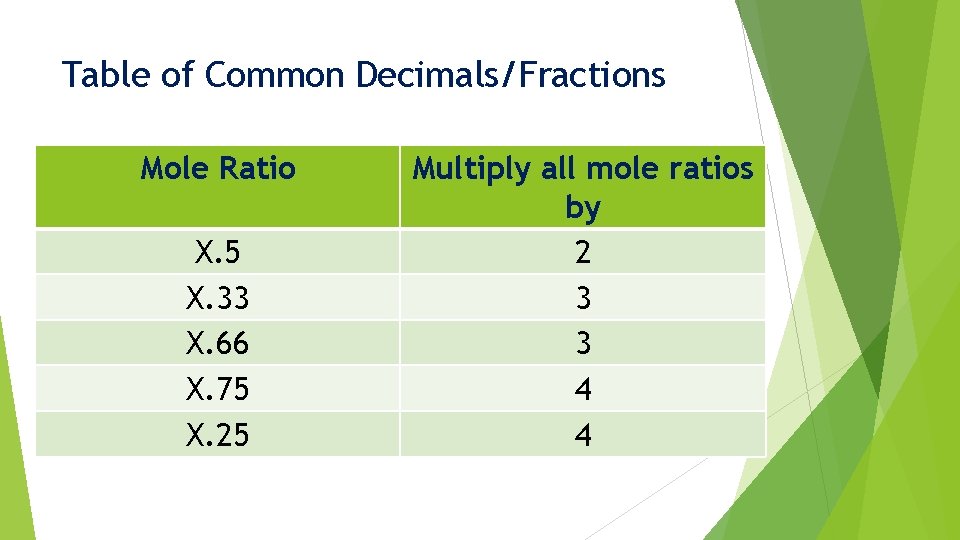
Common Decimals/Fractions When we find a mole ratio that ends up close to a common decimal/fraction: X. 33 X. 5 X. 75 X. 66 X. 25 We must multiply these ratio values by a specific number

Table of Common Decimals/Fractions Mole Ratio Multiply all mole ratios by

Table of Common Decimals/Fractions Mole Ratio X. 5 Multiply all mole ratios by

Table of Common Decimals/Fractions Mole Ratio X. 5 Multiply all mole ratios by 2

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 Multiply all mole ratios by 2

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 Multiply all mole ratios by 2 3

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 X. 66 Multiply all mole ratios by 2 3

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 X. 66 Multiply all mole ratios by 2 3 3

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 X. 66 X. 75 Multiply all mole ratios by 2 3 3

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 X. 66 X. 75 Multiply all mole ratios by 2 3 3 4

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 X. 66 X. 75 X. 25 Multiply all mole ratios by 2 3 3 4

Table of Common Decimals/Fractions Mole Ratio X. 5 X. 33 X. 66 X. 75 X. 25 Multiply all mole ratios by 2 3 3 4 4

Example 1: What is the empirical formula for a chemical compound that is 49. 1% Be and 50. 9% N?
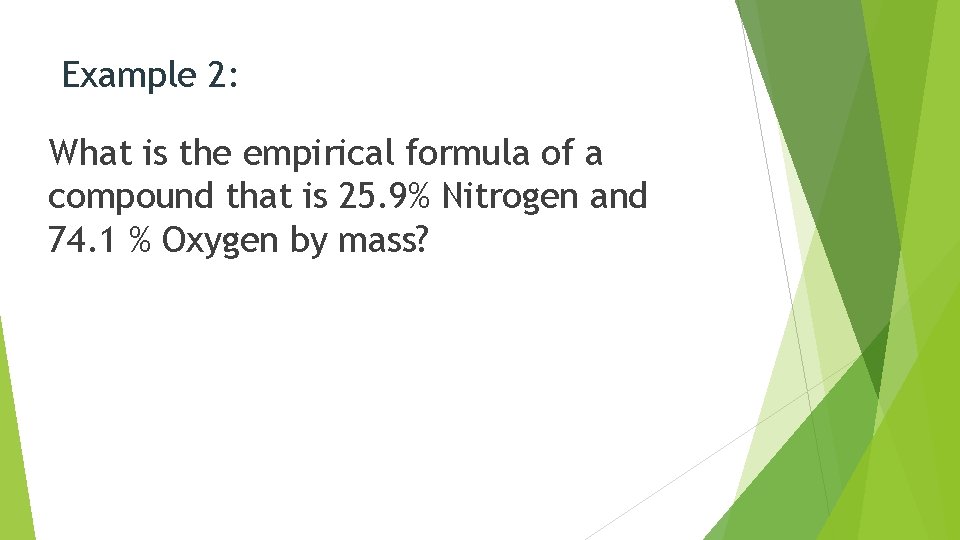
Example 2: What is the empirical formula of a compound that is 25. 9% Nitrogen and 74. 1 % Oxygen by mass?

Guided Practice Teacher: 1. Will show the problem on the board Students: 1. Take 22 seconds to identify the elements involved. 2. Take 68 seconds to solve the problem with your shoulder partner 3. Be ready to share when Mr. Ghosh says SWAG

Guided Practice 1: What is the Empirical Formula of a compound that is 25. 9% P and 74. 1% Cl? P 2 Cl 5

Guided Practice 2: What is the Empirical Formula of a compound that is 17. 4% Nitrogen and 82. 6% Fluorine? N 2 F 7

Guided Practice 3: What is the Empirical Formula of a compound that is made from 35. 9% Aluminum and 64. 1% Sulfur? Al 2 S 3

Independent Practice 85% Practice makes Perfect

Closing What How is the empirical formula? do you compose the empirical formula?

Warm Up: 4 Minutes Stay in your own seat Write the Learning Target You should be working SILENTLY 1. Log into your computer 2. Go to m. socrative. com 3. Enter room number: 230538 4. Finish all questions When you are finished, check your grades on PS Connect
![Agenda Warm Up 6 minutes NotesExamples Guided Quiz 11 minutes Practice 9 minutes 20 Agenda Warm Up [6 minutes] Notes/Examples Guided Quiz [11 minutes] Practice [9 minutes] [20](https://slidetodoc.com/presentation_image_h/09cbccc33e3488962e99354bcb24afd9/image-74.jpg)
Agenda Warm Up [6 minutes] Notes/Examples Guided Quiz [11 minutes] Practice [9 minutes] [20 minutes] Closing [1 Minute]

Goal For Today Student Given: Empirical Formula: CH 4 O Molar Mass of Molecular Formula: 96 g/mol Student Answer: Molecular Formula: C 3 H 12 O 3

Molecular Formulas Shows exactly how many of each atom is in a compound (unsimplified formula) Molecular formula of glucose = C 6 H 12 O 6

Check Point #1 Is CH 2 an empirical or a molecular formula? Empirical

Check Point #2 Is S 3 O 6 an empirical or a molecular formula? Molecular (Empirical Formula is SO 2)

Example 1: What is the molecular formula if the empirical formula is CH 4 O and the mass of the whole molecule is 96 g/mol?

How to Compose the Molecular Formula? Step 1: Find the Mass of Empirical Formula: CH 4 O the Empirical Formula Mass of Molecular Formula: 96 g/mol Step 2: Divide the mass of the Molecular Formula by the mass of the Empirical Formula

How to Compose the Molecular Formula? Step 3: Multiply the subscripts in the empirical formula by the answer from step 2. Empirical Formula: CH 4 O Mass of Molecular Formula: 96 g/mol

Example 2: Compose the molecular formula of the compound whose molar mass is 60. 0 g/mol and empirical formula is CH 4 N.

Guided Practice Teacher: 1. Will show the problem on the board Students: 1. Take 22 seconds to identify the elements involved. 2. Take 68 seconds to solve the problem with your shoulder partner 3. Be ready to share when Mr. Ghosh says SWAG

Guided Practice 1: Compose the molecular formula of the compound whose molar mass is 90 g/mol and empirical formula is CH 2 O. C 3 H 6 O 3

Guided Practice 2: Compose the molecular formula of the compound whose molar mass is 62 g/mol and empirical formula is CH 3 O. C 2 H 6 O 2

Independent Practice 85% Practice makes Perfect

Material Covered on Quiz Mole Calculations (1 and 2 step) Percent Composition Empirical Formulas

Goal To demonstrate mastery, we are shooting for 85%

Check Point What is your goal for this quiz? 85%

Expectations for Quiz Clear your desk of everything except a. . 1. Writing Utensil 2. Calculator Periodic Table is provided to you

Expectations Students will keep eyes on own paper Cheating will result in an automatic ZERO Students will remain SILENT for the duration of the quiz

Good Luck!!

Closing How was your quiz? What topics do you feel you still need review on?