Week 2 Mixing Substances What is Matter Matter

Week 2: Mixing Substances

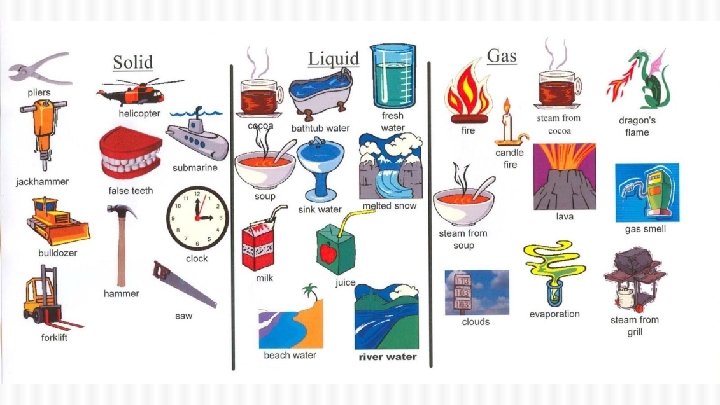
What is Matter? ● Matter is stuff! ● It is everything around you ● Scientifically, it is anything that takes up space and has mass ● Matter can be solid, liquid, or gas

Atoms and Elements ● Atoms are the smallest, most basic unit of matter ● There’s not just one kind of atom—there are 118!! (that we know of) ● Each of these is called an element


Compounds ● Compounds are atoms from different elements put together in a fixed combination ● These atoms are joined together by chemical bonds ● Compounds have different properties than the atoms that make it up

What is a Substance? ● A substance is a group of one type of matter ● It could be made of either an element or compound—but it has to be pure ● But it could be solid, liquid, or gas

Mixtures ● A mixture is a combination of two or more substances ● But, there is no chemical reaction -Each substance stays the same ● They just mix!

Homogenous vs. Heterogenous ● “Homogeneous” means uniform. ● In a homogeneous mixture, each substance is distributed evenly throughout and looks similar ● “Heterogeneous” means varied. ● In a heterogeneous mixture, the substances are visible, dissimilar, and irregularly distributed

Solutions ● A solution is a homogenous mixture made up of a solvent and one or more solutes ● The solvent is a substance that dissolves another substance that it is mixed with ● The solute is the substance that is dissolved

Suspension ● A suspension is a heterogeneous mixture that consists large particles spread throughout a liquid ● The particles are visible ● Some suspensions will settle, others will continue to be suspended

Vocabulary Review/List 1. Matter - anything that takes up space and has mass 1. Compound - 2 or more CHEMICALLY combined elements (not easily separated from each other) ex: water (H 2 O), CO 2 1. Substance - A substance is a group of one type of matter 1. Mixture - a mixture is a combination of two or more substances where there is no chemical combination or reaction. 2. Heterogeneous - substances are visible, dissimilar, and irregularly distributed 3. Homogenous - each substance is distributed evenly throughout and looks similar 1. Solution - a homogeneous mixture made up of a solvent and one or more solutes 2. Solvent - a substance that dissolves another substance that it is mixed with 3. Solute - the substance that is dissolved 4. Suspension - a heterogeneous mixture that consists large particles spread throughout a liquid
- Slides: 12