Webinar An introduction to NHS dmd and SNOMED

Webinar : An introduction to NHS dm+d and SNOMED CT UK Drug Extension Ian Spiers Pharmaceutical Terminology Specialist Data Standards and Products

Contents History of NHS dm+d and Use Cases dm+d 5 box data model NHS standard SNOMED CT UK Drug Extension Release and dm+d website
Information: Paper to IT Paper held Information Electronically held Information New Drugs Discontinued Product Change Price Drug Name Change Drug Name New Drugs HELP *July 13 th 2012 NHS dictionary of medicines and devices (dm+d) has 188, 527* drug concepts

Why a dictionary is needed? “There is a lack of standardisation in the UK in describing medicines, appliances and medical devices, in how such descriptions are organised, and in linking knowledge required for decision support to these descriptions” Section 3. 21

What will be done? “Work will begin on a national project to develop the UK Standard Clinical Products Reference Source (UKCPRS), bringing together all existing initiatives associated with the coding of drugs etc” Information for Health Section 3. 22

UKCPRS • The initial products of the Programme: – PCDD (Primary Care Drug Dictionary) • PPA (now NHS Prescription Services) – SCDD (Secondary Care Drug Dictionary) • NHSIA (now NHS CFH) – MDD (Medical Devices Dictionary) • NHSIA (now NHS CFH) • The three dictionaries (PCDD, SCDD, MDD) combined to become the NHS dictionary of medicines and devices (dm+d)

What the dm+d provides: • a unique stable identifier (code) for all drugs and devices • a standard term (description) for all drugs and devices • a structure in which this information will be held What the dm+d does NOT provide: • knowledge for decision support • application functionality

Use Cases Primary Care • GP Prescribing • GP Dispensing • Nurse Prescribing • NHS Dental Prescribing • Community Pharmacy Dispensing • GP Personal Administration • NHS Prescription Service (was PPD) Reimbursement • Decision Support links • Electronic Prescribing Guidelines Secondary Care • Prescribing – no product specified • Prescribing – product specified • In patient Dispensing • Out patient Dispensing • Tablets to Take Away (TTA) Dispensing • Administration • Patient Group Directive Protocols • Ward Stocks • Formulary/Protocol Development • Purchasing

dm+d 5 -box data model What are the dm+d concepts and how they fit in the 5 -box data model Take a look at the following description of tablets Paracetamol 500 mg Tablets This concept description is made up of Name Strength Form This concept is known as the Virtual Medicinal Product (VMP) in dm+d

dm+d 5 -box data model Lets look at some more descriptions that include Paracetamol in the name Paracetamol 500 mg soluble tablets Paracetamol 500 mg capsules Paracetamol 120 mg suppositories These are all VMP concepts in dm+d They all have a unique SNOMED CT identifier (Code) and a unique term (description) e. g. Paracetamol 500 mg Tablets has SNOMED CT Code 322236009

dm+d 5 -box data model In dm+d the VMPs Paracetamol 500 mg tablets Paracetamol 500 mg soluble tablets Paracetamol 500 mg capsules Paracetamol 120 mg suppositories are all associated with the higher level concept with the description Paracetamol This higher level concept is called a Virtual Therapeutic Moiety (VTM) As you can see one VTM may have many VMPs associated to it Each VTM also has a unique SNOMED CT identifier (Code) and a unique term (description) e. g. Paracetamol SNOMED CT Code 90332006
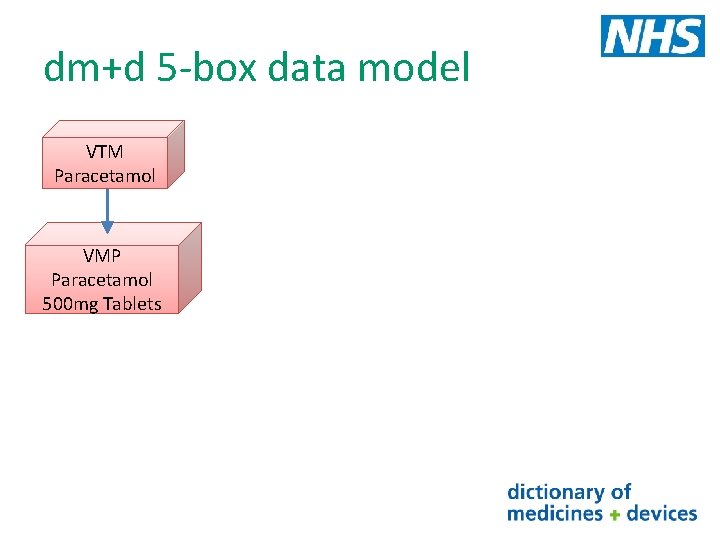
dm+d 5 -box data model VTM Paracetamol VMP Paracetamol 500 mg Tablets

dm+d 5 -box data model If we now look at the VMP : Paracetamol 500 mg tablets Many companies could/ make these tablets e. g. When we add brand supplier information to the description of a Paracetamol 500 mgatablets (Actavis VMP we describe concept known. UK as Ltd) the Actual Medicinal Paracetamol 500 mg tablets (Aspar Pharmaceuticals Ltd) Product (AMP) in dm+d Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) As you can see one VMP may have many AMPs associated to it Each AMP also has a unique SNOMED CT identifier (Code) and a term (description) e. g. Paracetamol 500 mg tablets (Aspar Pharmaceuticals Ltd) SNOMED CT Code 836511000001102
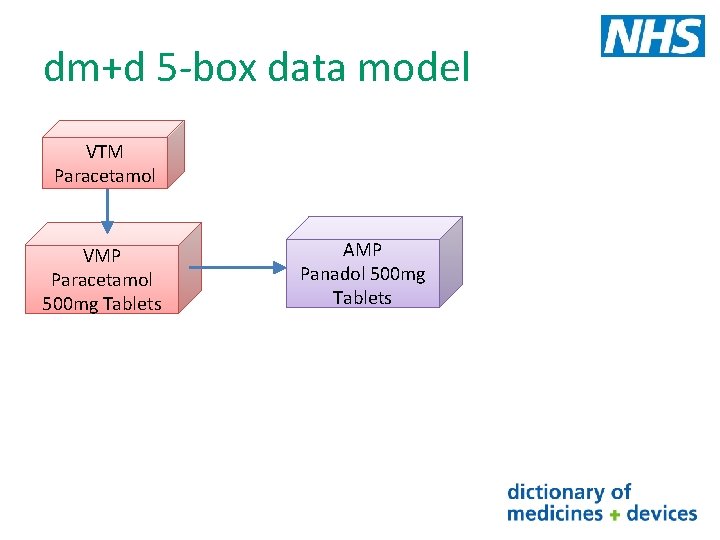
dm+d 5 -box data model VTM Paracetamol VMP Paracetamol 500 mg Tablets AMP Panadol 500 mg Tablets
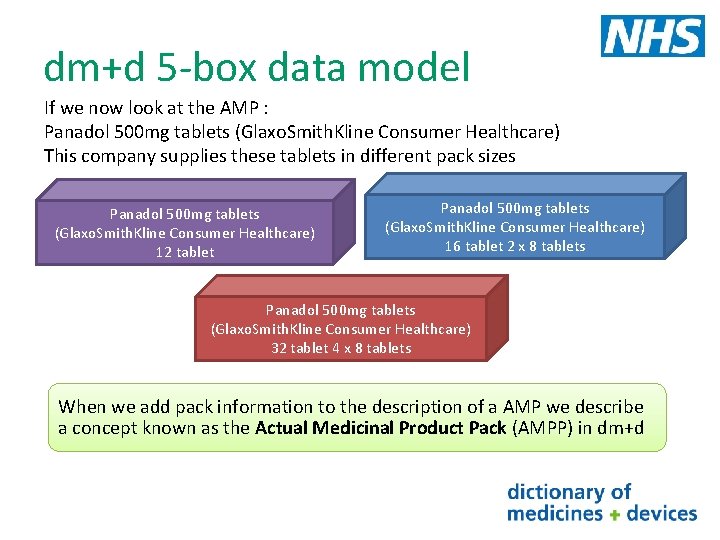
dm+d 5 -box data model If we now look at the AMP : Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) This company supplies these tablets in different pack sizes Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 12 tablet Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 16 tablet 2 x 8 tablets Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 32 tablet 4 x 8 tablets When we add pack information to the description of a AMP we describe a concept known as the Actual Medicinal Product Pack (AMPP) in dm+d

dm+d 5 -box data model So the AMP : Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) Can have many AMPPs Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 12 tablet Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 16 tablet 2 x 8 tablets Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 32 tablet 4 x 8 tablets Each AMPP also has a unique SNOMED CT identifier (Code) and a term (description) e. g. Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 12 tablet SNOMED CT Code 1437511000001107
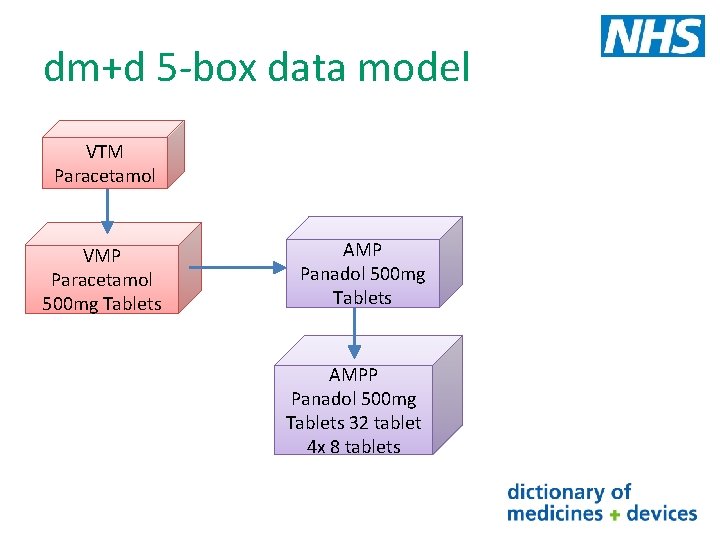
dm+d 5 -box data model VTM Paracetamol VMP Paracetamol 500 mg Tablets AMP Panadol 500 mg Tablets AMPP Panadol 500 mg Tablets 32 tablet 4 x 8 tablets

dm+d 5 -box data model Look at the following AMPPs Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 32 tablet 4 x 8 tablets Paracetamol 500 mg tablets (Teva UK Ltd) 32 tablet 4 x 8 tablets Paracetamol 500 mg tablets (Aspar Pharmaceuticals Ltd) 32 tablet 4 x 8 tablets They all have the same VMP Paracetamol 500 mg tablets and all have the same pack size The only difference is that they are supplied by different companies All the above AMPPs are associated with the higher level concept with the description Paracetamol 500 mg tablets 32 tablet This higher level concept is called a Virtual Medicinal Product Pack (VMPP)

dm+d 5 -box data model So the VMPP : Paracetamol 500 mg tablets 32 tablet Can have many AMPPs Panadol 500 mg tablets (Glaxo. Smith. Kline Consumer Healthcare) 32 tablet 4 x 8 tablets Paracetamol 500 mg tablets (Teva UK Ltd) 32 tablet 4 x 8 tablets Paracetamol 500 mg tablets (Aspar Pharmaceuticals Ltd) 32 tablet 4 x 8 tablets Each VMPP also has a unique SNOMED CT identifier (Code) and a unique term (description) e. g. Paracetamol 500 mg tablets 32 tablet SNOMED CT Code 1064711000001107
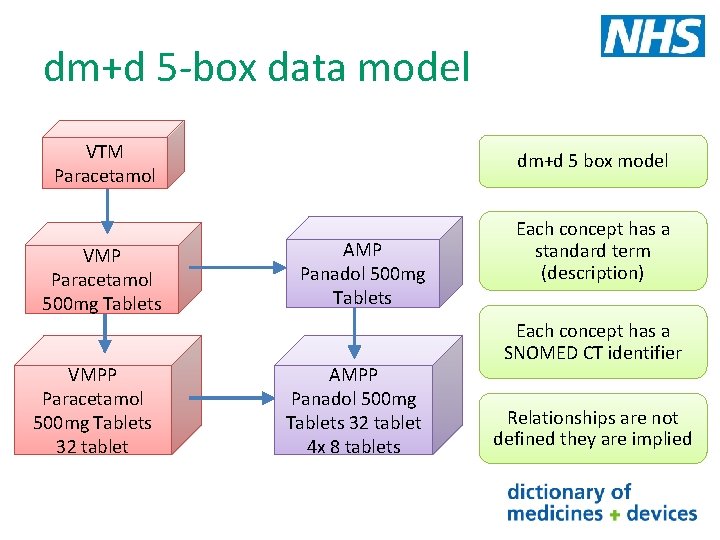
dm+d 5 -box data model VTM Paracetamol VMP Paracetamol 500 mg Tablets VMPP Paracetamol 500 mg Tablets 32 tablet dm+d 5 box model AMP Panadol 500 mg Tablets AMPP Panadol 500 mg Tablets 32 tablet 4 x 8 tablets Each concept has a standard term (description) Each concept has a SNOMED CT identifier Relationships are not defined they are implied

dm+d 5 -box data model Each dm+d concept has additional information associated to it (Attributes) e. g. VMP Paracetamol 500 mg Tablets Has the attribute of dose form populated with tablets Has the attribute of route populated with oral The different dm+d concepts have different attributes associated to them that are applicable to the concept level Some but not all of the attributes have SNOMED CT identifiers

Key dm+d documents dm+d Editorial Policy dm+d Data Model dm+d Technical Specifications Implementation guidance for both primary and secondary care

NHS Standard SNOMED CT as a Fundamental Standard • SNOMED CT has been approved as a Fundamental Standard by Information Standards Board for Health and Social Care (ISB Ha. SC). The Information Standards Notice (ISN) issued to notify this approval signals that SNOMED CT will be mandated for coded business interchanges between electronic healthcare record systems which support individual patient care by 2015. • Where pharmaceutical products are required to be represented then dm+d should be used.

NHS Standard dm+d as a ISB Fundamental Standard • The Information Standards Board (ISB) approved dm+d as NHS Standard for communicating medicines information (an Interoperability Standard). • Electronic systems that exchange or share information about medicines relating directly to a patients care must adhere to the Standard by using dm+d identifiers when transferring information and should also use dm+d descriptions • For licensed medicines only at this stage. Other content within dm+d such as medical devices are excluded in terms of complying with the Standard although users can use all the content within dm+d • Health and Care Organisations, System Suppliers and Pharmaceutical companies must comply with requirements in standard by 30 th June 2017 • The Electronic Prescription Service, Summary Care Record and Electronic Yellow Card Reporting already meet the requirements of this standard • For further information on the ISB standard http: //www. isb. nhs. uk/library/standard/84

Information Strategy The Power of Information: Putting all of us in control of the health and care information we need • DH Strategy document makes specific reference to the use of SNOMED CT and dm+d • It recommends that where IT systems reference medicines they use dm+d for coded information • Standardising on a single set of identifiers means that IT systems can more easily interoperate and information more readily shared

4 Key Points about dm+d • dm+d concepts have unique identifiers and associated textual descriptions • dm+d consists of a 5 -box data model • All dm+d concepts and some attributes use SNOMED CT concept identifiers • It is designed for both primary and secondary care

• Any questions? Please put yourself on mute when not talking. Many Thanks.

SNOMED CT UK Drug Extension • dm+d and SNOMED CT have a different structure and release format • SNOMED CT UK Drug Extension is produced to provide seamless links from SNOMED CT root node to dm+d concepts through one terminological structure • Mainly a technical transform which utilises some but not all information from dm+d • Defines relationships between concepts and has additional concept classes • Released 4 weekly
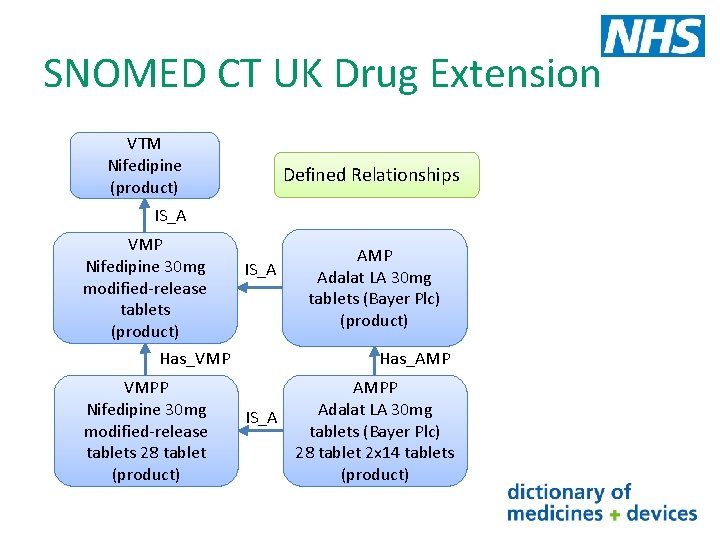
SNOMED CT UK Drug Extension VTM Nifedipine (product) Defined Relationships IS_A VMP Nifedipine 30 mg modified-release tablets (product) Has_VMP VMPP Nifedipine 30 mg modified-release tablets 28 tablet (product) IS_A AMP Adalat LA 30 mg tablets (Bayer Plc) (product) Has_AMP AMPP Adalat LA 30 mg IS_A tablets (Bayer Plc) 28 tablet 2 x 14 tablets (product)
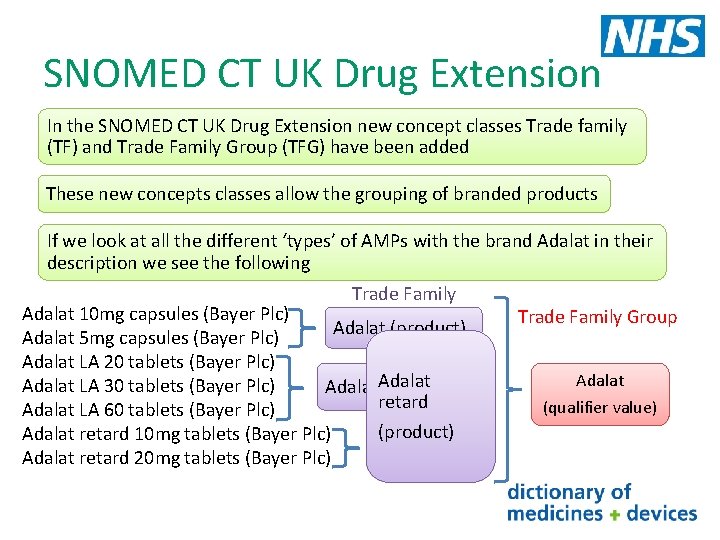
SNOMED CT UK Drug Extension In the SNOMED CT UK Drug Extension new concept classes Trade family (TF) and Trade Family Group (TFG) have been added These new concepts classes allow the grouping of branded products If we look at all the different ‘types’ of AMPs with the brand Adalat in their description we see the following Trade Family Adalat 10 mg capsules (Bayer Plc) Adalat (product) Adalat 5 mg capsules (Bayer Plc) Adalat LA 20 tablets (Bayer Plc) Adalat LA 30 tablets (Bayer Plc) Adalat LA (product) retard Adalat LA 60 tablets (Bayer Plc) (product) Adalat retard 10 mg tablets (Bayer Plc) Adalat retard 20 mg tablets (Bayer Plc) Trade Family Group Adalat (qualifier value)
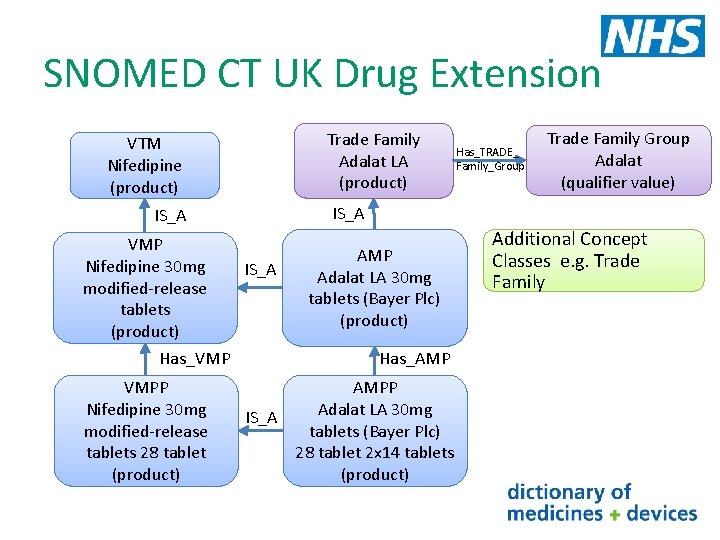
SNOMED CT UK Drug Extension Trade Family Adalat LA (product) VTM Nifedipine (product) Has_VMP VMPP Nifedipine 30 mg modified-release tablets 28 tablet (product) Trade Family Group Adalat (qualifier value) IS_A VMP Nifedipine 30 mg modified-release tablets (product) Has_TRADE_ Family_Group IS_A AMP Adalat LA 30 mg tablets (Bayer Plc) (product) Has_AMP AMPP Adalat LA 30 mg IS_A tablets (Bayer Plc) 28 tablet 2 x 14 tablets (product) Additional Concept Classes e. g. Trade Family
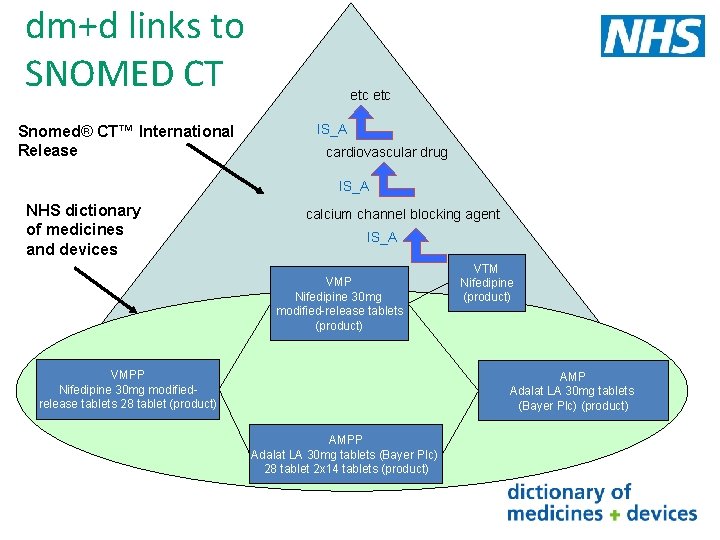
dm+d links to SNOMED CT Snomed® CT™ International Release etc IS_A cardiovascular drug IS_A NHS dictionary of medicines and devices calcium channel blocking agent IS_A VMP Nifedipine 30 mg modified-release tablets (product) VMPP Nifedipine 30 mg modifiedrelease tablets 28 tablet (product) VTM Nifedipine (product) AMP Adalat LA 30 mg tablets (Bayer Plc) (product) AMPP Adalat LA 30 mg tablets (Bayer Plc) 28 tablet 2 x 14 tablets (product)
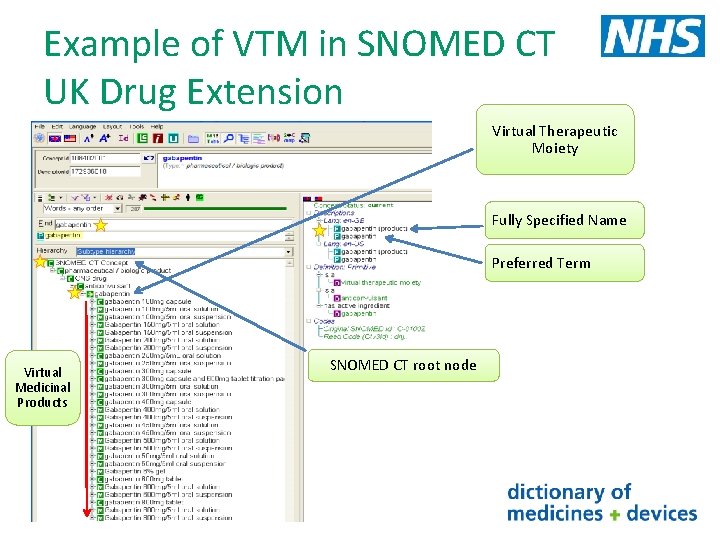
Example of VTM in SNOMED CT UK Drug Extension Virtual Therapeutic Moiety Fully Specified Name Preferred Term Virtual Medicinal Products SNOMED CT root node

4 Key Points about SNOMED CT UK Drug Extension • dm+d information forms the basis of the SNOMED CT UK Drug Extension • Not all dm+d information is used (e. g. Price) • Additional concept classes and information are added (e. g. Trade Family) • Defines relationships between concepts (only implied in the dm+d XML)

Release Schedule • dm+d data updated and released weekly in XML format Number of medicine and device concepts typically added per week: Concept type VTM VMPP AMPP added per week in dm+d 0 - 5 concepts 5 – 50 concepts 20 – 150 concepts 25 – 500 concepts 50 – 500 concepts Total No. in dm+d (13/07/12) 2, 286 concepts 15, 589 concepts 22, 683 concepts 68, 988 concepts 78, 981 concepts Total 188, 527 concepts • SNOMED CT UK Drug Extension released every 4 weeks • Both released via the TRUD website and FTP site – Technology Reference Data Update Distribution Service (TRUD) – www. uktcregistration. nss. cfh. nhs. uk
dm+d release on TRUD
SNOMED CT UK Drug Extension release on TRUD SNOMED CT UK Drug Extension Editorial Policy
dm+d website

dm+d information • dm+d Browser http: //www. ppa. org. uk/systems/pcddbrowserv 2_3 new/browser. jsp • dm+d Helpdesk – nhsbsa. dmdenquiries@nhs. net – 0845 850 0001 • In-Demand - is a web based system which provides pharmaceutical companies the facility to submit their product information changes and prices directly to the NHS Business Services Authority https: //indemand. medicines. org. uk/login/default. aspx? Return. Url=%2 f. Default. aspx

Governance and Issues • Information on dm+d governance structures, issues process and the current issues log can be found on the dm+d website: http: //www. dmd. nhs. uk/dmdgovernance/index. html http: //www. dmd. nhs. uk/issueslog/index. html

Key Benefits NHS dm+d • • • Stable, well defined national dictionary that is freely available published editorial policy and stringent governance process for change control continually updated and released weekly dm+d concepts have unique SNOMED CT identifiers and associated textual descriptions Allows unambiguous recording of medicines and devices for prescribing, dispensing, administration and reimbursement Enables unambiguous electronic data transfer SNOMED CT UK Drug Extension • • Contains dm+d concepts with defined relationships to the international terminology SNOMED CT, allows international interoperability New concept classes : Trade Family and Trade Family Group supports brand prescribing and enhanced allergy recording

• Any questions? Please put yourself on mute when not talking. Many Thanks. A recorded webinar can be found on the TRUD website under UKTC Education and Training Resources as well as other webinars on SNOMED CT
- Slides: 42