Weather Studies Introduction to Atmospheric Science American Meteorological




















- Slides: 24

Weather Studies Introduction to Atmospheric Science American Meteorological Society Chapter 5 Air Pressure Credit: This presentation was prepared for AMS by Michael Leach, Professor of Geography at New Mexico State University - Grants

Case-in-Point § Mount Everest – World’s tallest mountain – 8850 m (29, 035 ft) – Same latitude as Tampa, FL – Due to declining temperature with altitude, the summit is always cold § January mean temperature is -36 °C (-33 °F) § July mean temperature is -19° C (-2 °F) – Shrouded in clouds from June through September § Due to monsoon winds – November through February – Hurricane-force winds § Due to jet stream moving down from the north – Harsh conditions make survival at the summit difficult § Very thin air § Wind-chill factor – Most ascents take place in May 2

Driving Question § What is the significance of horizontal and vertical variations in air pressure? – Air pressure is an element of weather we do not physically sense as readily as temperature and humidity changes – This chapter examines: § The properties of air pressure § How air pressure is measured § The reasons for spatial and temporal variations in air pressure 3

Defining Air Pressure § Air exerts a force on the surface of all objects it contacts – The air is a gas, so the molecules are in constant motion – The air molecules collide with a surface area in contact with air § The force of these collisions per unit area is pressure § Dalton’s Law – total pressure exerted by mixture of gases is sum of pressures produced by each constituent gas § Air pressure depends on: – Mass of the molecules and kinetic molecular energy § Air pressure can be thought of as the weight of overlying air acting on a unit area – Weight is the force of gravity exerted on a mass § Weight = (mass) x (acceleration of gravity) § Average sea-level air pressure – 1. 0 kg/cm 2 (14. 7 lb/in. 2) § Air pressure acts in all directions – That is why structures do not collapse under all the weight 4

Air Pressure Measurement § A mercury thermometer employs air pressure to support a column of mercury in a tube § Air pressure at sea level will support the mercury to a height of 760 mm (29. 92 in. ) § Height of the mercury column changes with air pressure § Adjustments required for: – The expansion and contraction of mercury with temperature – Gravity variations with latitude and altitude 5

Air Pressure Measurement § An aneroid barometer is less precise, but more portable than a mercury barometer § It has a chamber with a partial vacuum § Changes in air pressure collapse or expand the chamber § This moves a pointer on a scale calibrated equivalent to mm or in. of mercury § New ones are piezoelectric – depend on the effect of air pressure on a crystalline substance § Home-use aneroid barometers often have a fair, changeable, and stormy scale – These should not be taken literally 6

Air Pressure Measurement § Forecasting uses air pressure and tendency values – changes over time § Barometers may keep a record of air pressure – These are called barographs 7

§ Units of length – Millimeters or inches § Inches typical for TV Air Pressure Units § Units of pressure – Pascal – worldwide standard § Metric scale § Sea-level pressure = – 101, 325 pascals (Pa) – 1013. 25 hectopascals (h. Pa) – 101. 325 kilopascals (k. Pa) – Bars – U. S. § A bar is 29. 53 inches of mercury § A millibar (mb) is the standard used on weather maps, meaning 1/1000 of a bar – Usual worldwide range is 970 – 1040 mb – Lowest ever recorded - 870 mb (Typhoon Tip in 1979) – Highest ever recorded – 1083. 8 mb (Agata, Siberia) 8

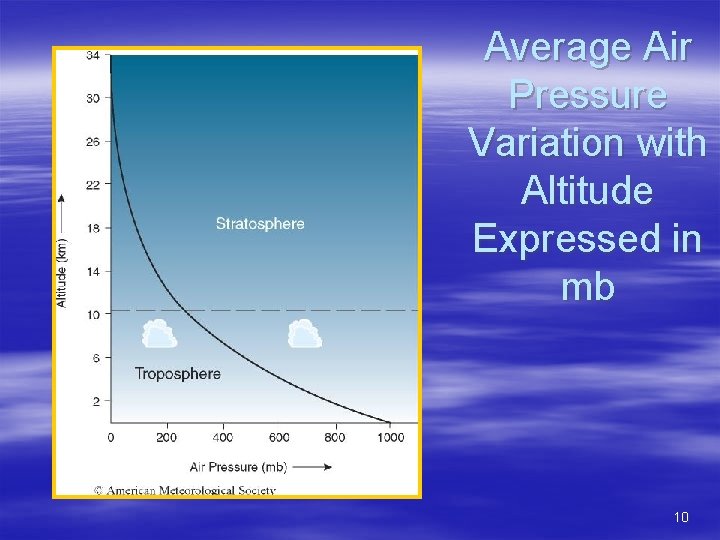
Variations in Air Pressure w/Altitude § Overlying air compresses the atmosphere – the greatest pressure is at the lowest elevations § § § Gas molecules are closely spaced at the surface Spacing increases with altitude At 18 km (11 mi), air density is only 10% of that at sea level § Because air is compressible, the drop in pressure with altitude is greater in the lower troposphere – Then it becomes more gradual aloft § Vertical profiles of average air pressure and temperature are based on the standard atmosphere – state of atmosphere averaged for all latitudes and seasons § Even though density and pressure drop with altitude, it is not possible to pinpoint a specific altitude at which the atmosphere ends – – – ½ the atmosphere’s mass is below 5500 m (18, 000 ft) 99% of the mass is below 32 km (20 mi) Denver, CO average air pressure is 83% of Boston, MA 9

Average Air Pressure Variation with Altitude Expressed in mb 10

Horizontal Variations in Air Pressure § Horizontal variations are much more important to weather forecasters than vertical differences – In fact, local pressures at elevations are adjusted to equivalent sea-level values – This shows variations of pressure in the horizontal plane – This is mapped by connecting points of equal equivalent sea-level pressure, producing isobars 11

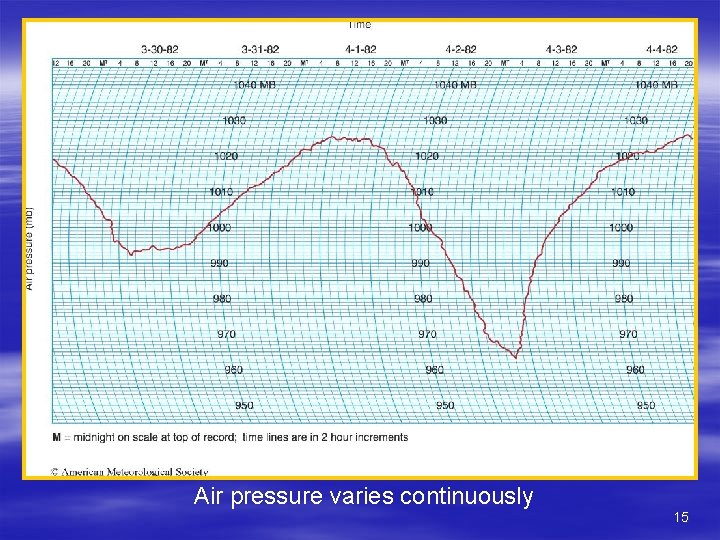
Horizontal Variations in Air Pressure § Horizontal changes in pressure can be accompanied by significant changes in weather § In middle latitudes, a continuous procession of different air masses brings changes in pressure and weather – Temperature has a much more pronounced affect on air pressure than humidity § In general, the weather becomes stormy when air pressure falls but clears or remains fair when air pressure rises 12

Horizontal Variations in Air Pressure § Influence of temperature and humidity – Rising air temperature = rise in the average kinetic energy of the individual molecules § In a closed container, heated air exerts more pressure on the sides – Density in a closed container does not change – No air has been added or removed § The atmosphere is not like a closed container – Heating the atmosphere causes the molecules to space themselves farther apart – This is due to increased kinetic energy – Molecules placed farther apart have a lower mass per unit volume, or density – The heated air is less dense, and lighter. 13

Horizontal Variations in Air Pressure § Influence of temperature and humidity, continued – Air pressure drops more rapidly with altitude in a column of cold air § Cold air is denser, has less kinetic energy, so the molecules are closer together – 500 mb surfaces represent where half of the atmosphere is above and half below by mass § This surface is at a lower altitude in cold air vs. in warm air – Increasing humidity decreases air density § The greater the concentration of water vapor, the less dense is the air due to Avogadro’s Law § We often refer to muggy air as heavy air, but the opposite is true – Muggy air only weighs heavily on our personal comfort factor 14

Air pressure varies continuously 15

Horizontal Variations in Air Pressure § Influence of temperature and humidity, continued – Cold, dry air masses are the densest § These generally produce higher surface pressures – Warm, dry air masses generally exert higher pressure than warm, humid air masses – These pressure differences create horizontal pressure gradients § Pressure gradients cause cold or warm air advection – Air mass modifications can also produce changes in surface pressures – Conclusion: local conditions and air mass advection can influence air pressure 16

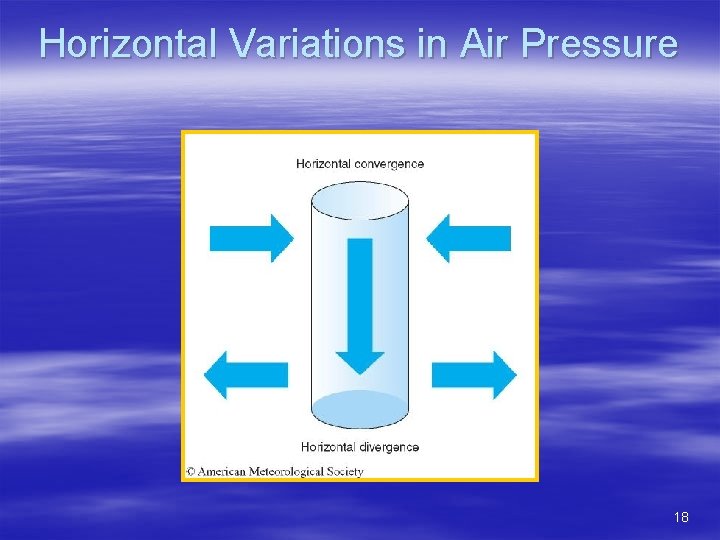
Horizontal Variations in Air Pressure § Influence of diverging and converging winds – Diverging = winds blowing away from a column of air – Converging = winds blowing towards a column of air – Diverging/converging caused by : § Horizontal winds blowing toward or away from some location (this chapter) § Wind speed changes in a downstream direction (Chapter 8) 17

Horizontal Variations in Air Pressure 18

Highs and Lows § Isobars are drawn on a map as previously discussed – U. S. convention – these are drawn at 4 -mb intervals (e. g. , 996 mb, 1000 mb, 1004 mb) § A High is an area where pressure is relatively high compared to the surrounding air § A Low is an area where pressure is relatively low compared to the surrounding air § Highs are usually fair weather systems § Lows are usually stormy weather systems – Rising air is necessary for precipitation formation – Lows are rising columns of air. Highs are sinking columns of air. 19

The Gas Law § We have discussed variability of temperature, pressure, and density → these properties are known as variables of state; their magnitudes change from one place to another across Earth’s surface, with altitude above Earth’s surface, and with time § The three variables of state are related through the ideal gas law, which is a combination of Charles’ law and Boyle’s law – The ideal gas law states that pressure exerted by air is directly proportional to the product of its density and temperature, i. e. pressure = (gas constant) x (density) x (temperature) 20

The Gas Law, Continued § Conclusions from the ideal gas law – Density of air within a rigid, closed container remains constant. Increasing the temperature leads to increased pressure – Within an air parcel, with a fixed number of molecules: – Volume can change, mass remains constant – Compressing the air increases density because its volume decreases – Within the same air parcel: – With a constant pressure, a rise in temperature is accompanied by a decrease in density. – Expansion due to increased kinetic energy increases volume – Hence, at a fixed pressure, temperature is inversely proportional to density 21

Expansional Cooling and Compressional Warming § Expansional cooling – when an air parcel expands, the temperature of the gas drops § Compressional warming – when the pressure on an air parcel increases, the parcel is compressed and its temperature rises § Conservation of energy – Law of energy conservation/1 st law of thermodynamics → heat energy gained by an air parcel either increases the parcel’s internal energy or is used to do work on the parcel – A change in internal energy is directly proportional to a 22 change in temperature

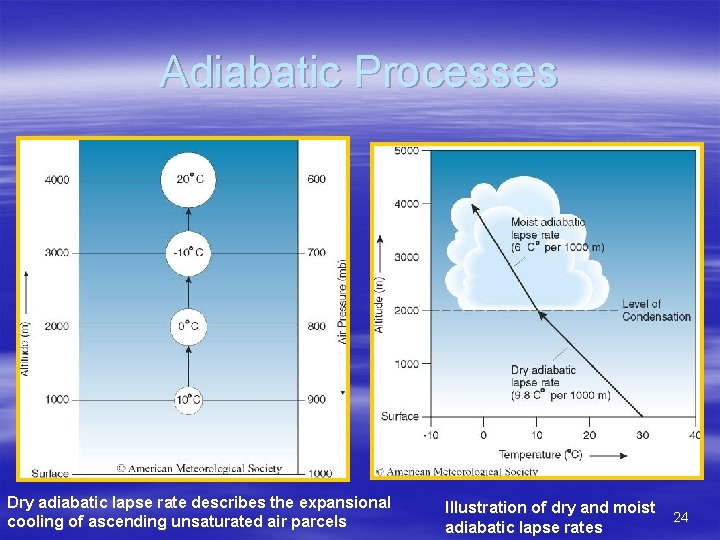
Adiabatic Processes § During an adiabatic process, no heat is exchanged between an air parcel and its surroundings – The temperature of an ascending or descending unsaturated parcel changes in response to expansion or compression only – Dry adiabatic lapse rate = 9. 8 C°/1000 m (5. 5 °F/1000 ft) – Once a rising parcel becomes saturated, latent heat released to the environment during condensation or deposition partially counters expansional cooling – Moist adiabatic lapse rate = 6 C°/1000 m (3. 3 °F/1000 ft) → this is an average rate 23

Adiabatic Processes Dry adiabatic lapse rate describes the expansional cooling of ascending unsaturated air parcels Illustration of dry and moist adiabatic lapse rates 24