Weak AcidStrong Base Titration Lab Sam White Pd

Weak Acid-Strong Base Titration Lab Sam White Pd. 2

Titrations are a neutralization reaction between a base and an acid n The 2 types of titrations you need for this lab are: n ¨ Strong Acid-Strong Base ¨ Weak Acid-Strong Base

Strong Acid-Strong Base n In a proper Strong Acid-Strong Base titration, the H+ and OH- are used up to create water in the following reaction: H+ + OHH 2 O n The ions attached to the H+ ion (like Cl-) and to the OHion (like Na+) form a soluble salt (like Na. Cl) which splits into ions in water. These become spectator ions in the reaction, and do not affect the p. H When done properly, this type of titration creates a solution with a p. H of 7, since the H+ and OHconcentrations are equal, and water is neutral. n
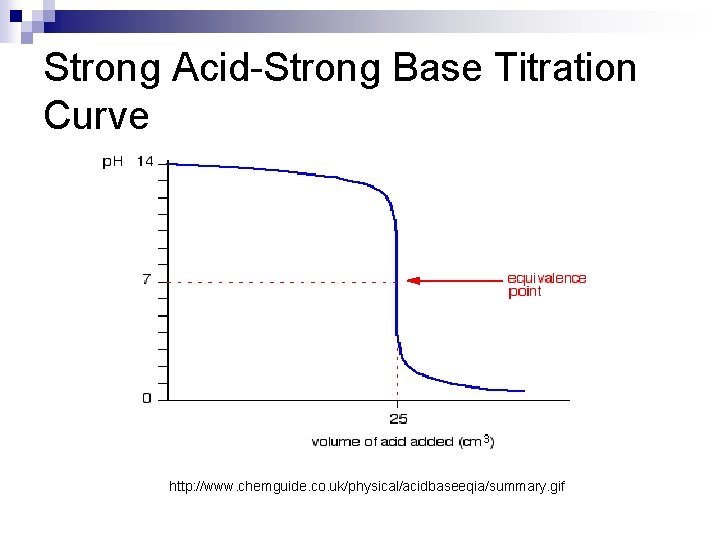
Strong Acid-Strong Base Titration Curve http: //www. chemguide. co. uk/physical/acidbaseeqia/summary. gif

Weak Acid-Strong Base n Weak Acid-Strong Base reactions follow this reaction: HA + OHH 2 O + A - n While the acid is fully neutralized (all H+ ions are used up to make water), the conjugate base of the acid (denoted generically as A-) remains. This means your titrated solution will have a p. H above 7 (usually somewhere between 8 -9) n
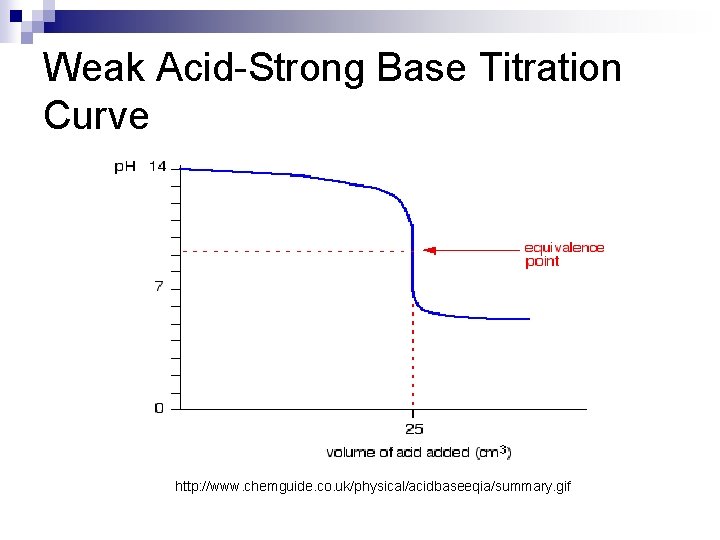
Weak Acid-Strong Base Titration Curve http: //www. chemguide. co. uk/physical/acidbaseeqia/summary. gif

Diprotic Acids So far we’ve only seen examples of monoprotic acids (acids with one H+ ion) n Weak diprotic acids (acids with two H+ ions) behave a little differently: n H 2 A + OHHA- + OH- n H 2 O + HAH 2 O + A-2 As you can see, weak diprotic acids must dissociate twice, resulting in unique titration curves
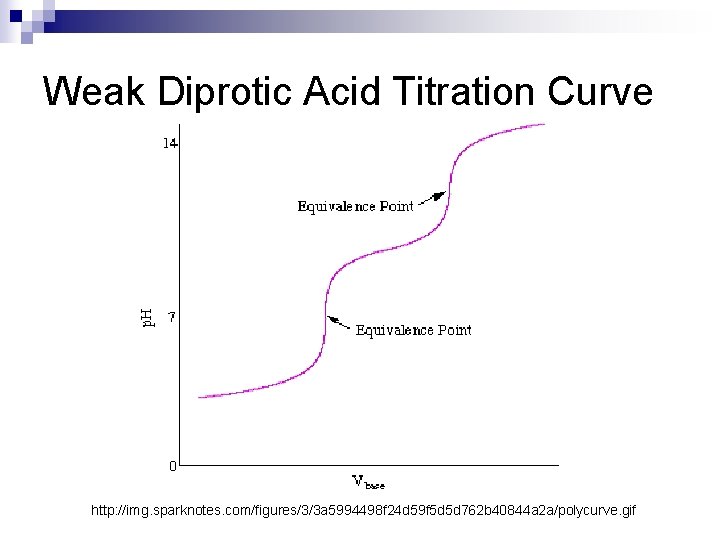
Weak Diprotic Acid Titration Curve http: //img. sparknotes. com/figures/3/3 a 5994498 f 24 d 59 f 5 d 5 d 762 b 40844 a 2 a/polycurve. gif

Purpose n To determine the concentration of a sample of an unknown weak acid through titration with Na. OH

Materials n n n n 50 m. L Buret Ring Stand Buret Clamp. 1 M Na. OH Ehrlenmeyer Flask Graduated Cylinder Acetic Acid Phenolphthalein
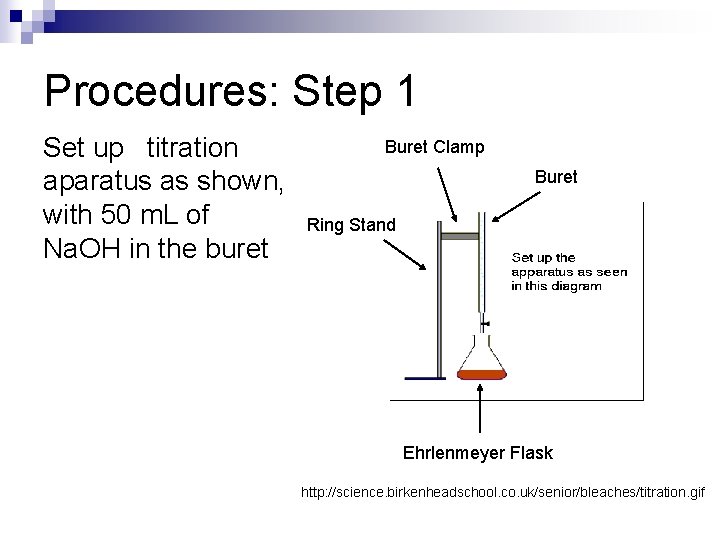
Procedures: Step 1 Set up titration aparatus as shown, with 50 m. L of Na. OH in the buret Buret Clamp Buret Ring Stand Ehrlenmeyer Flask http: //science. birkenheadschool. co. uk/senior/bleaches/titration. gif

Procedures: Step 2 Acid 25 m. L Acid Measure out 25 m. L of acetic acid. Add to Ehrlenmeyer Flask. From Clipart
Procedures: Step 3 Add 3 drops of Phenolphthalein indicator to the flask of acid. Acid Made From Clipart

Procedures: Step 4 Slowly add drops of Na. OH from the buret to the flask of acid. Make sure to stop when it is a light pink color. Before Stop Here Way too Far

Procedures: Step 5 Record the amount of Na. OH needed to neutralize the acid.
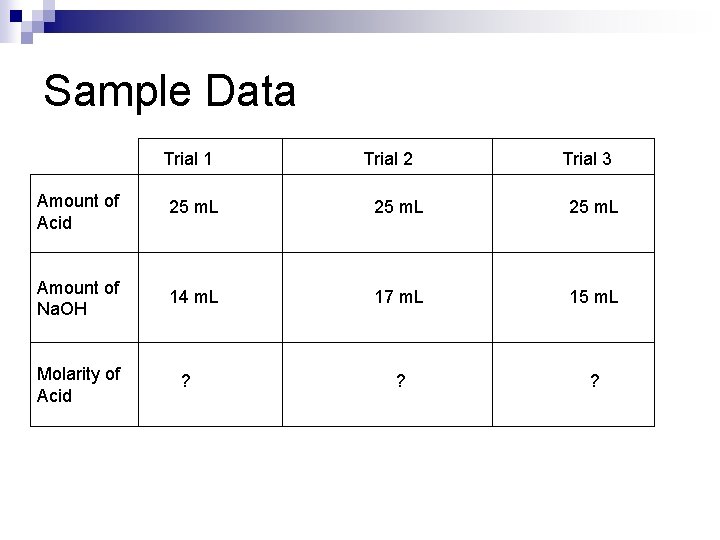
Sample Data Trial 1 Trial 2 Trial 3 Amount of Acid 25 m. L Amount of Na. OH 14 m. L 17 m. L 15 m. L ? ? Molarity of Acid ?

Calculations Moles of Na. OH: 0. 1 M = moles Na. OH Liters solution

Calculations Each trial Na. OH: 0. 1 M = moles Na. OH. 014 L moles of Na. OH =. 014 x. 1 =. 0014 0. 1 M = moles Na. OH. 017 L moles of Na. OH =. 017 x. 1 =. 0017 0. 1 M = moles Na. OH. 015 L moles of Na. OH =. 015 x. 1 =. 0015

Calculations Average moles of Na. OH: 0. 0014 + 0. 0017 + 0. 0015 = 0. 0046 / 3 = 0. 00153 moles of Na. OH

Calculations This means, on average, 0. 00153 moles of Na. OH were used in neutralization. This means, on average, there were 0. 00153 moles of H+ neutralized. This also means, on average, 0. 00153 moles of Acetic acid were used.

Calculations Average molarity of Acetic Acid: M = 0. 00153 moles 0. 025 L M = 0. 0621 This means the concentration of the acetic acid solution was 0. 0621 M

Bibliography n For 3 images on Procedure 4: http: //images. google. com/imgres? imgurl=http: //www. sciencebuddies. org/mentoring/project_i deas/Chem_img 030. jpg&imgrefurl=http: //www. sciencebuddies. org/science-fairprojects/project_ideas/Chem_p 045. shtml&usg=__Pf. ZM_64 ir. O 37 n. Sl. QQbvg. F 2 Gj. Gao=&h=40 0&w=300&sz=16&hl=en&start=21&tbnid=LNlcn. R 6 dl. BX 7 z. M: &tbnh=124&tbnw=93&prev=/ima ges%3 Fq%3 Dtitration%26 gbv%3 D 2%26 ndsp%3 D 20%26 hl%3 Den%26 sa%3 DN%26 start%3 D 20 ¨ http: //educ. queensu. ca/~science/main/concept/gen/g 09/N. %20 Sabet/titration 1. gif ¨ http: //www. titrations. info/img/phenolphthalein-s. jpg ¨
- Slides: 22