Weak acid strong base titrations Initial p H
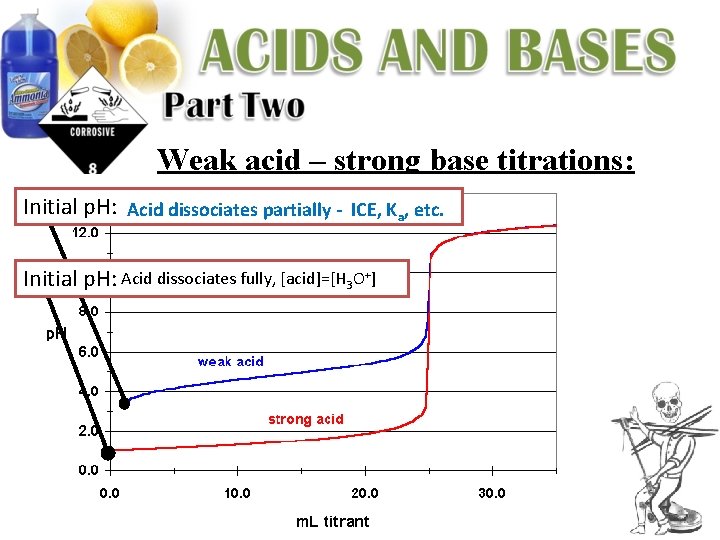
Weak acid – strong base titrations: Initial p. H: Acid dissociates partially - ICE, Ka, etc. Initial p. H: Acid dissociates fully, [acid]=[H 3 O+]

Weak acid – strong base titrations: Initial p. H: Acid dissociates partially - ICE, Ka, etc. If 0. 1 M Na. OH is being used to titrate a 0. 1 M solution of acetic acid (Ka = 1. 8 x 10 -5), calculate the following: 1. Initial p. H: (before titrating)
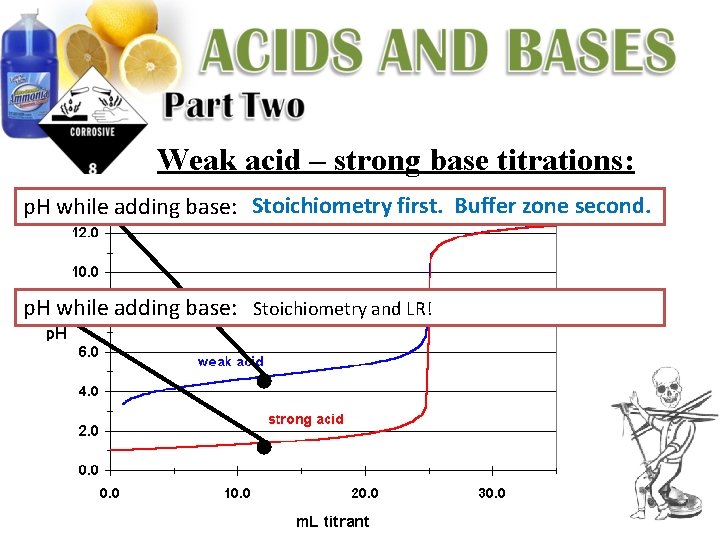
Weak acid – strong base titrations: p. H while adding base: Stoichiometry first. Buffer zone second. p. H while adding base: Stoichiometry and LR!

p. H while adding base: Stoichiometry first. Buffer zone second. If 0. 1 M Na. OH is being used to titrate a 0. 1 M solution of acetic acid (Ka = 1. 8 x 10 -5), calculate the following: 2. p. H while adding base: (before equivalence point) 50 m. L of HC 2 H 3 O 2 is titrated with 10 m. L of Na. OH

Weak acid – strong base titrations: Equivalence point: If acid and base are gone, why it slightly basic? ? ? Equivalence point: Point at which all moles of acid have reacted with all moles of base (p. H=7)

Equivalence point: If acid and base are gone, why it slightly basic? ? ? If 0. 1 M Na. OH is being used to titrate a 0. 1 M solution of acetic acid (Ka = 1. 8 x 10 -5), calculate the following: 3. p. H at equivalence point: 50 m. L of HC 2 H 3 O 2 is titrated with Na. OH to the equivalence point.

Weak acid – strong base titrations: p. H if “over-titrated”: Stoichiometry and LR! p. H if “over-titrated”: Excess strong base + conj. Base together

If 0. 1 M Na. OH is being used to titrate a 0. 1 M solution of acetic acid (Ka = 1. 8 x 10 -5), calculate the following: 4. p. H after equivalence point: (over-titrating) 50 m. L of HC 2 H 3 O 2 is titrated with 55 m. L of Na. OH p. H if “over-titrated”: Excess strong base + conj. Base together

Acids and Bases
- Slides: 9