We will become the world leading arterial stiffness

We will become the world leading arterial stiffness measurement company Marshall Ring, CEO May 2020 Canada 1
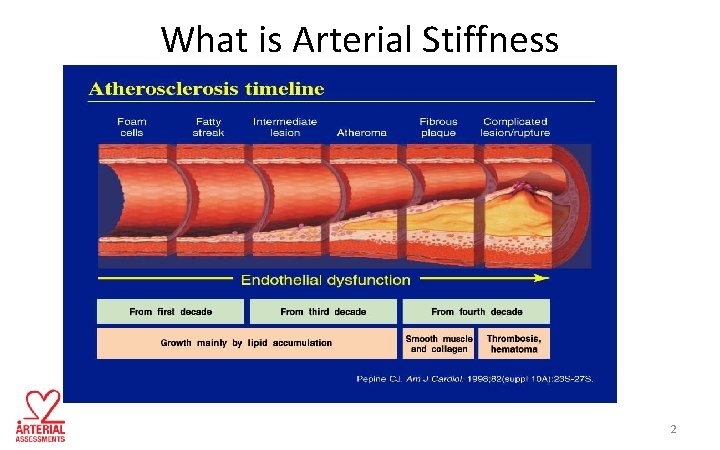
What is Arterial Stiffness 2

Current Problem and Opportunity • Measuring arterial stiffness is expensive, time consuming and requires centralized testing with trained technicians. • A quick, fast, easy, affordable test system which can be used outside of hospitals will deliver great value. 3

Our solution • A device able to reliably calculate the amount of atherosclerosis in a person. • Less expensive than current tests. • Easy to use and patients do not need to be tested in a hospital with skilled technicians. • Can test a person in half the time of current tests, but will still earn the full billing code rate. • Capital costs are 10% of competitive devices. 4

Product 1. Finger clip to secure pulse wave signal 2. Computer / smart phone for data input 3. Algos for big data and processing of signal 5
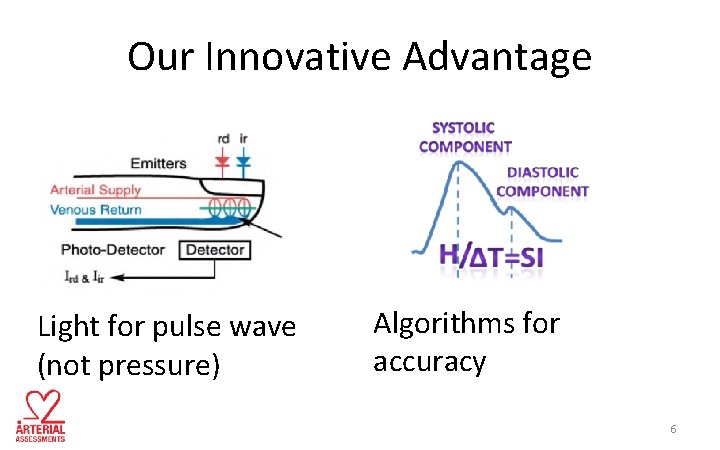
Our Innovative Advantage Light for pulse wave (not pressure) Algorithms for accuracy 6

Patents Pending • US application 13/825, 225 - effective filing date September 20, 2011 • Canada 2, 846, 650 – effective filing date September 20, 2011 • Europe 11826275. 7 effective filing date September 20, 2011 7

Business Model – Recurring Revenue • Move away from selling a capital expense and towards a recurring revenue model. • Confirmed Meinian will revenue share at $2 CDN per test. • Goal is build a business similar to the cell phone business model. 8
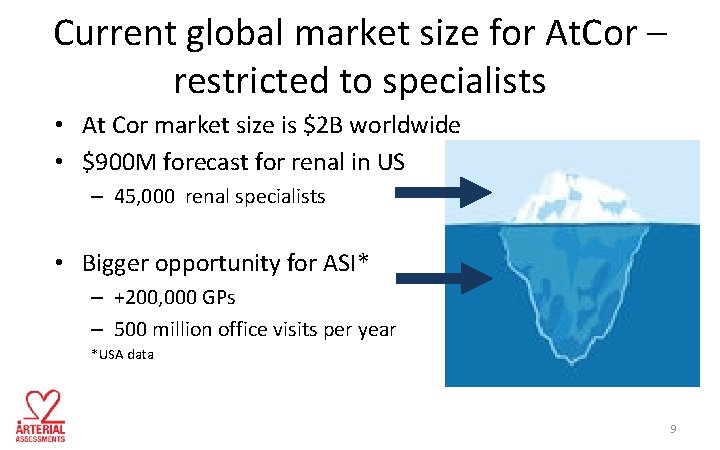
Current global market size for At. Cor – restricted to specialists • At Cor market size is $2 B worldwide • $900 M forecast for renal in US – 45, 000 renal specialists • Bigger opportunity for ASI* – +200, 000 GPs – 500 million office visits per year *USA data 9

US Market Emerging • American Heart Association endorses measuring for arterial stiffness • AMA Category 1 CPT code 9300 X 1 for $54 – “We re-emphasize our view that ACG is significantly de -risked following the AMA’s initial acceptance of the RPAs’ Category 1 CPT Code application” • 4 major pharma companies have drugs in Phase 3 trials for abating plaque – Therapeutics will enter the market driving the need for cheap and easy diagnostics. 10

Market Size in China • Active reimbursement code in China (about 250 RMB per test) • Arterial Stiffness devices are already sold into China (60% is OMRON) • More market will be available if cost and technical requirements are reduced – Healthy China 2030 – Community Hospitals and clinics 11

Go to Market Timeframe • 2020 – Secure US FDA – Secure early customers in China (can sell in some areas of China with US FDA). • 2021 – Secure China FDA to scale in China – Pursue US market 12
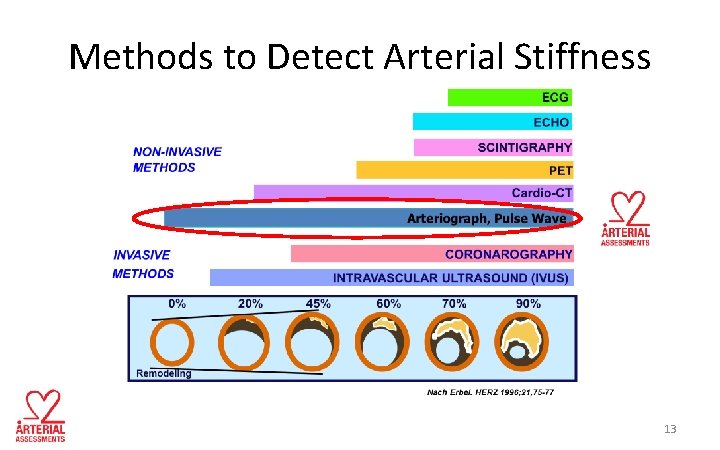
Methods to Detect Arterial Stiffness 13
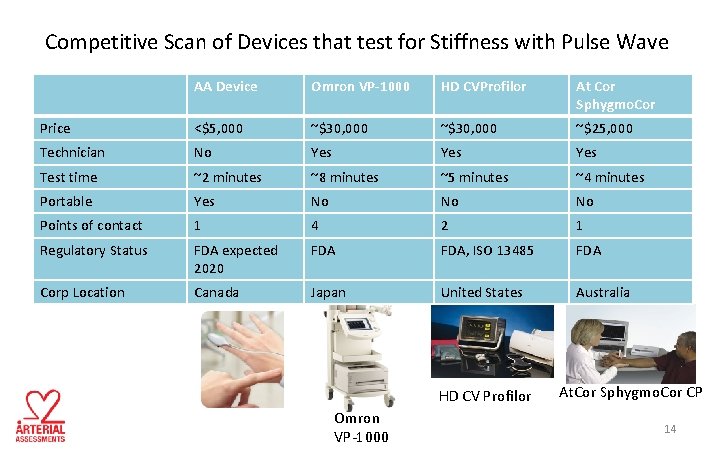
Competitive Scan of Devices that test for Stiffness with Pulse Wave AA Device Omron VP-1000 HD CVProfilor At Cor Sphygmo. Cor Price <$5, 000 ~$30, 000 ~$25, 000 Technician No Yes Yes Test time ~2 minutes ~8 minutes ~5 minutes ~4 minutes Portable Yes No No No Points of contact 1 4 2 1 Regulatory Status FDA expected 2020 FDA, ISO 13485 FDA Corp Location Canada Japan United States Australia HD CV Profilor Omron VP-1000 At. Cor Sphygmo. Cor CP 14

Team • Marshall Ring, CEO – Built and grew technology incubator to one of most recognized in Canada • Dr. Michael Zhang – Scientific founder, recognized international speaker with chronic disease management • Marc Caron, VP Product – Systems Architect, clients currently include Twitter, Nike, American Express. 15
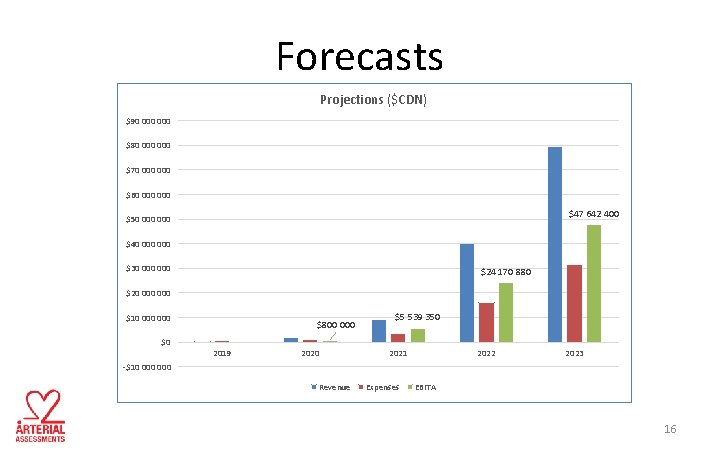
Forecasts Projections ($CDN) $90 000 $80 000 $70 000 $60 000 $47 642 400 $50 000 $40 000 $30 000 $24 170 880 $20 000 $10 000 $0 $800 000 2019 2020 $5 539 350 2021 2022 2023 -$10 000 Revenue Expenses EBITA 16

Researchers and Supporters 17

Major Accomplishments – Technology • Correlation to existing vascular elasticity devices, including At. Cor and Meridian • US FDA clinical trials completed May 2018 successful. • FDA expected 2020. • China FDA expected 2021. 18

Major Accomplishments – Qualified Customer Prospects in China • Meinian One Health +200 clinics in China – Value of this customer is over $100 M CDN annually. • Dr. Wang, PLA 301 – “This device should be used across China” • Wellness Institutes – currently using it 19

Major Accomplishments – Investment Partners in China • Hefei Goaxin VC fund has invested $1 M CDN • Canadian investors have invested $400, 000 CDN into the China company. 20
- Slides: 20