We now understand crystal structure and how to


































- Slides: 41

We now understand crystal structure and how to measure it We now move into bigger condensed matter questions. First up: What holds a crystal together? (A hot topic in early CMP)

We now understand crystal structure and how to measure it We now move into bigger condensed matter questions. First up: What holds a crystal together? (A hot topic in early CMP) Generalization to nonzero temps (Helmholtz free en(V, T)) gives eq. thermodynamics

We now understand crystal structure and how to measure it We now move into bigger condensed matter questions. First up: What holds a crystal together? (A hot topic in early CMP) Generalization to nonzero temps (Helmholtz free en(V, T)) gives eq. thermodynamics Mostly electrostatics of the outer electrons, which can describe the form of materials.

Today’s Objectives By the end of this section you should be able to: p Use a simple harmonic oscillator model to understand attractive forces in noble gases p Apply a van der Waals-London interaction

Today’s Objectives By the end of this section you should be able to: p Use a simple harmonic oscillator model to understand attractive forces in noble gases p Apply a van der Waals-London interaction p Use the Lennard-Jones potential to understand equilibrium lattice parameters (noble gases)

Today’s Objectives By the end of this section you should be able to: p Use a simple harmonic oscillator model to understand attractive forces in noble gases p Apply a van der Waals-London interaction p Use the Lennard-Jones potential to understand equilibrium lattice parameters (noble gases) p Simply by looking at the periodic table, begin to explain why other types of bonding might

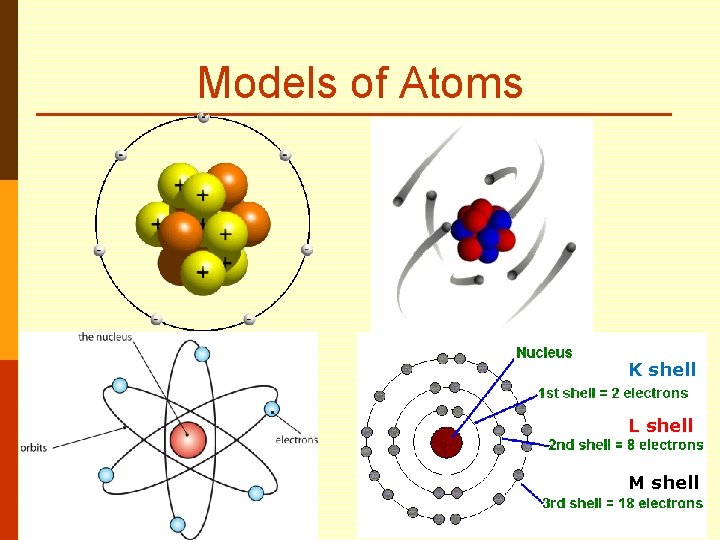
Models of Atoms K shell L shell M shell

Organization of Periodic Table Dmitri Mandeleev Columns: groups with similar shells, similar properties Rows: periods with elements with increasingly-full shells

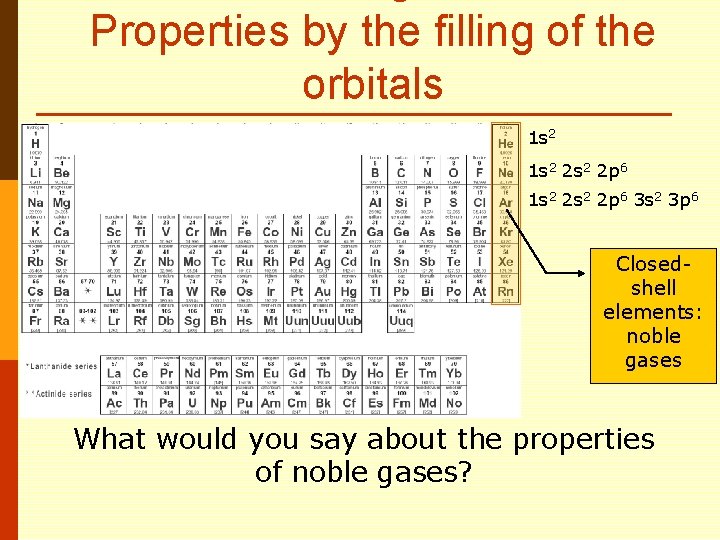
Properties by the filling of the orbitals 1 s 2 2 s 2 2 p 6 1 s 2 2 p 6 3 s 2 3 p 6 Closedshell elements: noble gases What would you say about the properties of noble gases?

Properties by the filling of the orbitals 1 s 2 2 s 2 2 p 6 1 s 2 2 p 6 3 s 2 3 p 6 Closedshell elements: noble gases What would you say about the properties of noble gases? What causes these elements to stick together?

van der Waals Bond p Arise from charge fluctuations in atoms due to zero-point motion (due to Heisenberg uncertainty principle); these create dipole moments that are attractive graphite

van der Waals Bond Arise from charge fluctuations in atoms due to zero-point motion (due to Heisenberg uncertainty principle); these create dipole moments that are attractive p Depends on p 2/r 6, short ranged p graphite

van der Waals Bond p p Arise from charge fluctuations in atoms due to zero-point motion (due to Heisenberg uncertainty principle); these create dipole moments that are attractive Depends on p 2/r 6, short ranged Typical strength of 0. 2 e. V/atom ~1% of other bonds Always present, but significant only when other types of bonding not possible (closed electron shells, saturated molecules) graphite

van der Waals Bond p p p Arise from charge fluctuations in atoms due to zero-point motion (due to Heisenberg uncertainty principle); these create dipole moments that are attractive Depends on p 2/r 6, short ranged Typical strength of 0. 2 e. V/atom ~1% of other bonds Always present, but significant only when other types of bonding not possible (closed electron shells, saturated molecules) What do weak bonds mean in terms of properties? graphite

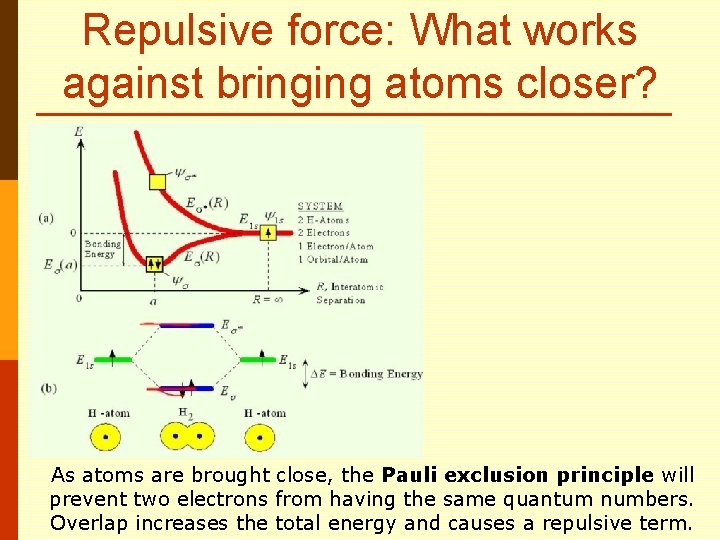
Repulsive force: What works against bringing atoms closer?

Repulsive force: What works against bringing atoms closer? As atoms are brought close, the Pauli exclusion principle will prevent two electrons from having the same quantum numbers. Overlap increases the total energy and causes a repulsive term.

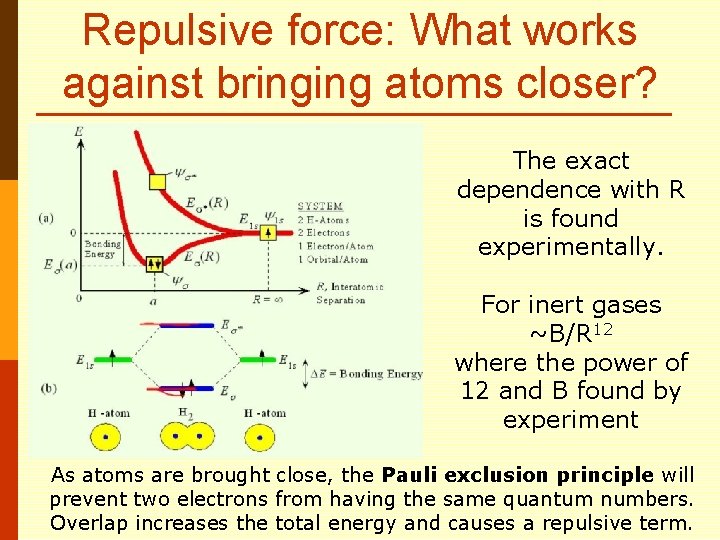
Repulsive force: What works against bringing atoms closer? The exact dependence with R is found experimentally. For inert gases ~B/R 12 where the power of 12 and B found by experiment As atoms are brought close, the Pauli exclusion principle will prevent two electrons from having the same quantum numbers. Overlap increases the total energy and causes a repulsive term.

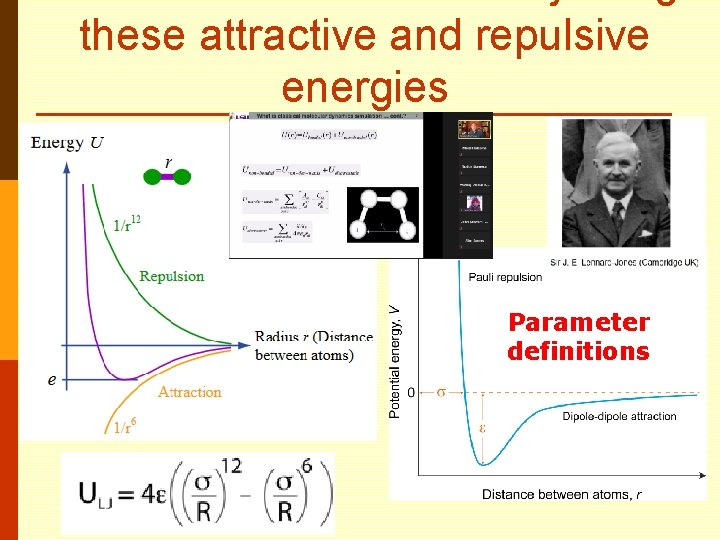
these attractive and repulsive energies Parameter definitions

What about the properties of non-noble gases? Closedshell elements: noble gases

Consider the case of sodium (atomic # 11) p On your own write down the electronic configuration: 1 s 2…

Consider the case of sodium (atomic # 11) p On your own write down the electronic configuration: 1 s 2…

Consider the case of sodium (atomic # 11) p On your own write down the electronic configuration: 1 s 2… Note: 4 s before 3 d

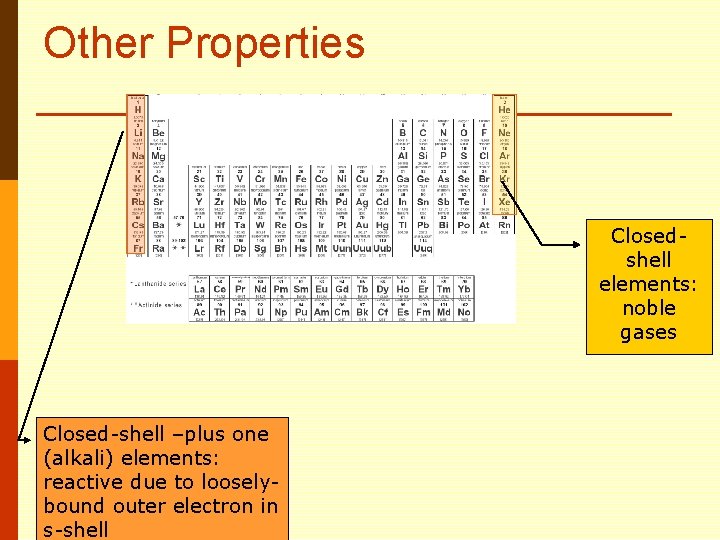
Other Properties Closedshell elements: noble gases Closed-shell –plus one (alkali) elements: reactive due to looselybound outer electron in s-shell

Why mobile electrons appear in some solids and not others? p According to the very simple Drude model, the valance electrons are responsible for the conduction of electricity, thus termed conduction electrons. p Na 11 → 1 s 2 2 p 6 3 s 1 Core electrons Valance electron (loosely bound)

Why mobile electrons appear in some solids and not others? p According to the very simple Drude model, the valance electrons are responsible for the conduction of electricity, thus termed conduction electrons. p Na 11 → 1 s 2 2 p 6 3 s 1 Valance electron (loosely bound) Core electrons p This valance electron, which occupies the third atomic shell, is the electron which is responsible chemical properties of Na.

Why mobile electrons appear in some solids and not others? p According to the very simple Drude model, the valance electrons are responsible for the conduction of electricity, thus termed conduction electrons. p Na 11 → 1 s 2 2 p 6 3 s 1 Core electrons p Valance electron (loosely bound) Metallic 11 Na, 12 Mg and 13 Al are assumed to have 1, 2 and 3 mobile electrons per atom respectively. This valance electron, which occupies the third atomic shell, is the electron which is responsible chemical properties of Na.

Terminology There exist many forms of valence! p By valence electrons here we mean outer electrons. . . the ones that could easily be pulled from the core to form an electron gas

Terminology There exist many forms of valence! p By valence electrons here we mean outer electrons. . . the ones that could easily be pulled from the core to form an electron gas p Sometimes people talk about different valence states of an element

Terminology There exist many forms of valence! p By valence electrons here we mean outer electrons. . . the ones that could easily be pulled from the core to form an electron gas (In chemistry) Valence = Maximum number of bonds formed by atom

Terminology There exist many forms of valence! p By valence electrons here we mean outer electrons. . . the ones that could easily be pulled from the core to form an electron gas (In chemistry) Valence = Maximum number of bonds formed by atom Where would these definitions differ?

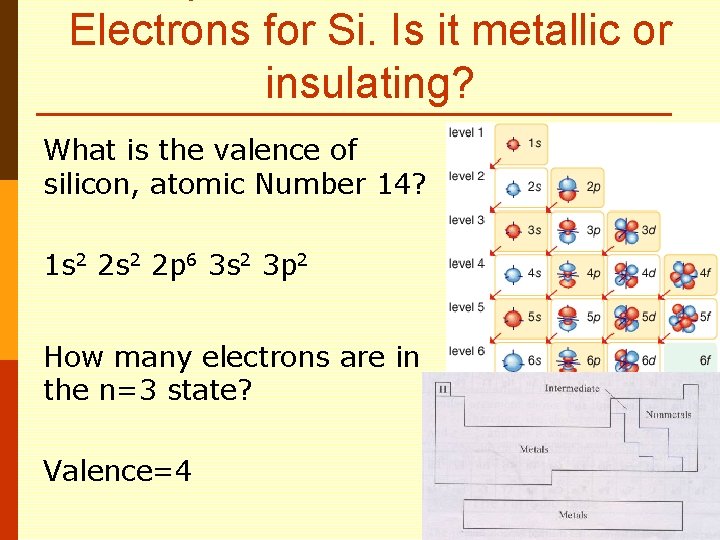
Electrons for Si. Is it metallic or insulating? What is the valence of silicon, atomic Number 14?

Electrons for Si. Is it metallic or insulating? What is the valence of silicon, atomic Number 14?

Electrons for Si. Is it metallic or insulating? What is the valence of silicon, atomic Number 14? 1 s 2 2 p 6 3 s 2 3 p 2

Electrons for Si. Is it metallic or insulating? What is the valence of silicon, atomic Number 14? 1 s 2 2 p 6 3 s 2 3 p 2 How many electrons are in the n=3 state?

Electrons for Si. Is it metallic or insulating? What is the valence of silicon, atomic Number 14? 1 s 2 2 p 6 3 s 2 3 p 2 How many electrons are in the n=3 state? Valence=4

Group: Find the Valence of Fe (26) What is the valence of iron, atomic Number 26?

Group: Find the Valence of Fe (26) What is the valence of iron, atomic Number 26?

Group: Find the Valence of Fe (26) What is the valence of iron, atomic Number 26? 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 Number of 3 d electrons = 6

Group: Find the Valence of Fe (26) What is the valence of iron, atomic Number 26? 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 Number of 3 d electrons = 6 Except, are these the outer most? No, the 4 s is farther! So elemental valence =2!

Group: Find the Valence of Fe (26) What is the valence of iron, atomic Number 26? 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 Number of 3 d electrons = 6 What about in compounds? (Ba. Fe. O 3) Except, are these the outer most? No, the 4 s is farther! So elemental valence =2!

Valence often confused/used for oxidation state Let’s determine the type, valence and oxidation states of the atoms and ions in the material Ba. Fe. O 3. Anion = Negative ion (atom with extra electrons) Cation = Positive ion (atom missing electrons) Which is oxygen? Warning: The "oxidation state" of an atom in a molecule gives the number of valence electrons it has gained or lost through bonding. In contrast to the valence number, the oxidation state can be positive or negative. But sometimes called valence.