We Dont Need No Stinking Badges Movie Clip

“We Don’t Need No Stinking Badges” Movie Clip Research & Discovery Grand Rounds (R&D GR)

Making Research Safer & Better: The KUMC, KUHS & RI Research Committee Structure Research & Discovery Grand Rounds (R&D GR) June 15 th, 2017 Richard J. Barohn, M. D. Chair, Department of Neurology Gertrude and Dewey Ziegler Professor of Neurology University Distinguished Professor Vice Chancellor for Research President, KUMC Research Institute Gregory A. Ator, M. D. , F. A. C. S. Associate Professor Director, Division of Otology/Neurology Director, Audiology/Vestibular Rehabilitation Laboratory
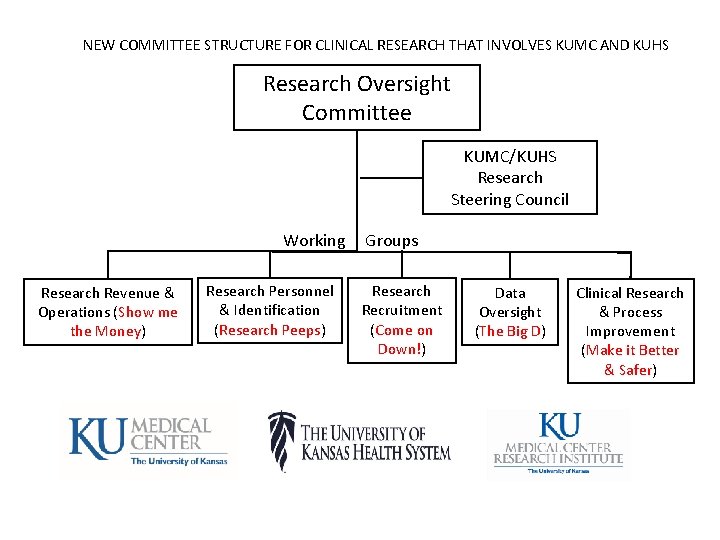
NEW COMMITTEE STRUCTURE FOR CLINICAL RESEARCH THAT INVOLVES KUMC AND KUHS Research Oversight Committee KUMC/KUHS Research Steering Council Working Research Revenue & Operations (Show me the Money) Research Personnel & Identification (Research Peeps) Groups Research Recruitment (Come on Down!) Data Oversight (The Big D) Clinical Research & Process Improvement (Make it Better & Safer)

Research Oversight Committee • • • New: Created in the System merger 2016 5 representatives from KUMC & 5 from, KUHS Richard Barohn serves as Chair Meets every other month Steering Committees & Subcommittee's report to ROC Final Decision Making Body on KUMC/KUHS/RI research issues when needed Members: University Membership: Barohn, Richard, M. D. (Chair) Girod, Doug, MD Jensen, Roy, MD Simari, Robert, MD Weiner, Carl, MD Hospital Membership: Marting, Bill Lasack, Colette Peters, Dan Ruder, Chris Member TBD Ex-officio: Ator, Greg, MD Caldwell, Jamie Gibbens, Kim Hansen, Chris Hoebelheinrich, Lisa Stites, Steven, MD Sokol, Amy

KUMC/KUHS/RI Research Steering Council • • Richard Barohn, MD & Greg Ator, MD, Co-Chairs Established 2014, Before ROC 30 representatives from KUMC faculty and staff, RI & KUHS Want to add more investigators and coordinators – possibly one from each department Charges: • Inform other working groups of activities • Inform investigators and coordinators from clinical departments about activities • Review work of working groups and make suggestions, especially before issues go to ROC • Meets every other month that ROC is not meeting Members: Greg Ator (Steering Committee co-chair) Rick Barohn (Steering Committee co-chair) Debra Brogden (Committee Support) Sandy Billinger Karen Blackwell Marge Bott Jamie Caldwell Chad Cannon Ethan Carter Carol Cleek Mazen Dimachkie Hilary Dubinsky Ed Ellerbeck Bill Marting Peter Griffith Joseph Mc. Guirk Chris Hansen Tamara Mc. Mahon Lisa Hoebelheinrich Andrea Nicol Colette Lasack Dinesh Pal Lisa Mische Lawson Mudaranthakam (FARC Chair) Matt Mayo and/or Byron Jeff Reene Michael Rippee Gajewski David Robbins Steve Simpson Peter Smith Jim Vacek Russ Waitman Dawn Walters Susanne Welborn Jennifer Williams Steve Williamson Jeff Wright
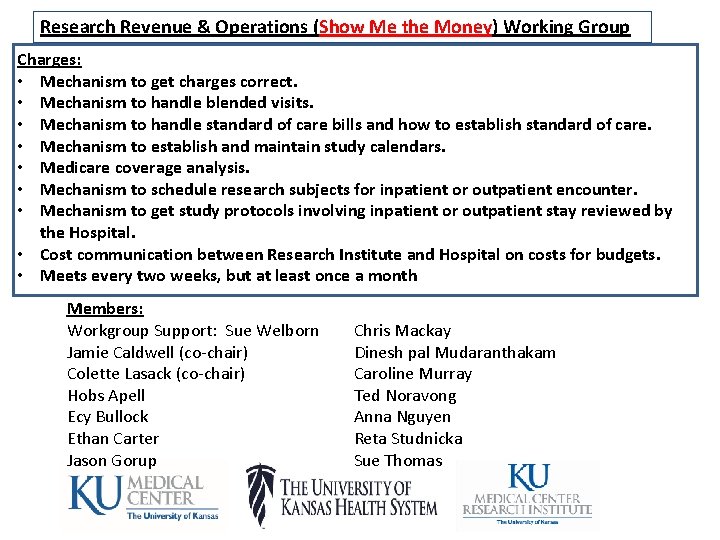
Research Revenue & Operations (Show Me the Money) Working Group Charges: • Mechanism to get charges correct. • Mechanism to handle blended visits. • Mechanism to handle standard of care bills and how to establish standard of care. • Mechanism to establish and maintain study calendars. • Medicare coverage analysis. • Mechanism to schedule research subjects for inpatient or outpatient encounter. • Mechanism to get study protocols involving inpatient or outpatient stay reviewed by the Hospital. • Cost communication between Research Institute and Hospital on costs for budgets. • Meets every two weeks, but at least once a month Members: Workgroup Support: Sue Welborn Jamie Caldwell (co-chair) Colette Lasack (co-chair) Hobs Apell Ecy Bullock Ethan Carter Jason Gorup Chris Mackay Dinesh pal Mudaranthakam Caroline Murray Ted Noravong Anna Nguyen Reta Studnicka Sue Thomas

Research Revenue & Operations (Show Me the Money) Working Group Three Major Activities Financial Epic go live , Research DNB avg $18 million, release days averaged 60 days. Current – Research DNB avg $2 million, release days average less than 15 days. 2016 - Outstanding study balances average $2. 5 million Current – Outstanding study balances under $1 million Coverage Analysis Developed a coverage analysis guideline and added 2 Clinical Trial Financial Analysts and Nurse Auditor. Continue to grow these positions and increase their knowledge and education. Have identified and confirmed additional solutions in non-qualifying studies and compliant way of getting what appears to be a non-qualified study, qualified. Work closely with RI budgets and contracts team to establish qualifying status, appropriateness of soc services, handle pricing requests, and create billing matrices used in budgeting and compliant billing. Work continues to grow the process and continue education for the team members. Nursing Impact Formerly reviewed only nursing/clinical impact for studies with Inpatient services. Currently reviewing all studies with health system services to ensure ancillary departments is aware of the study/protocol, have appropriate resources, and can safely provide the services as required by the protocol. Work continues to make this process efficient.
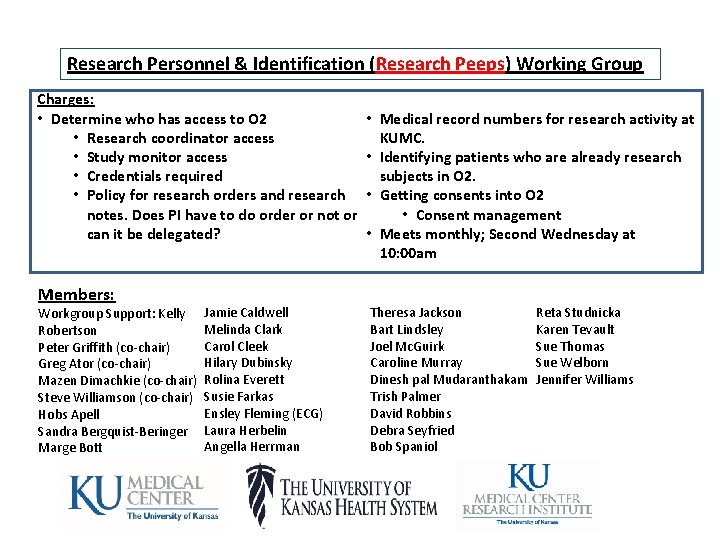
Research Personnel & Identification (Research Peeps) Working Group Charges: • Determine who has access to O 2 • Research coordinator access • Study monitor access • Credentials required • Policy for research orders and research notes. Does PI have to do order or not or can it be delegated? Members: Workgroup Support: Kelly Robertson Peter Griffith (co-chair) Greg Ator (co-chair) Mazen Dimachkie (co-chair) Steve Williamson (co-chair) Hobs Apell Sandra Bergquist-Beringer Marge Bott Jamie Caldwell Melinda Clark Carol Cleek Hilary Dubinsky Rolina Everett Susie Farkas Ensley Fleming (ECG) Laura Herbelin Angella Herrman • Medical record numbers for research activity at KUMC. • Identifying patients who are already research subjects in O 2. • Getting consents into O 2 • Consent management • Meets monthly; Second Wednesday at 10: 00 am Theresa Jackson Bart Lindsley Joel Mc. Guirk Caroline Murray Dinesh pal Mudaranthakam Trish Palmer David Robbins Debra Seyfried Bob Spaniol Reta Studnicka Karen Tevault Sue Thomas Sue Welborn Jennifer Williams

Research Personnel & Identification (Research Peeps) Working Group Ongoing Work: • Advocate Epic Care. Link tool for Study Monitor (nonemployee) Access • Timetable: Piloting this summer • Rationale: • Study source data in the EMR (formerly required a site visit) which reduces or eliminates: • Scarce coordinator time devoted to uncompensated service to sponsors • Use of unsecure work -arounds such as printing, over-theshoulder glimpses, or universal access Future: • Adopt new Security Approach for Clinical Research Study Staff • Timetable: Active rollout now • Rationale: • Reform “Clinical Heritage” security template use (Ex. Former hospital employee transfers to university with hospital related security profile) • Replace proliferating and unmanageable individual security templates • Allows for building out the research feature set of O 2 – more to come

Research Recruitment (Come on Down!) Working Group Charges: • Link Pioneers and Frontiers registries to EMR. • • Putting inclusion/exclusion criteria in O 2. • Figure out how to integrate Velos and • Pioneers with O 2. • Determine ways to find perspective subjects • from EMR for research studies. Members: Workgroup Support: Debra Brogden *Tamara Mc. Mahon (cochair) Dinesh pal Mudaranthakam (co-chair) Bhargav Adagarla Hobs Apell Greg Ator, MD Hilary Dubinsky Susie Farkas Peter Griffith Angella Herrman Matt Mayo Joel Mc. Guirk Kevin Smilor Carol Smith Bob Spaniol Determine efficient ways to refer subjects for research projects to research teams. Distribute build - putting outcomes forms in EMR Meets quarterly; will meet monthly when CRIO arrives Terry Tsue Jeff Wright Amy Sokol *will be replaced by future CRIO hire

Research Recruitment (Come on Down!) Working Group Previous Work • Worked with IRB to determine best approach to obtain inclusion and exclusion criteria for studies • The working group decided on clincialtrials. gov as the source for the inclusion and exclusion criteria available in Pioneers. • Worked with HITS team to review the use of Epic alerts for recruitment at other institutions • Higher impact with My. Chart versus alerts for patient recruitment • Medical Informatics is working to obtain distributed build access to add a research tab and a Pioneers link to My. Chart. • HSC will review consent prior to adding to My. Chart. • Explored the use of problem list SNOWMED codes to tag studies in both Pioneers and Velos Next steps • New Chief Research Informatics Officer (CRIO), Tamara Winden, joins July 17, 2017 • Continue work to integrate Frontiers and Pioneers into My. Chart once distributed build access is available • Further working group discussions will focus on linking the problem list codes to trials and subsequently making this information available in O 2.

Data Oversight (The Big D) Working Group Charges: • Joint oversight of clinical data being used for • research. • Ensuring proper IT and legal infrastructure is • in place to support research needs of investigators and protect Health System and • KUMC. Members: Greg Ator (co-chair) Rick Barohn (co-chair) Lisa Hoebelheinrich (vice co-chair) Dan Peters Amy Sokol Bob Spaniol Juli Wessel Develop policies and agreements to accomplish this. Assuring adequate plans are in place for security of research data Meets monthly

Data Oversight (The Big D) Working Group Activities: • Identifying and alleviating barriers to the development of researchrelated data sharing infrastructure • Supporting implementation of research-related data sharing initiatives by assuring that applicable regulatory requirements are met • Updating existing University/Health System data use agreements to provide for more efficient project start-up and oversight Ongoing Work: • Clearing the way for the Frontiers/IDX transition • Work with the EPIC infrastructure • Update informatics and data use agreements

Clinical Research & Process Improvement (Make it Better & Safer) Working Group • Charges: • Establish process for monitoring of clinical trials • • Establish and disseminate SOPs to manage clinical trials • • Recommend policies for research compliance in clinical setting Members: Carol Cleek (co-chair) Lisa Hoebelheinrich (co-chair) Hobs Apell Greg Ator (Ex-Officio) Rick Barohn (Ex-Officio) Hilary Dubinsky Establish training policies and programs for clinical research Identify and recommend strategies to reduce compliance risk Meets monthly Mary Hindle Monica Lubek Kevin Smilor Amy Sokol Kyle Stephens Amber Styles

Clinical Research & Process Improvement (Make it Better & Safer) Working Group Primary work thus far has focused on establishing and disseminating SOPs to manage clinical trials • Phase 1 SOPs are posted on the KUMC intranet (search “clinical research standard operating procedures”) • Phase 1 SOPs address such things as study team responsibilities, Good Clinical Practice and study documentation, sponsor/monitor visits and FDA audits • They are not intended to take the place of departmental SOPs that are needed to assure effective and consistent functioning at the department level. Phase 2 SOPs are currently in development. They are intended to provide guidance and streamline processes in areas such as: • Research record management, and what research records should be included in O 2. • Health System research review • Single patient INDs, expanded access and compassionate use • Use of sponsor provided equipment within the Health System

Making Research Safer & Better: The KUMC/KUHS/RI Committee Structure QUESTIONS?
- Slides: 16