WAYS ENERGY CAN BE TRANSFORMED CHEMICAL REACTIONS Energy

WAYS ENERGY CAN BE TRANSFORMED

CHEMICAL REACTIONS • Energy is stored in chemical bonds • During a reaction bonds break and reform • Results in energy release or absorption between the system and its surroundings • Energy is usually thermal but might be released as light or sound
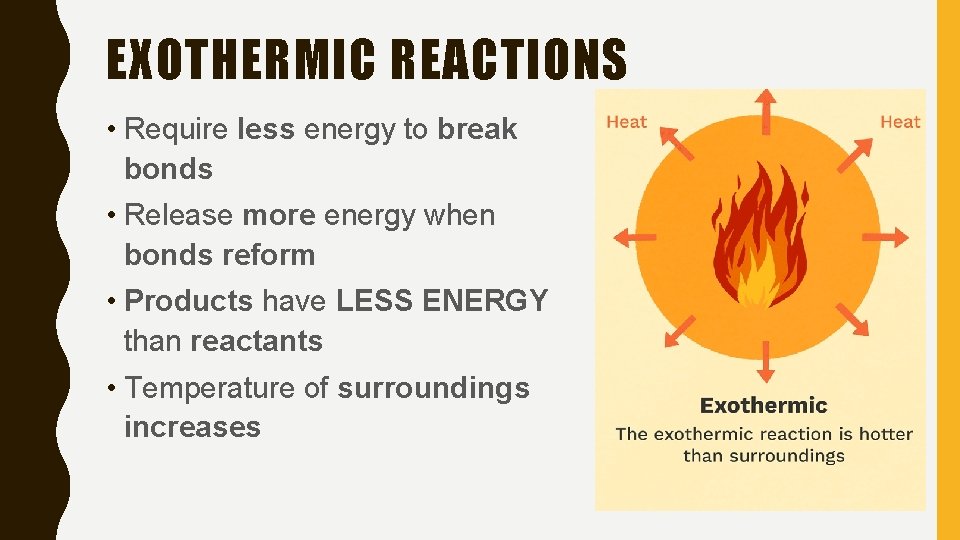
EXOTHERMIC REACTIONS • Require less energy to break bonds • Release more energy when bonds reform • Products have LESS ENERGY than reactants • Temperature of surroundings increases

EXAMPLES

CELLULAR RESPIRATION • An EXOTHERMIC process • Glucose reacts with oxygen to form carbon dioxide, water and ENERGY!

FOSSIL FUELS & FUEL CELLS • Chemical potential energy in living things came from solar energy (the sun) • Ancient remains of plants and animals buried and converted into fossil fuels • Includes oil, coal and natural gas
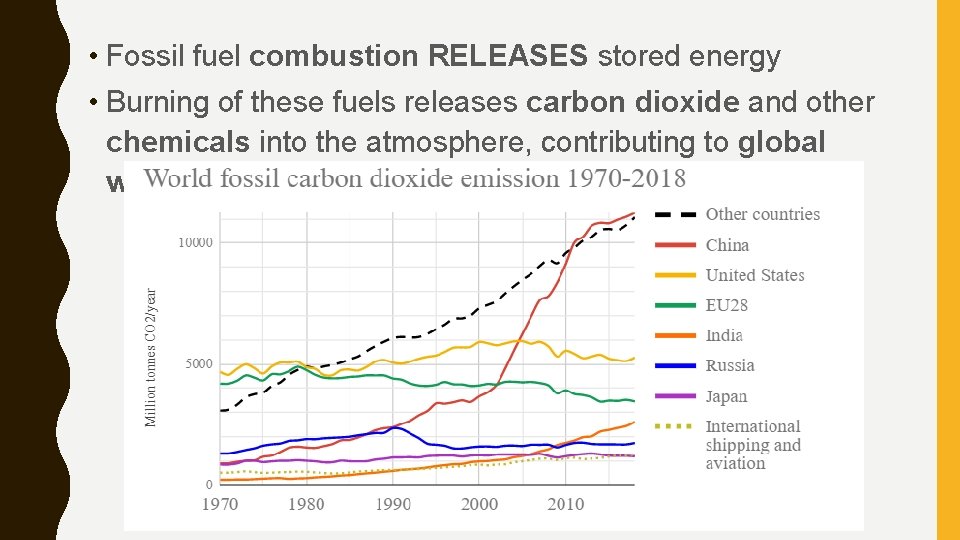
• Fossil fuel combustion RELEASES stored energy • Burning of these fuels releases carbon dioxide and other chemicals into the atmosphere, contributing to global warming
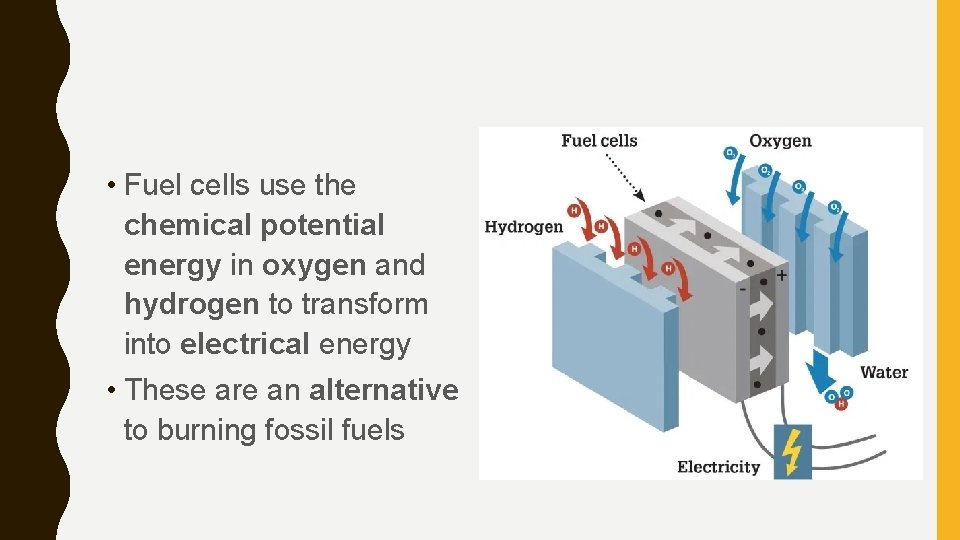
• Fuel cells use the chemical potential energy in oxygen and hydrogen to transform into electrical energy • These are an alternative to burning fossil fuels
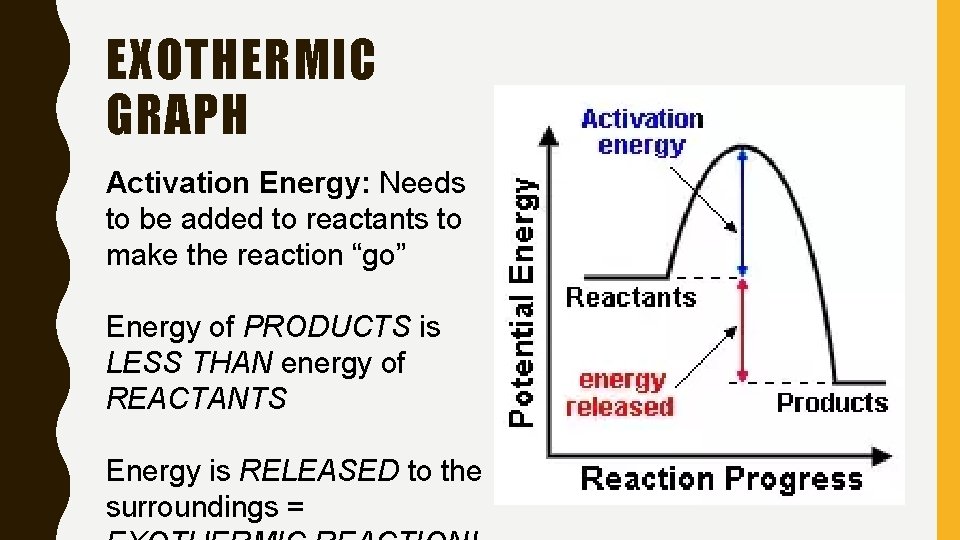
EXOTHERMIC GRAPH Activation Energy: Needs to be added to reactants to make the reaction “go” Energy of PRODUCTS is LESS THAN energy of REACTANTS Energy is RELEASED to the surroundings =

ENDOTHERMIC REACTIONS • Need a large amount of energy to break bonds • Release less energy when bonds reform • Products have MORE chemical potential energy than the reactants • Temperature of the surroundings decreases
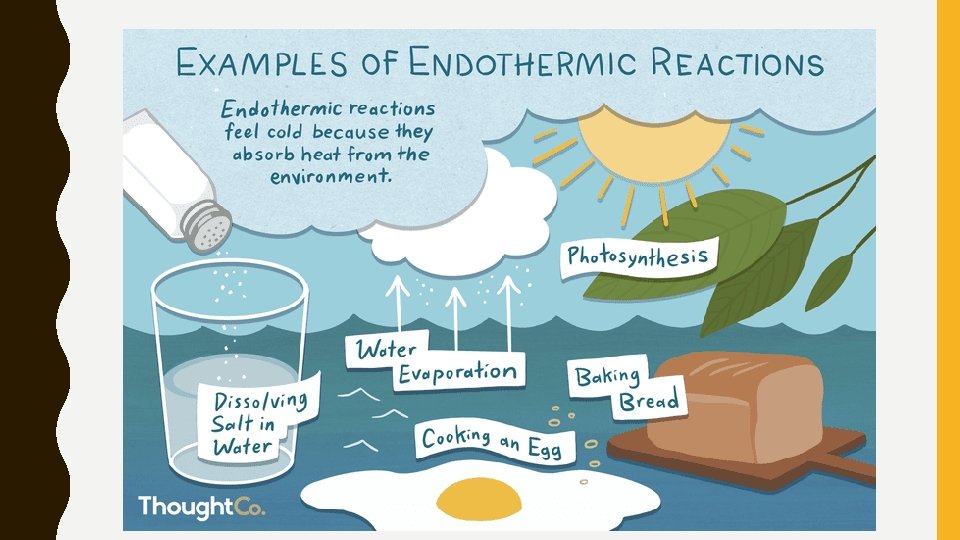
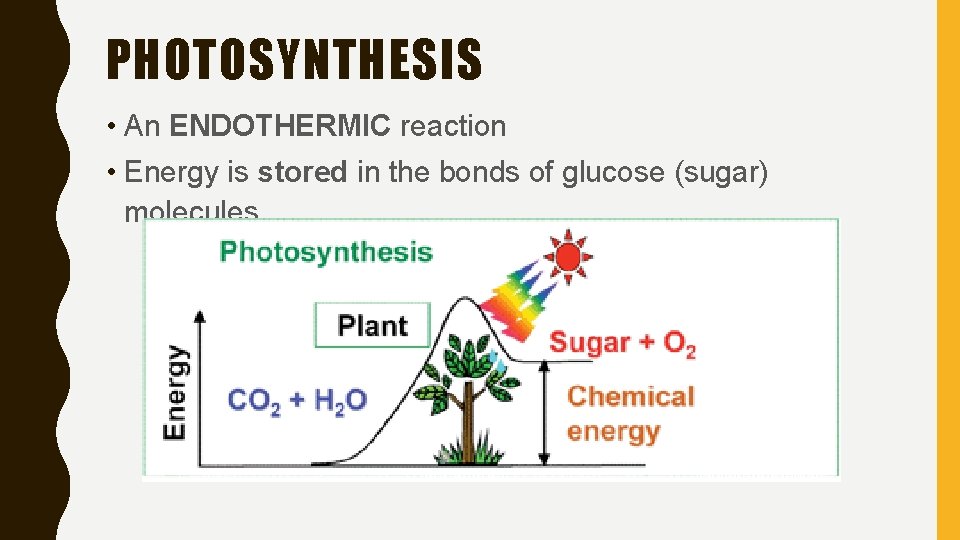
PHOTOSYNTHESIS • An ENDOTHERMIC reaction • Energy is stored in the bonds of glucose (sugar) molecules
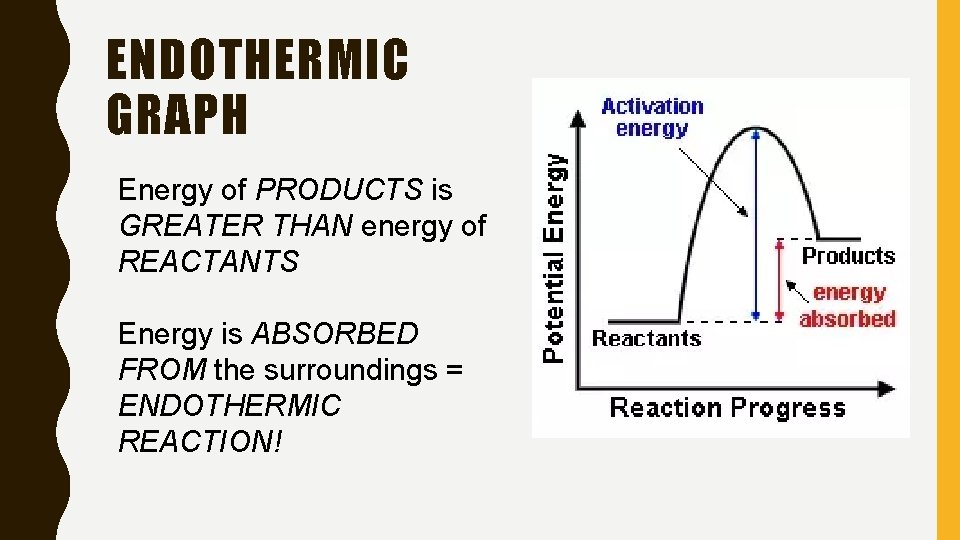
ENDOTHERMIC GRAPH Energy of PRODUCTS is GREATER THAN energy of REACTANTS Energy is ABSORBED FROM the surroundings = ENDOTHERMIC REACTION!

LAB ACTIVITIES- EXOTHERMIC VS ENDOTHERMIC REACTIONS The lab activities today should emphasize the difference between exothermic and endothermic reactions! …There also a few easy workbook questions to help you review the lesson from today. I will write these on the board
- Slides: 14