Waveparticle duality Quantum mechanics Prof Marko B Popovic

Wave-particle duality. Quantum mechanics Prof. Marko B Popovic

Particle wave duality As you can learn in depth if you take Modern Physics PH 1130, “things” can have dual personalities also referred to as the principle of complementarity. What you assume is a wave can also behave as a particles Example electromagnetic wave and massless particles called photons What you assume is a particle can also behave as a wave More on this today, especially in the context of De Broglie relationship
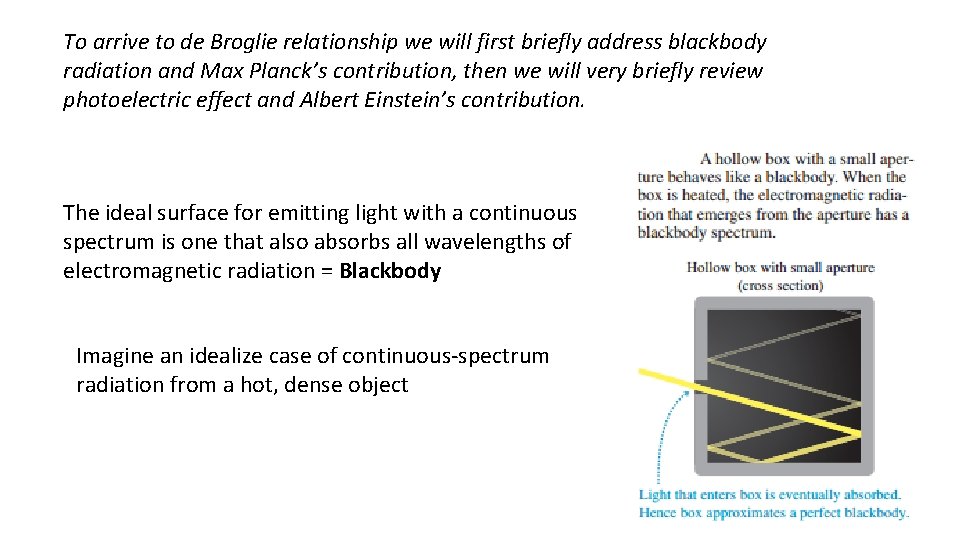
To arrive to de Broglie relationship we will first briefly address blackbody radiation and Max Planck’s contribution, then we will very briefly review photoelectric effect and Albert Einstein’s contribution. The ideal surface for emitting light with a continuous spectrum is one that also absorbs all wavelengths of electromagnetic radiation = Blackbody Imagine an idealize case of continuous-spectrum radiation from a hot, dense object

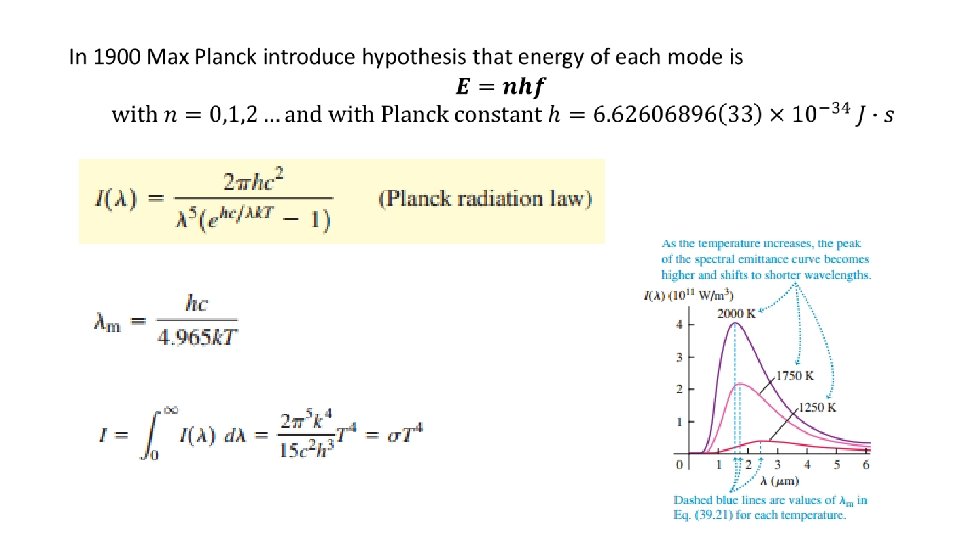
By 1900 three features of black body radiation were experimentally established: Scaling principle

However there was no good theoretical explanation of spectral emittance “ultraviolet catastrophe” Infinitely large total radiated intensity
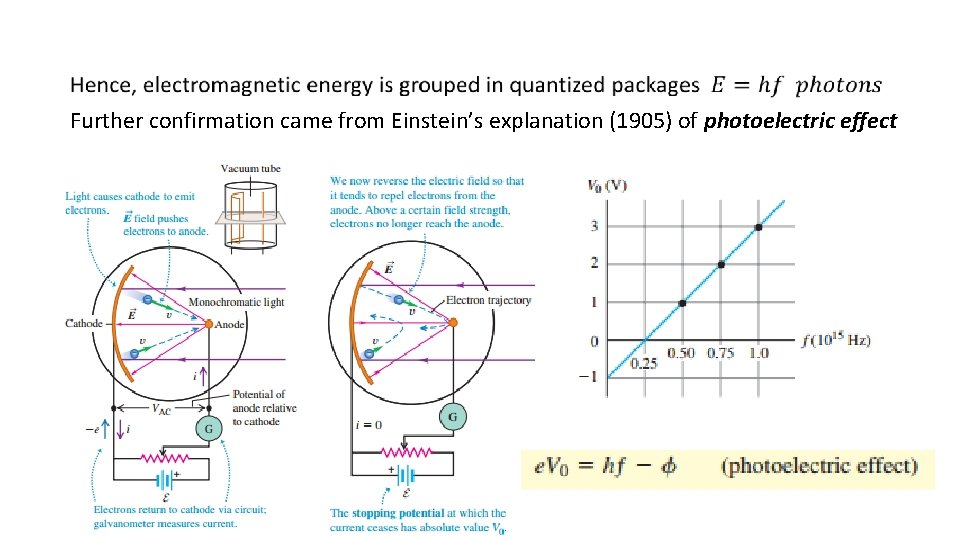
Further confirmation came from Einstein’s explanation (1905) of photoelectric effect

Same relationship for particles with mass
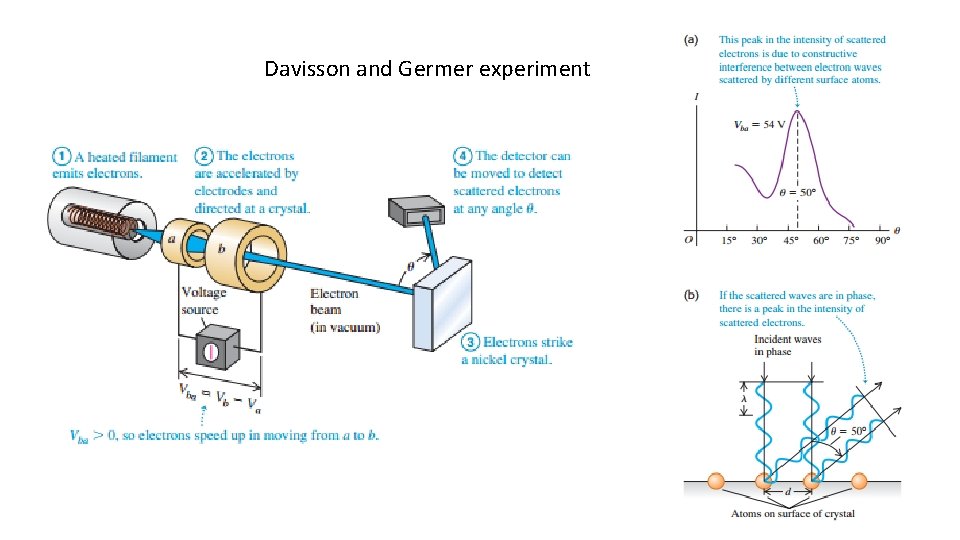
Davisson and Germer experiment
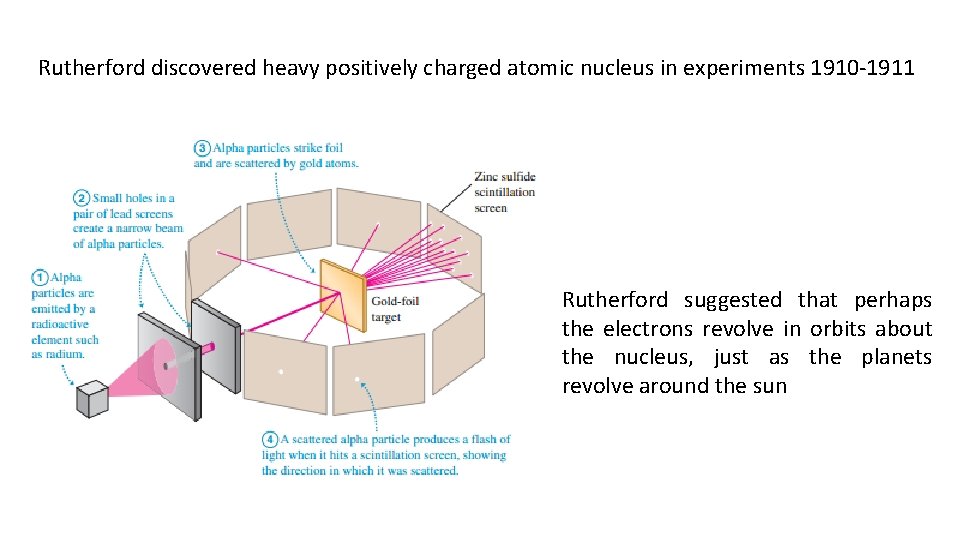
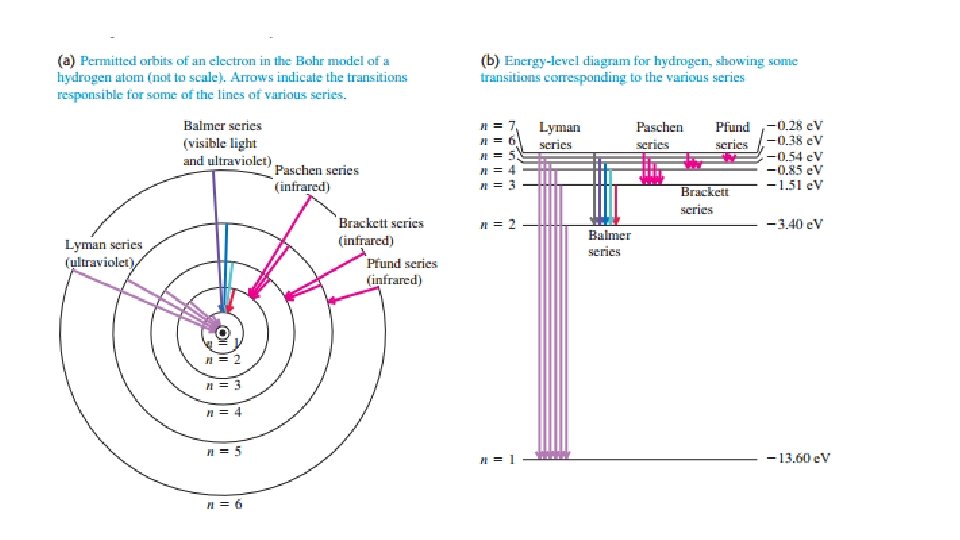
Rutherford discovered heavy positively charged atomic nucleus in experiments 1910 -1911 Rutherford suggested that perhaps the electrons revolve in orbits about the nucleus, just as the planets revolve around the sun
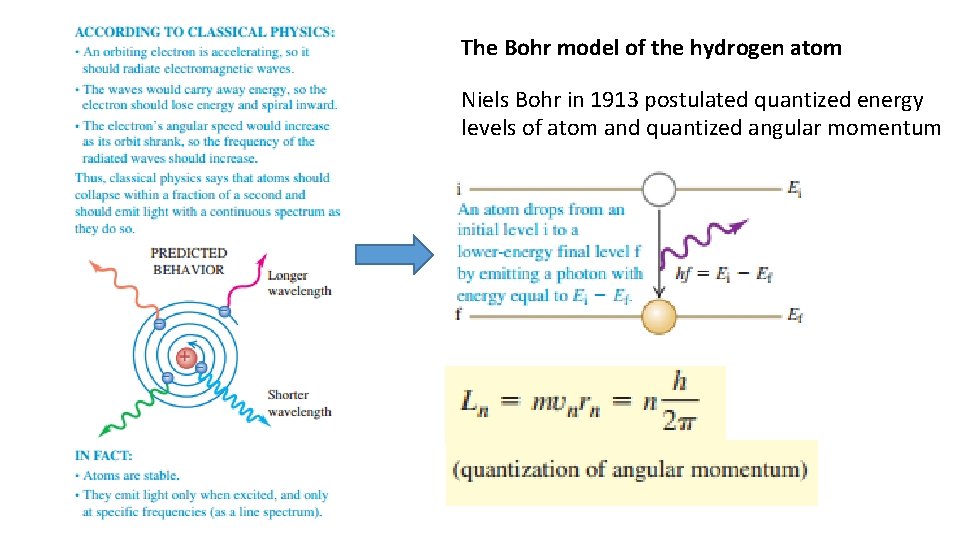
The Bohr model of the hydrogen atom Niels Bohr in 1913 postulated quantized energy levels of atom and quantized angular momentum
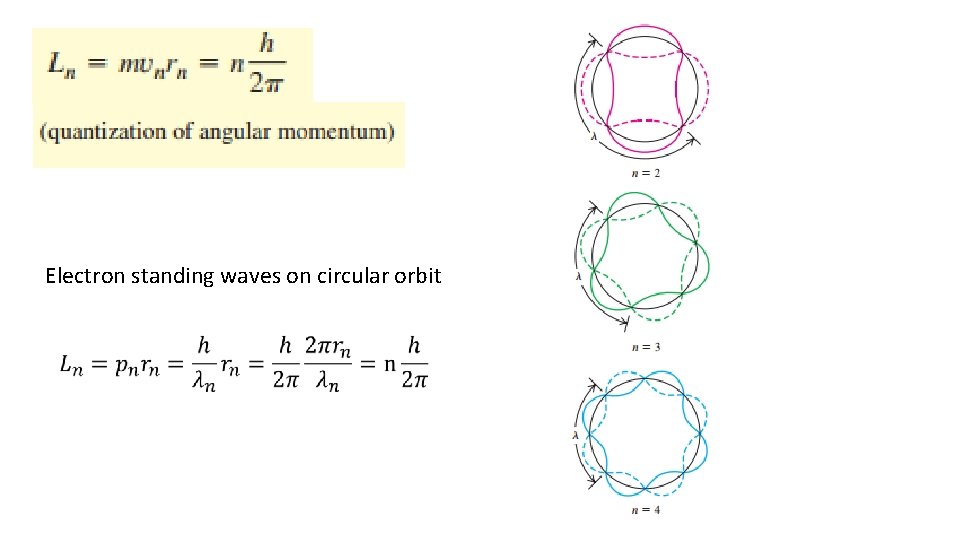
Electron standing waves on circular orbit
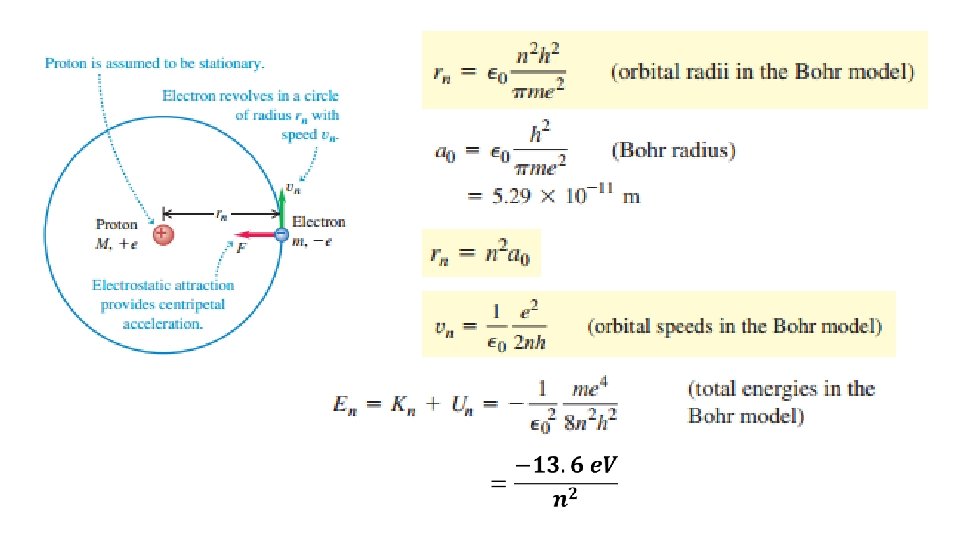
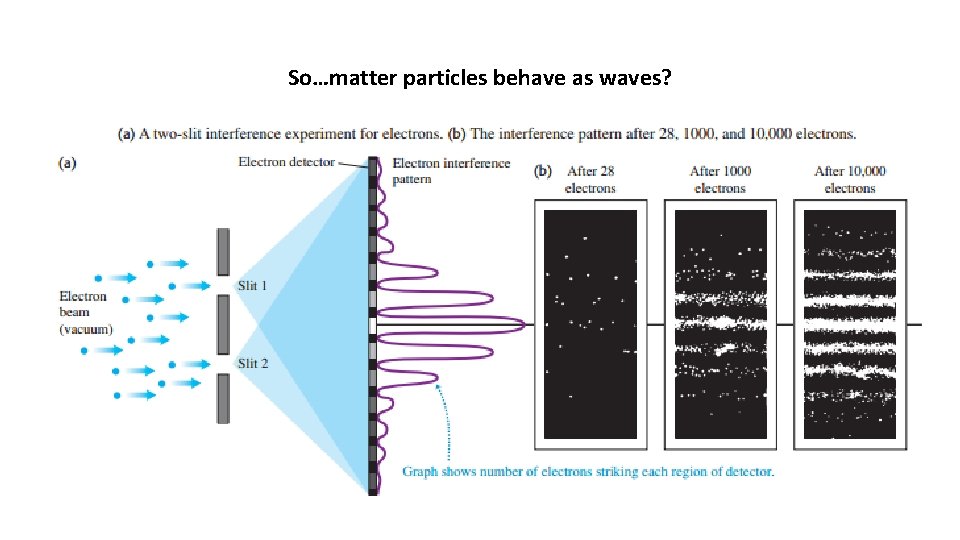
So…matter particles behave as waves?

Quantum mechanical approach

Introduce potential energy
- Slides: 17